 |
 |
 |
| |
Renal Safety Profile of Elvitegravir/Cobicistat/Emtricitabine/Tenofovir DF (STB)
and of Cobicistat-boosted Atazanavir plus Emtricitabine/Tenofovir DF in HIV Patients
|
| |
| |
Reported by Jules Levin
52nd ICAAC Sept 9-12 2012 SF
P Benson1, C Mayer2, J Morales Ramirez3, J Winston4, MS Rhee5, J Szwarcberg5
1Be Well Medical Center, Berkley, MI, US; 2St. Joseph's Comprehensive Research Institute, Tampa, FL, US; 3Clinical Research PR, Inc., San Juan, PR;
4Mount Sinai School of Medicine, New York, NY, US; 5Gilead Sciences, Foster City, CA, US
Program Abstract
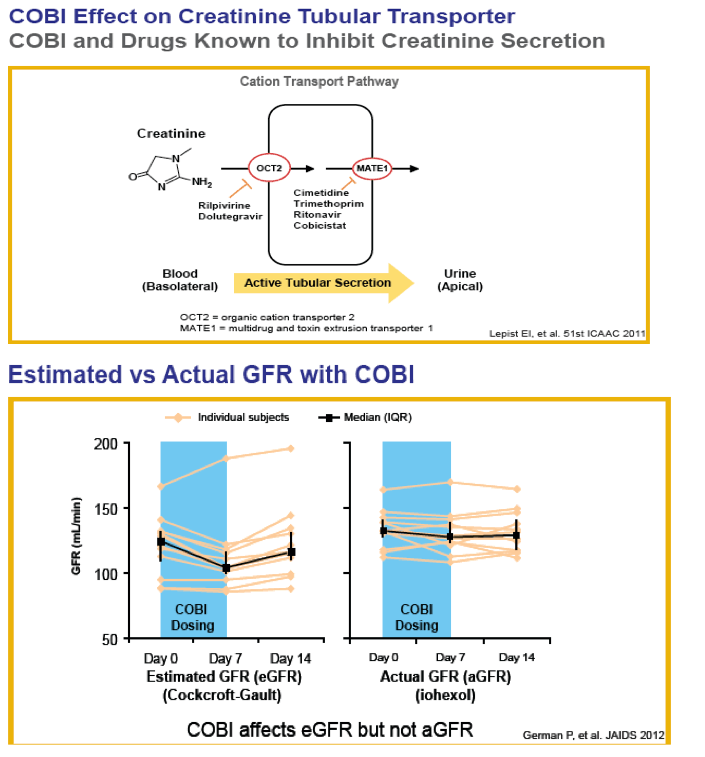
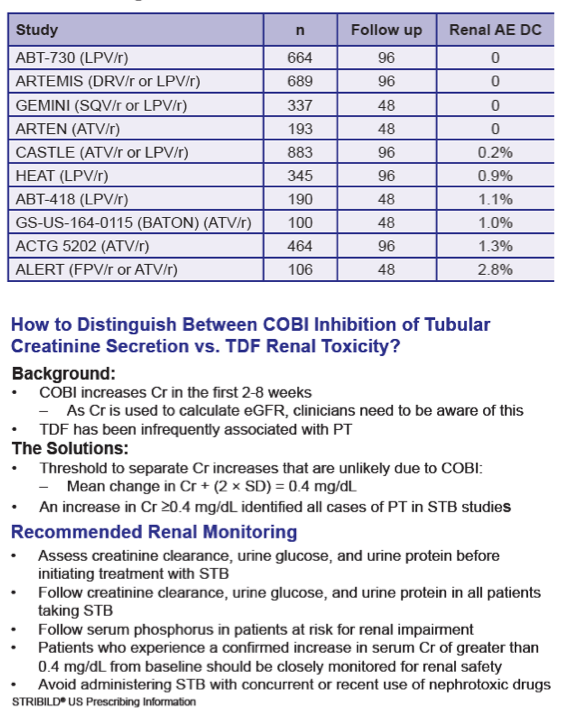
Background: Tenofovir DF (TDF) has infrequently been associated with renal toxicity. Cobicistat (COBI), like ritonavir, can increase creatinine (Cr) without affecting glomerular filtration by inhibiting renal tubular creatinine secretion via transporter (MATE-1). We examined the renal safety of regimens containing TDF and COBI.
