 |
 |
 |
| |
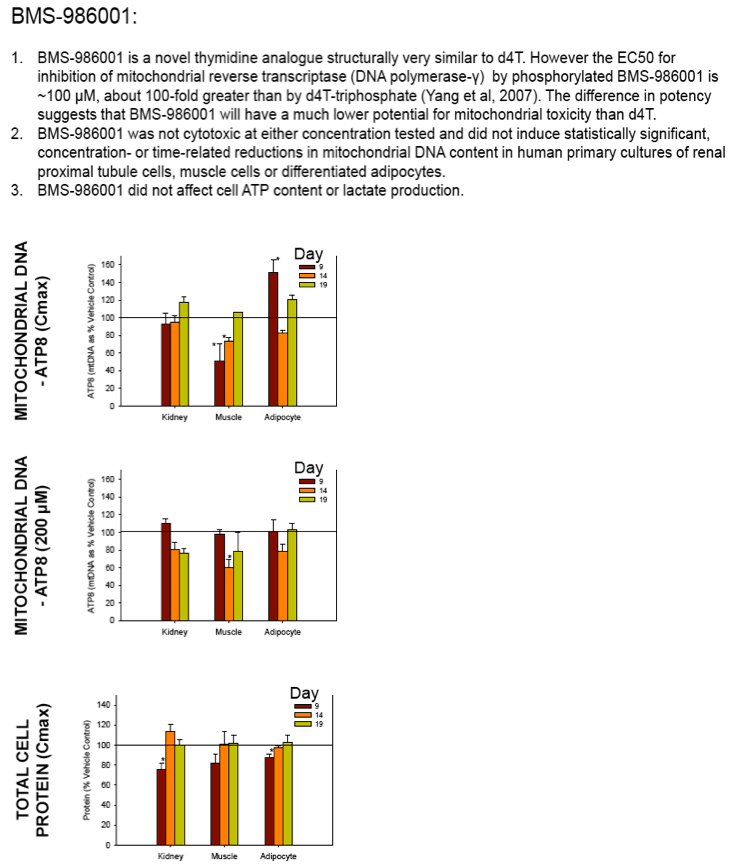
THE HIV NRTI BMS-986001 DOES NOT DEGRADE MITOCHONDRIAL DNA IN LONG TERM PRIMARY CULTURES
OF CELLS ISOLATED FROM HUMAN KIDNEY, MUSCLE AND SUBCUTANEOUS FAT
|
| |
| |
Reported by Jules Levin
XIX International AIDS Conference, July 22-27, 2012, Washington, DC
Faye Wang M.S., M.D. and Oliver Flint, Ph.D., Bristol-Myers Squibb, Hopewell, NJ<
INTRODUCTION
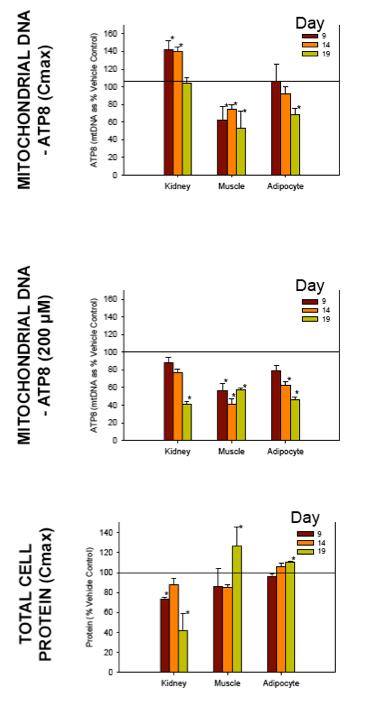
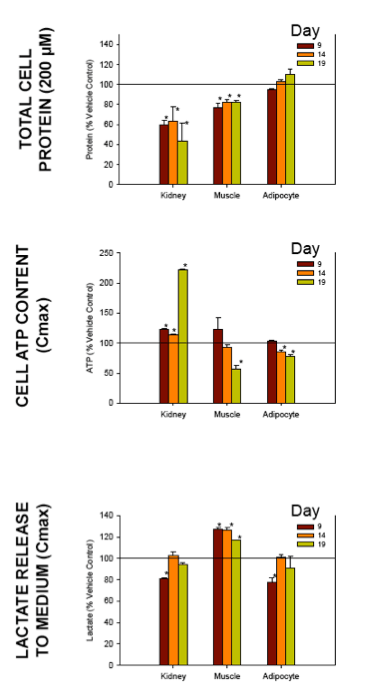
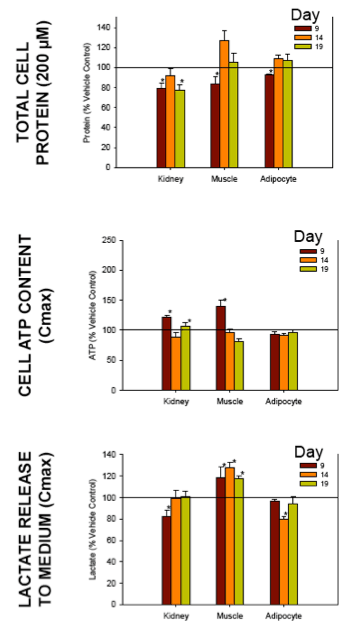
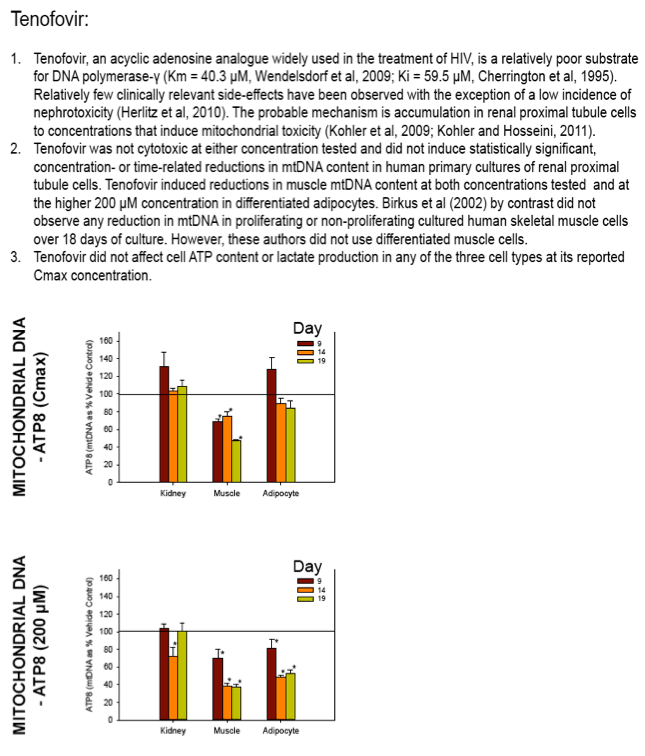
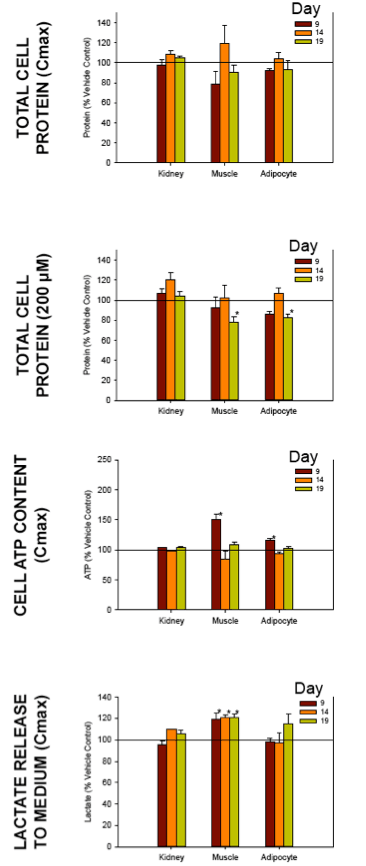
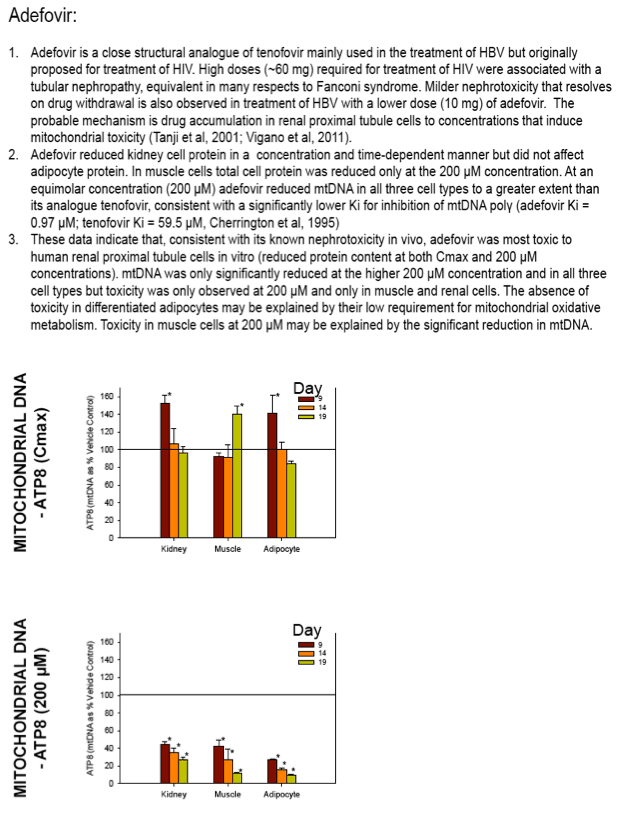
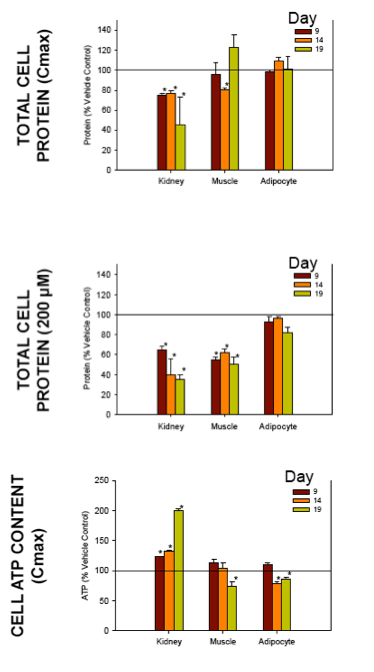
