 |
 |
 |
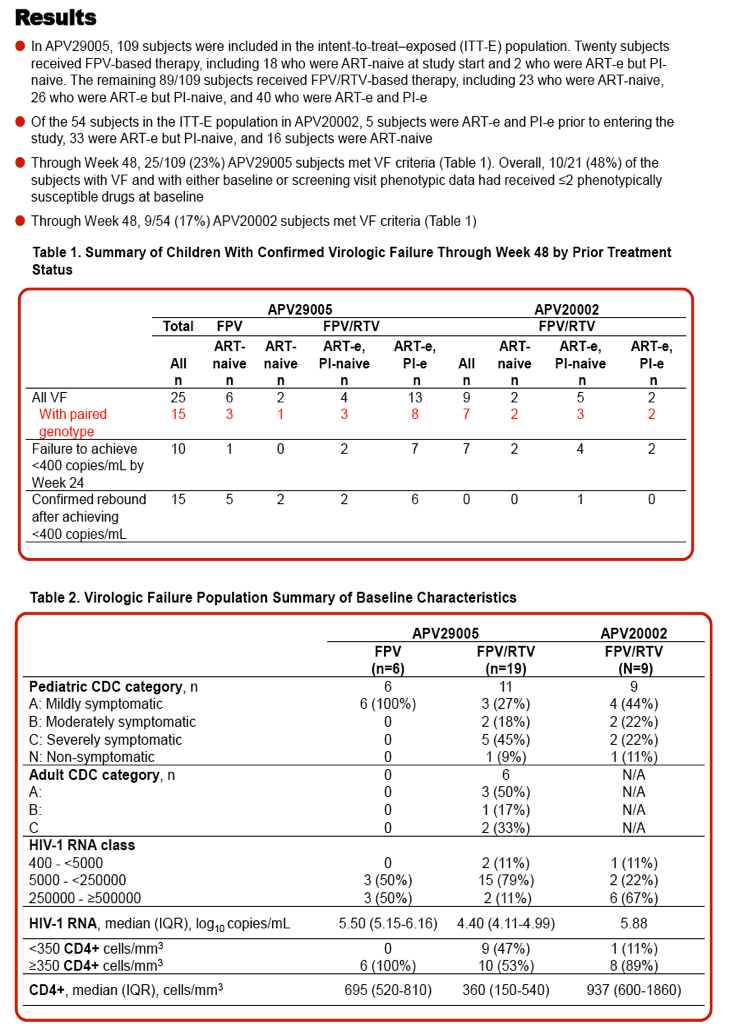
| |
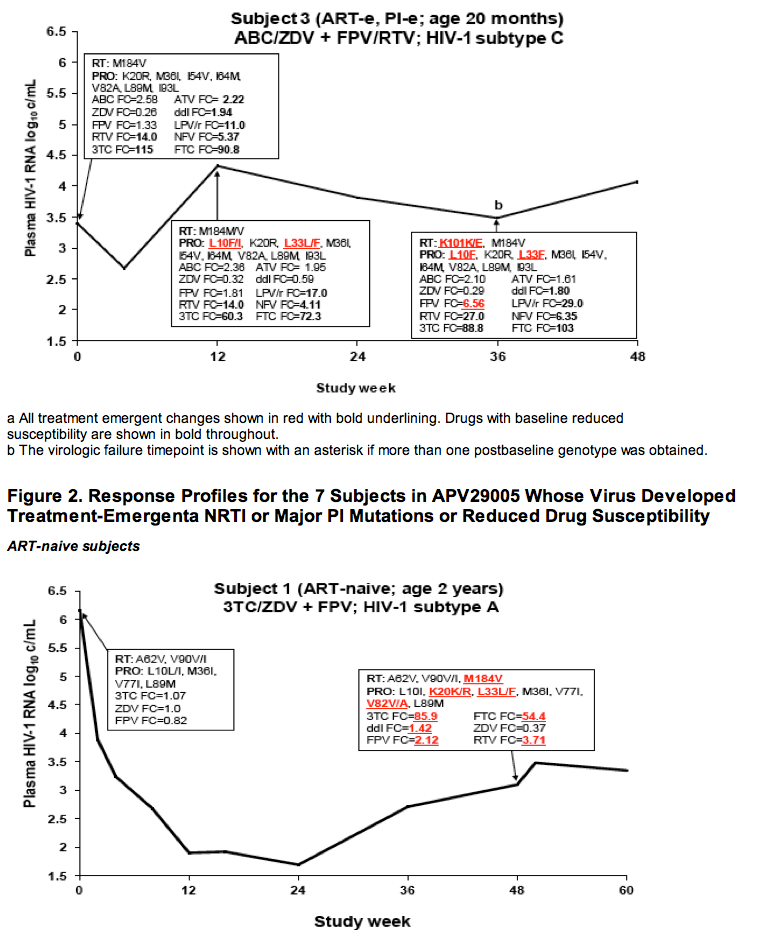
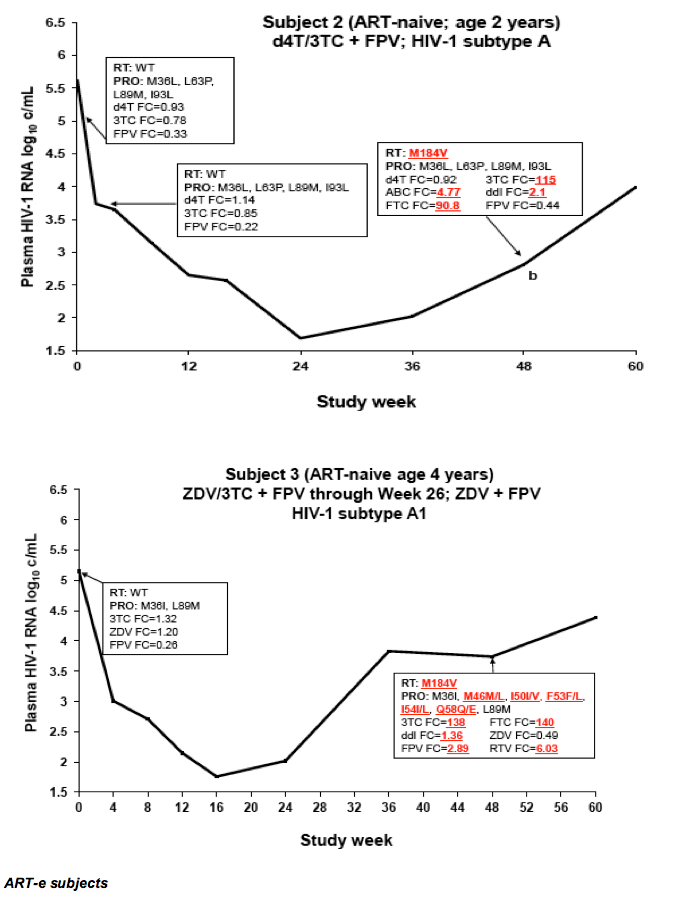
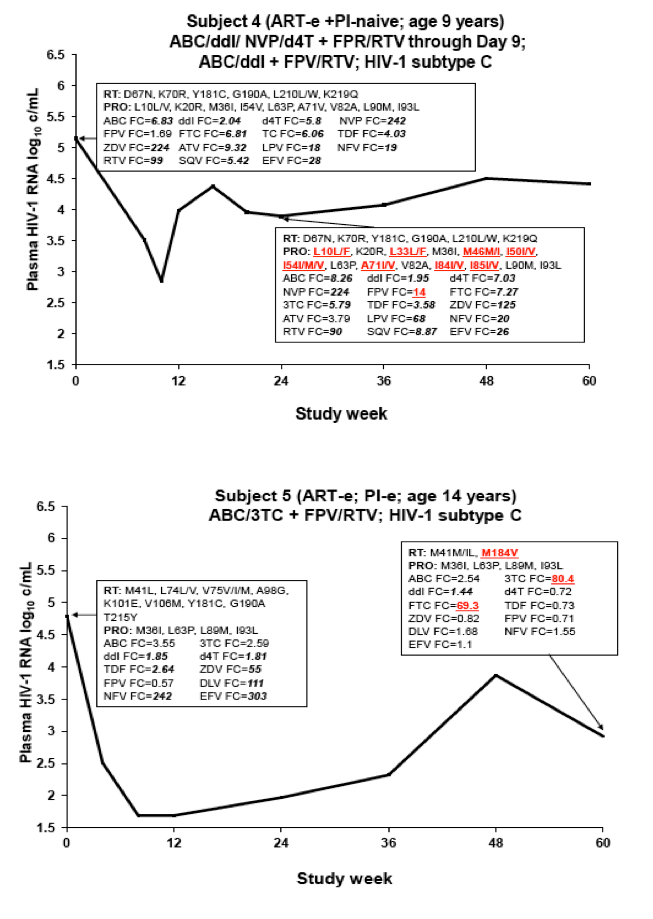
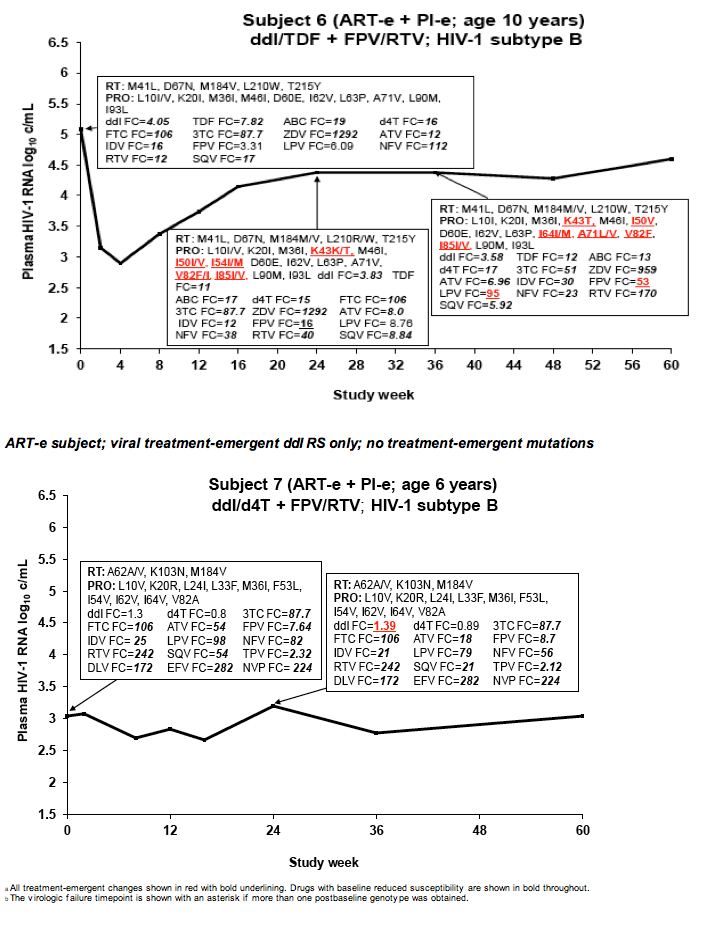
Drug Resistance and Mutational Profile in