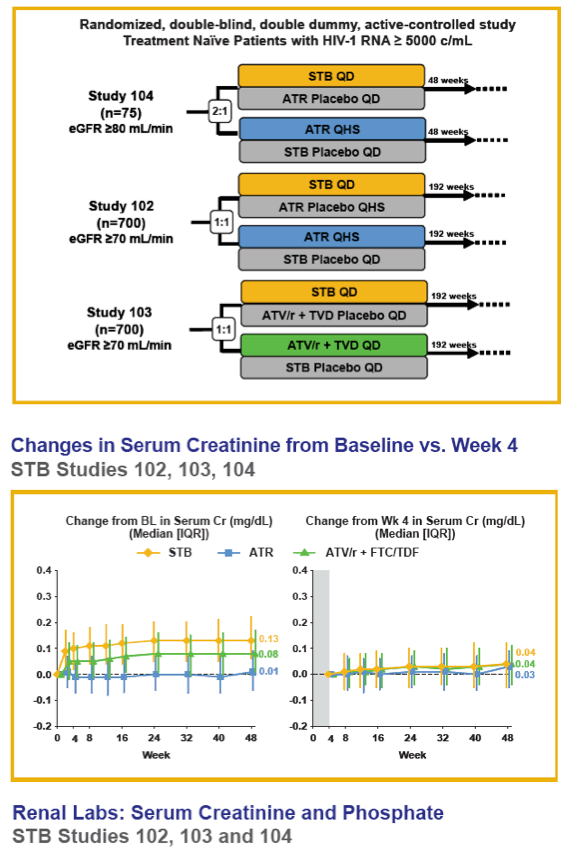
Methods: Data were analyzed using pooled data from three "QUAD studies" (236-0102 and -0104: "QUAD" (elvitegravir [EVG]/COBI/emtricitabine [FTC]/TDF) vs. FTC/TDF/efavirenz [ATR]; 236-0103: QUAD vs. ritonavir-boosted atazanavir [ATV/r] + FTC/TDF [TVD]) and pooled data from two "COBI studies" (216-0105 and -0114: COBI-boosted ATV [ATV/co] vs. ATV/r in combination with TVD).
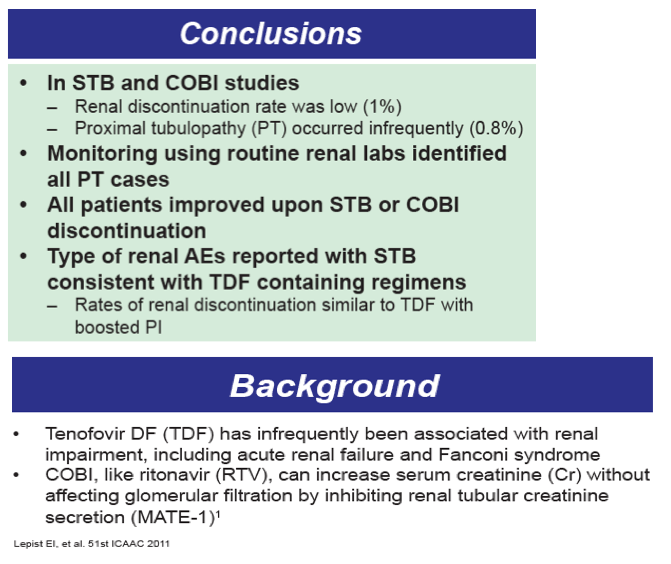
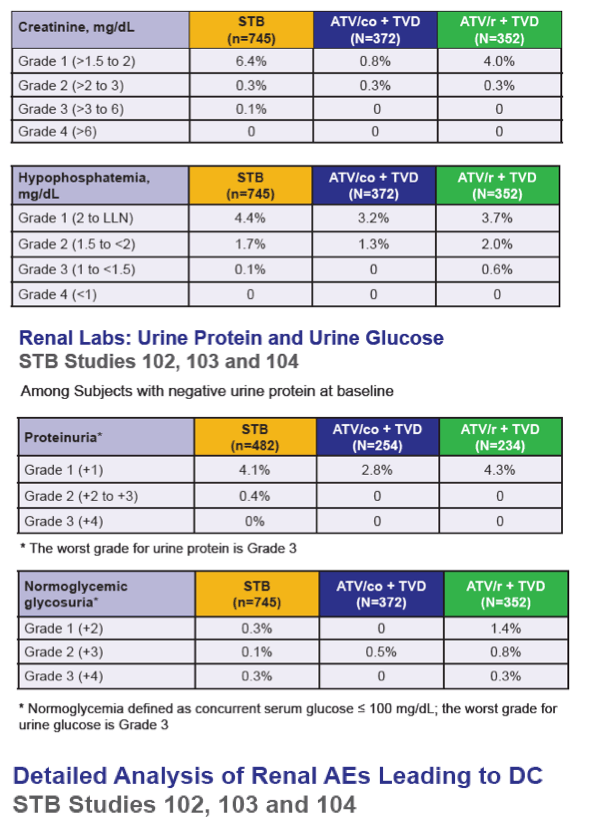
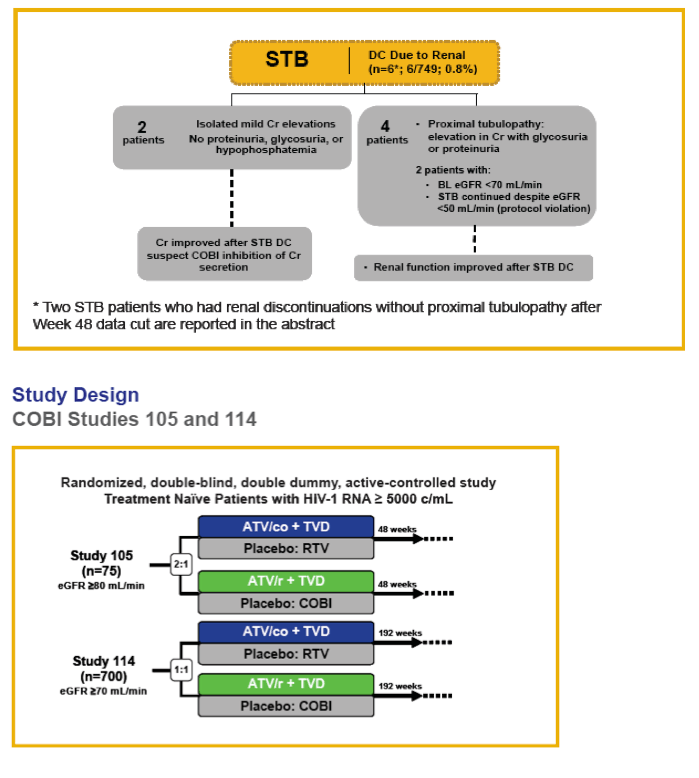
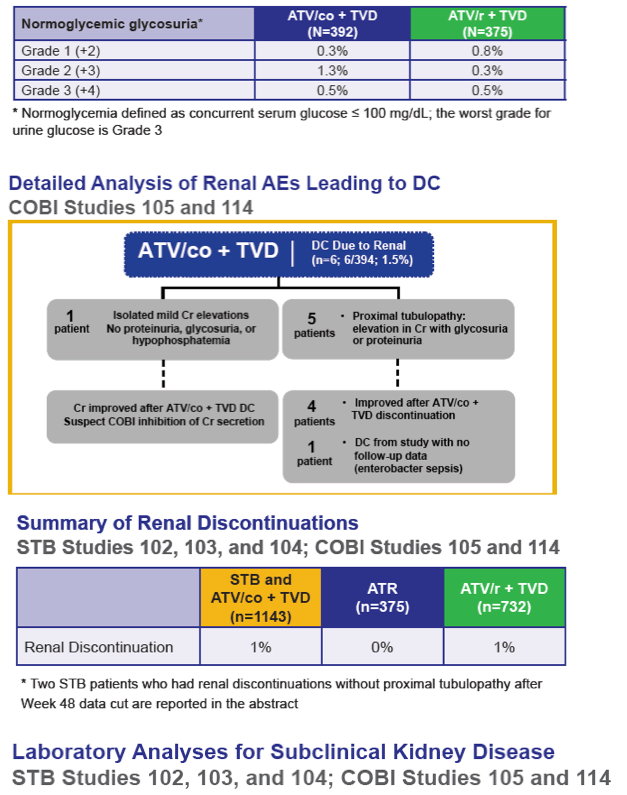
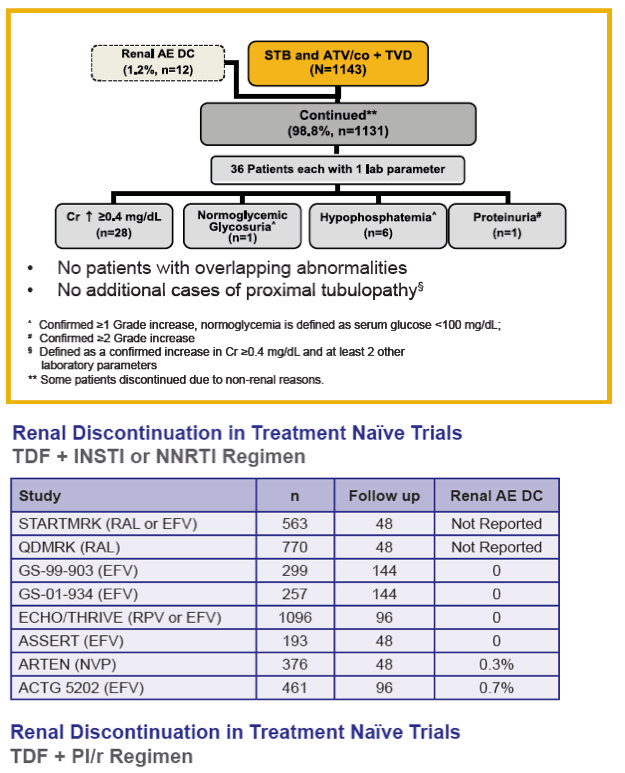