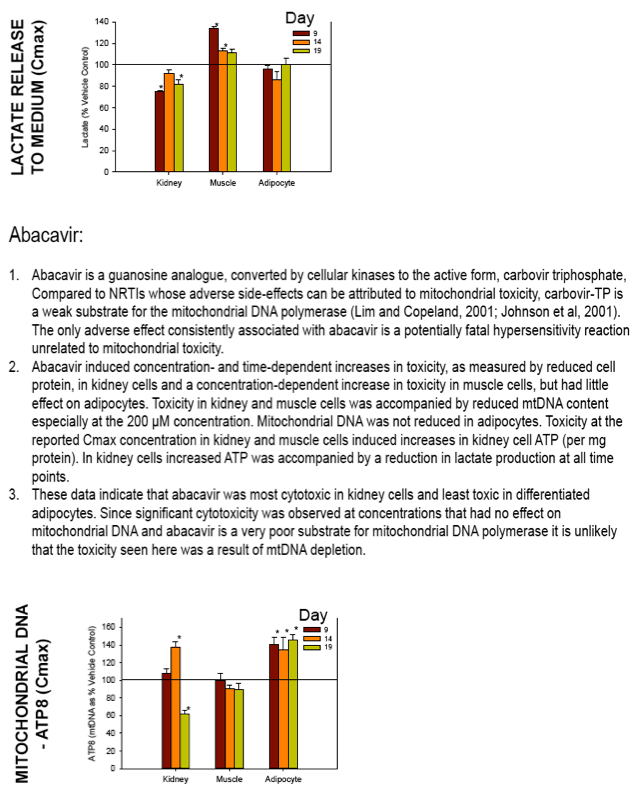
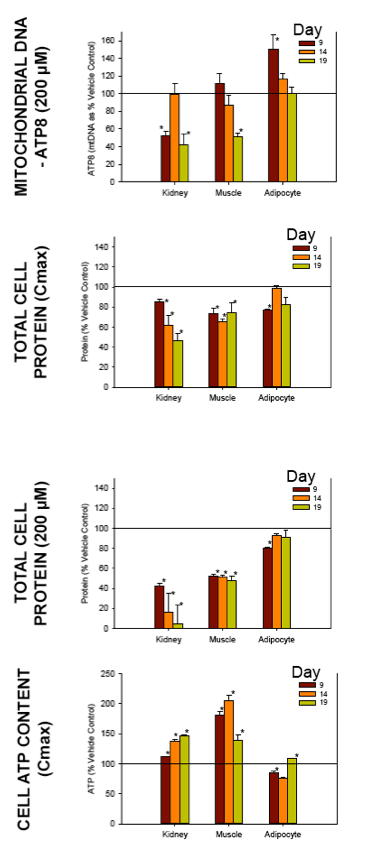
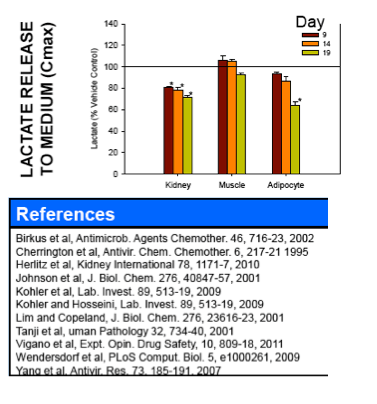
Nucleoside reverse transcriptase inhibitors (NRTIs) are used in the treatment of HIV infections in combination with other NRTIs, protease inhibitors and/or non-nucleoside reverse transcriptase inhibitors. The NRTIs prevent viral replication by inhibiting viral reverse transcriptase. The adverse side effects of NRTI treatment, including lactic acidosis, peripheral neuropathy, myopathy, pancreatitis, renal toxicity, and hepatotoxicity have been attributed to inhibition of eukaryotic mitochondrial DNA polymerase (mtDNA polγ). The primary objective of this study was to compare the in vitro toxicity of the NRTI BMS-986001 to that of four other NRTIs: tenofovir (TDF), adefovir (ADV), stavudine (D4T), and abacavir (ABC). In vitro toxicity was assessed in three human primary cell culture systems: renal proximal tubule epithelium, differentiated subcutaneous preadipocytes, and differentiated muscle cells (myotubes). Cell cultures were exposed to NRTIs at concentrations approximating the plasma Cmax in humans, or 200 μM, for 19 days. Four parameters of in vitro cytotoxicity were measured: total cell protein and ATP content, cell mitochondrial ATP8 DNA content, and lactate concentration in the media.



|
| |
|
 |
 |
|
|