Fosamprenavir-Treated HIV-Infected Children Aged 2 Months to 18 Years at Start of Therapy
|
| |
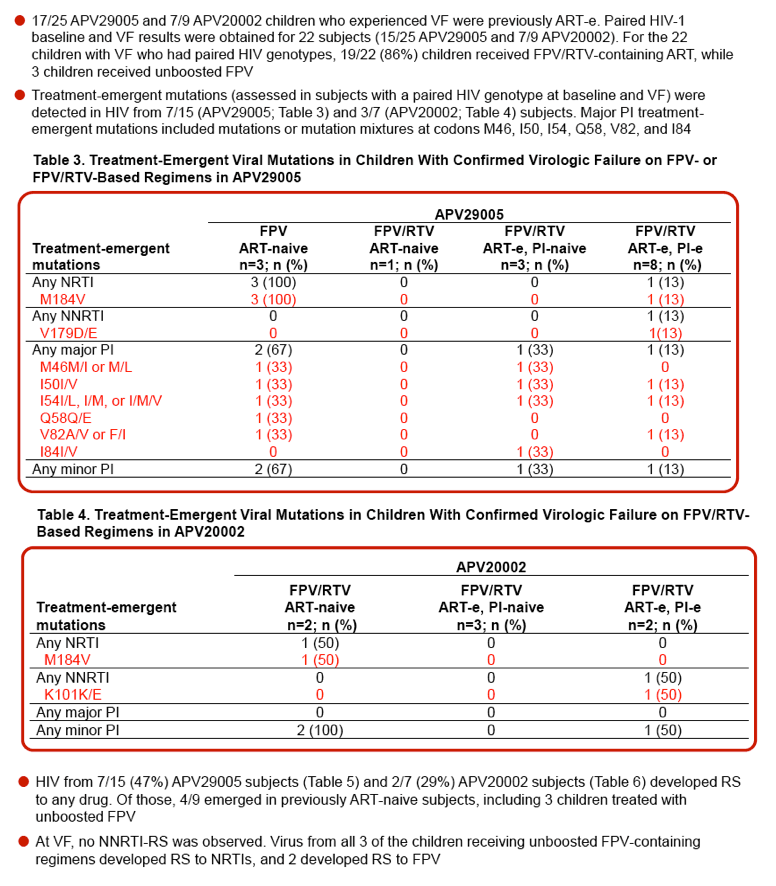
| |
Reported by Jules Levin
IAC 2012 Wash DC USA July 22-27
L Ross,1 M Cotton,2 H Cassim,3 E Voronin,4 N Givens,5 J Sievers,5 K Cheng,5 