Results: In the pooled QUAD studies, the median exposures to QUAD (n=749), ATR (n=375), and ATV/r+TVD (n=355) were 48, 59, and 48 weeks. The median (IQR) changes in Cr (mg/dL) at Week 48 in the three groups were 0.13 (0.06 to 0.22), 0.01 (-0.06 to 0.09), and 0.08 (0.01 to 0.17). The rates of renal AEs leading to study drug discontinuation (renal AE DC) were QUAD 1.1 % (8/749), ATR 0 %, and ATV/r+TVD 0.3 % (1/355). One patient in QUAD reported a serious renal AE not leading to study drug discontinuation. In the pooled COBI studies, the median exposures to ATV/co+TVD (n=394) and ATV/r+TVD (n=377) were 48 weeks. The median (IQR) changes in Cr (mg/dL) at Week 48 in the two groups were 0.13 (0.05 to 0.22) and 0.09 (0.01 to 0.17). The rates of renal AE DC were ATV/co+TVD 1.5 % (6/394), ATV/r+TVD 1.6 % (6/377); two and three renal AE DCs from each group were serious AEs.
Conclusions: As expected, small increases in Cr were seen with the use of QUAD or COBI. The rates of renal event leading to study drug discontinuation with TDF-containing COBI-boosted EVG or ATV regimen were low at 1.2% (14/1143), and comparable to historical rates seen with TDF containing ritonavir-boosted protease inhibitor regimen.

|
| |
|
 |
 |
|
|