 |
 |
 |
| |
Associations of Inflammatory Markers with AIDS and non-AIDS Clinical Events after Initiation of Antiretroviral Therapy (ART): AIDS Clinical Trials Group A5224s, a substudy of ACTG A5202
|
| |
| |
Reported by Jules Levin
IAC 2012
Grace A McComsey, Douglas Kitch, Paul E Sax, Camlin Tierney, Nasreen C Jahed, Kathleen Melbourne, Belinda Ha, Todd T Brown, Anthony Bloom, Neal Fedarko, and Eric S Daar on behalf of the ACTG A5224s team

PROGRAM ABSTRACT
Associations of inflammatory markers with AIDS and non-AIDS clinical events after initiation of antiretroviral therapy (ART): AIDS Clinical Trials Group A5224s, a substudy of ACTG A5202
Presented by Grace A McComsey (United States).
G.A. McComsey1, D. Kitch2, P.E. Sax3, C. Tierney2, N.C. Jahed4, K. Melbourne5, B. Ha6, T.T. Brown7, A. Bloom8, N. Fedarko7, E.S. Daar9, Adult AIDS Clinical Trials Group A5224s
1Case Western Reserve University, Cleveland, United States, 2Harvard School of Public Health, Boston, United States, 3Brigham and Women's Hospital and Harvard Medical School, Boston, United States, 4Social & Scientific Systems, Inc, Silver Spring, United States, 5Gilead