for the APV29005 & APV20002 Pediatric Study Groups
1GlaxoSmithKline, Infectious Disease Medicines Development Center, Research Triangle Park, NC, USA; 2Tygerberg Children's Hospital, Tygerberg, South Africa; 3Perinatal HIV Research Unit, Johannesburg, South Africa; 4Republic Hospital of Infectious Disease, St. Petersburg, Russian Federation; 5GlaxoSmithKline, Infectious Disease Medicines Discovery and Development, Uxbridge, UK

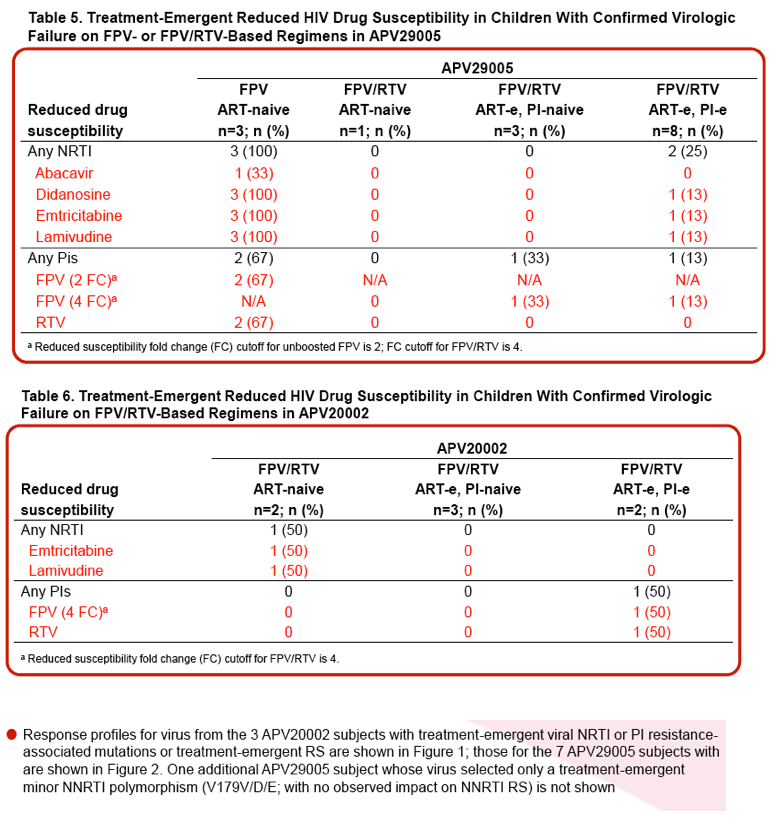

|
| |
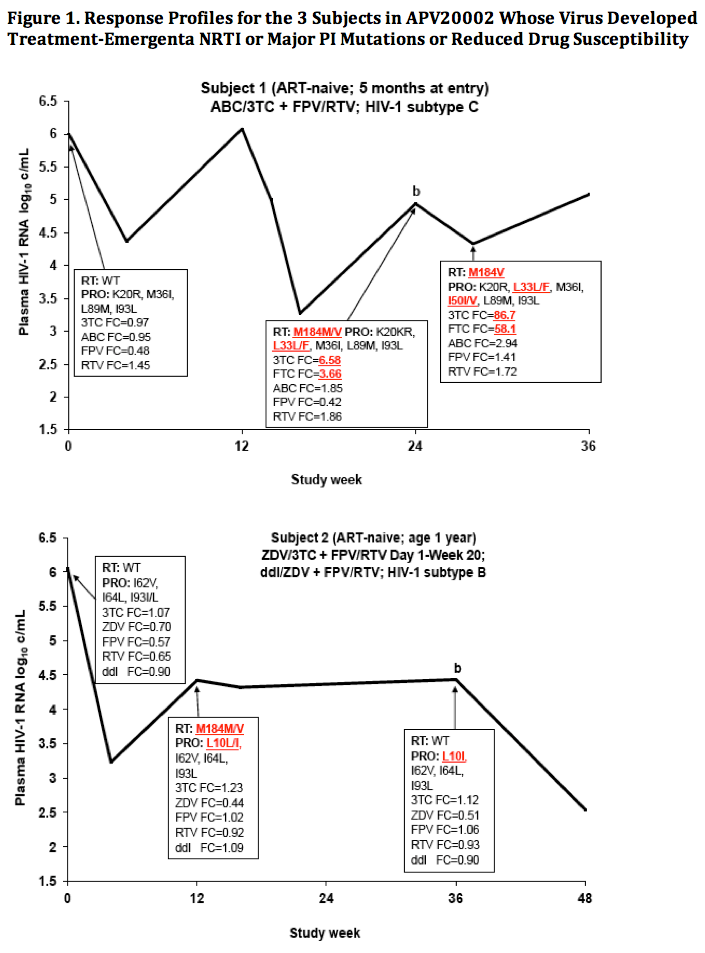
|
 |
 |
|
|