Sciences, Foster City, United States, 6GlaxoSmithKline, Research Triangle, United States, 7Johns Hopkins, Baltimore, United States, 8Frontier Science and Technology Research Foundation, Inc., Amherst, United States, 9Los Angeles Biomedical Research Institute at Harbor-UCLA Medical Center, Torrance, United States
Background: The association of inflammatory biomarkers with clinical events after ART initiation is unclear.
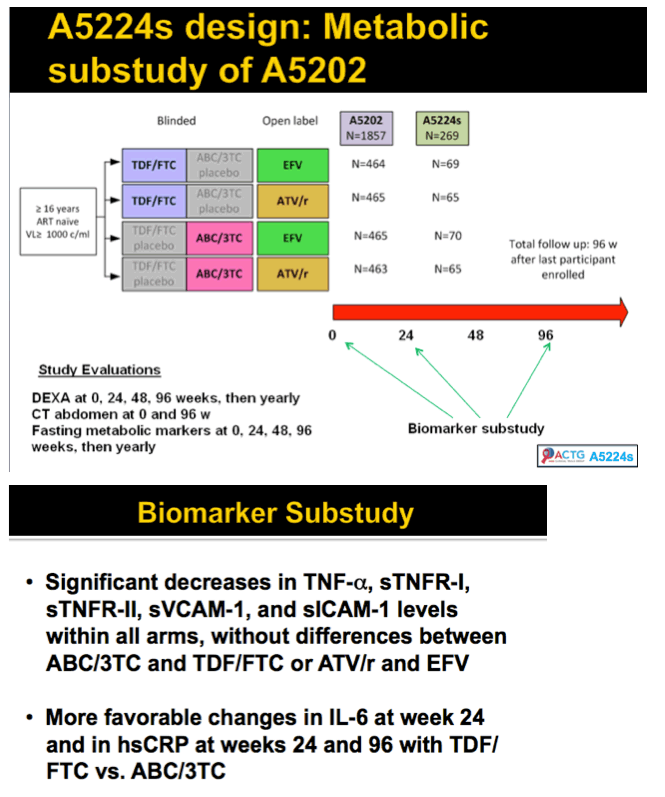
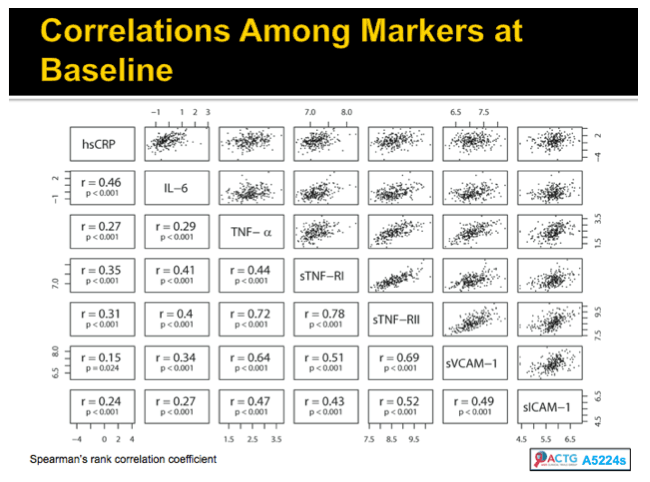
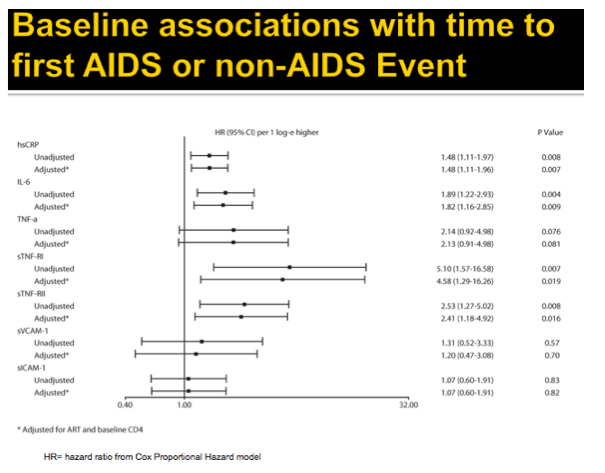
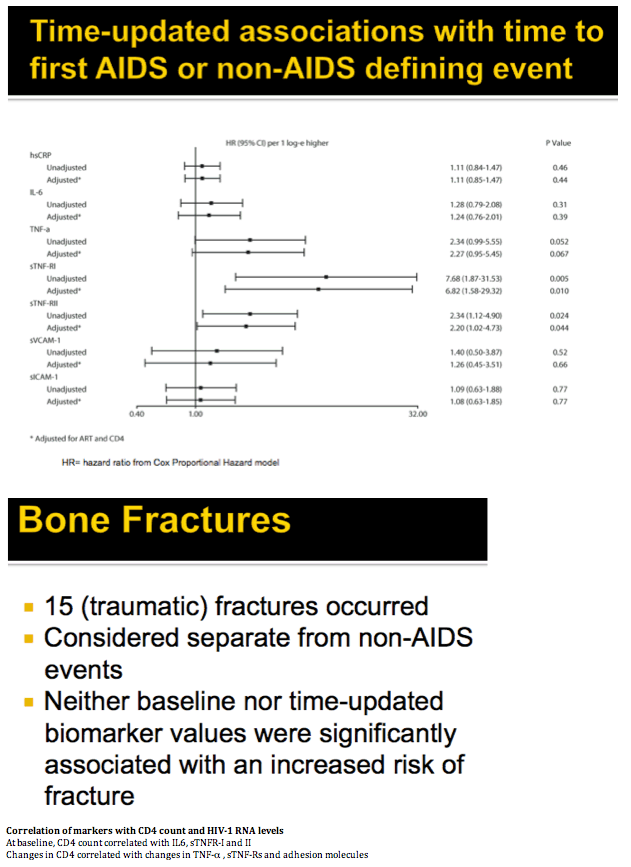
Methods: A5202 randomized 1857 treatment-naive subjects to abacavir/lamivudine or tenofovir DF/emtricitabine with efavirenz or atazanavir/ritonavir. Substudy A5224s measured inflammatory biomarkers on all substudy subjects with available plasma from baseline and weeks 24 or 96. The association of hsCRP, IL-6, sTNF-RI, sTNF-RII, TNF-a, sVCAM-1, and sICAM-1 with times to AIDS and non-AIDS defining events was analyzed with Cox proportional hazards models, with adjustment by ART assignment, and HIV-1 RNA or CD4. Time-updated analyses used the most current value.
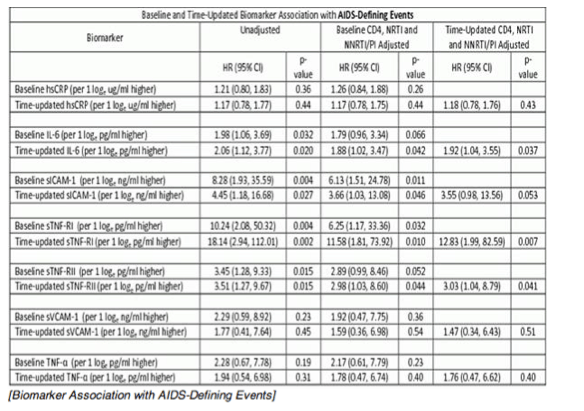
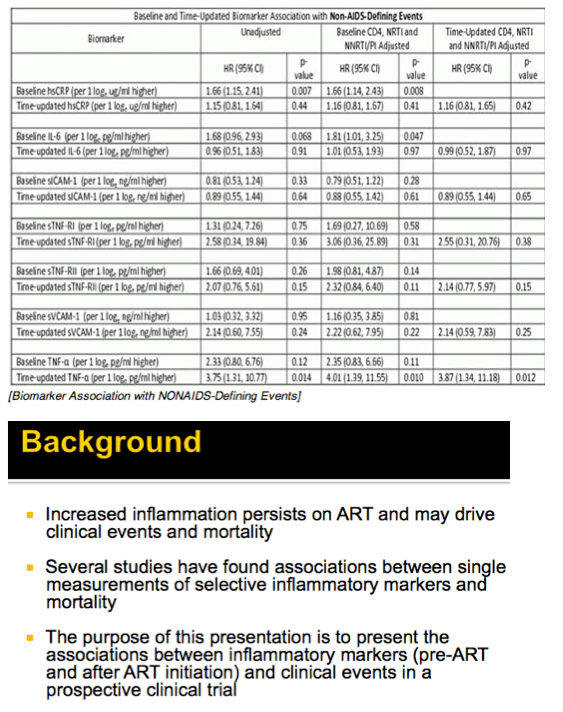
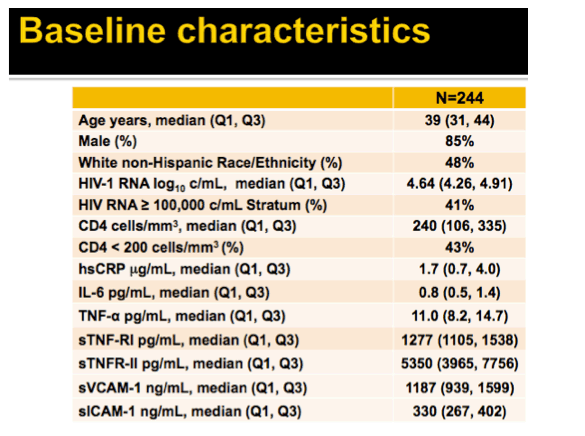
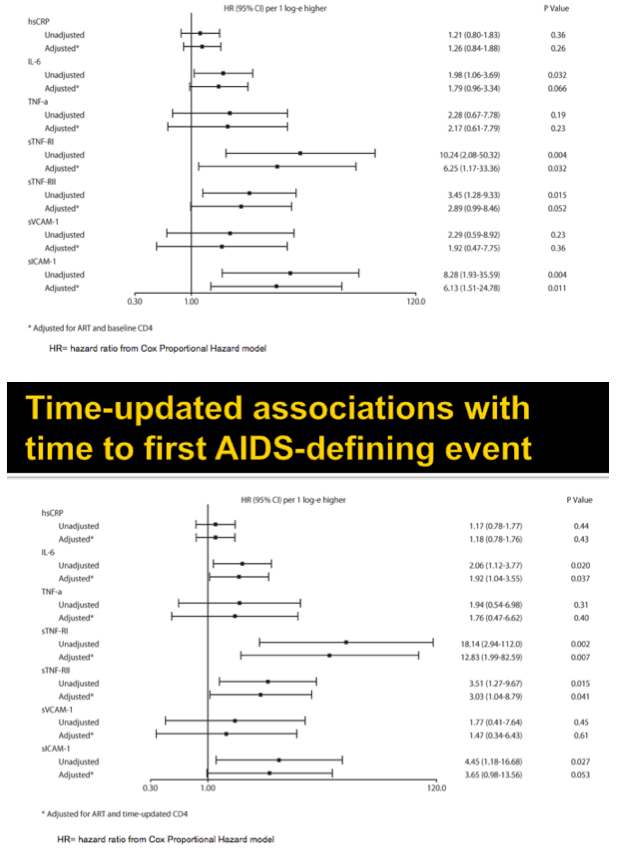
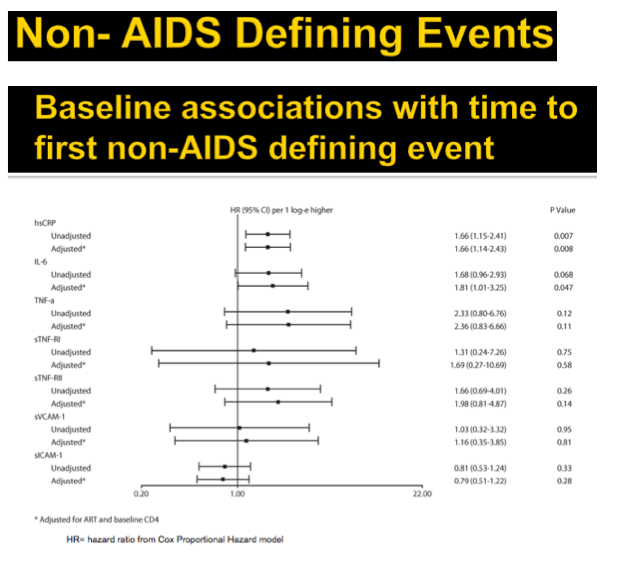
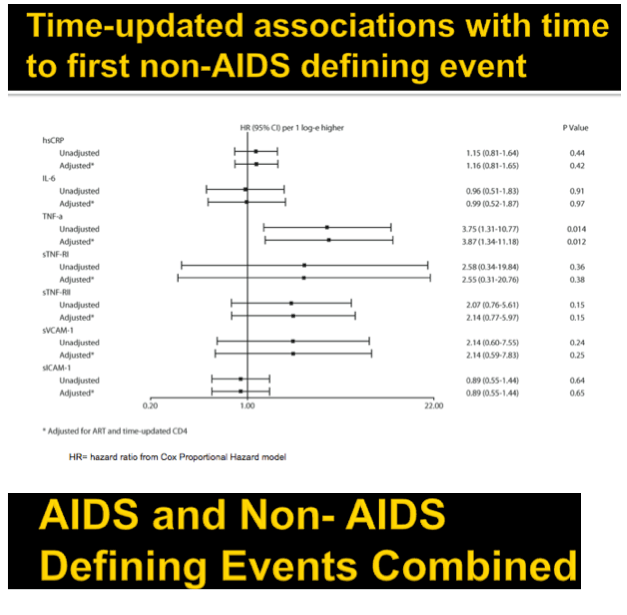
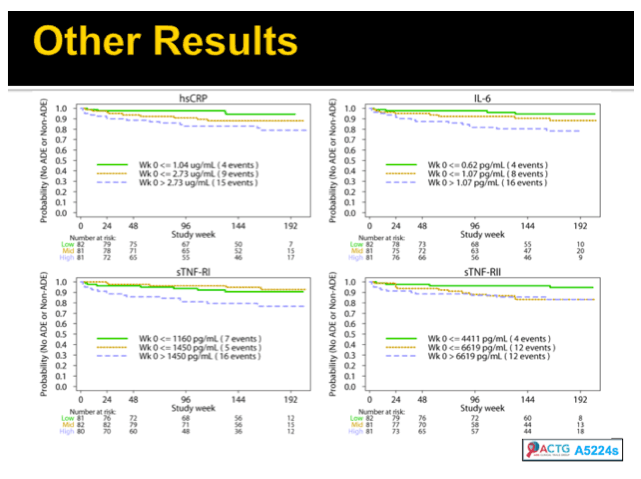
Results: Analysis included 244 subjects; 85% male, 48% white non-Hispanic, with median age 39 years, HIV-1 RNA 4.6 log10 copies/mL, and CD4 240 cells/µL. A total of 13 AIDS-defining events (9 opportunistic infections; 3 AIDS-cancers, 1 recurrent bacterial pneumonia) and 18 non-AIDS defining events (6 diabetes, 4 cancers, 3 cardiovascular, 5 pneumonias) occurred. Higher baseline IL-6, sTNF-RI, sTNF-RII, and sICAM-1 were significantly associated with increased risk of AIDS-defining events. Adjustment for baseline HIV-1 RNA did not change results, while adjusting for CD4 count left sTNF-RI and sICAM-1 significantly associated with increased AIDS-defining events risk. Time-updated values of these biomarkers were also associated with increased risk of AIDS-defining events, even after adjusting for ART assignment, baseline and changes in CD4 and HIV-1 RNA. For non-AIDS events, only baseline hsCRP was significantly associated with increased risk; after adjustment for baseline CD4 count, IL-6 became significantly associated with higher risk. Analyses of time-updated biomarker value showed TNF-a to be significantly associated with increased risk of non-AIDS-defining events, even after adjustment for ART, baseline and changes in CD4 and HIV-1 RNA.
Conclusions: Higher levels of several inflammatory biomarkers were associated independently of CD4 count with increased risk of AIDS and non-AIDS events. Larger and longer studies should investigate the use of these markers as predictors of clinical endpoints.




|
| |
|
 |
 |
|
|