 |
 |
 |
| |
The Pharmacokinetic Interactions of the HCV Protease Inhibitor Simeprevir (TMC435) With HIV Antiretroviral Agents in Healthy Volunteers
|
| |
| |
Reported by Jules Levin
IDSA Oct 17-21 2012 San Diego
Sivi Ouwerkerk-Mahadevan, PhD,1 Vanitha Sekar, PhD,2 Alexandru Simion, MD, PhD,3 Monika Peeters, MSc,3 Maria Beumont-Mauviel, MD3
1Janssen Research & Development, Beerse, Belgium; 2Janssen Research & Development, Titusville, NJ, USA; 3Janssen Infectious Diseases, Beerse, Belgium
CONCLUSIONS
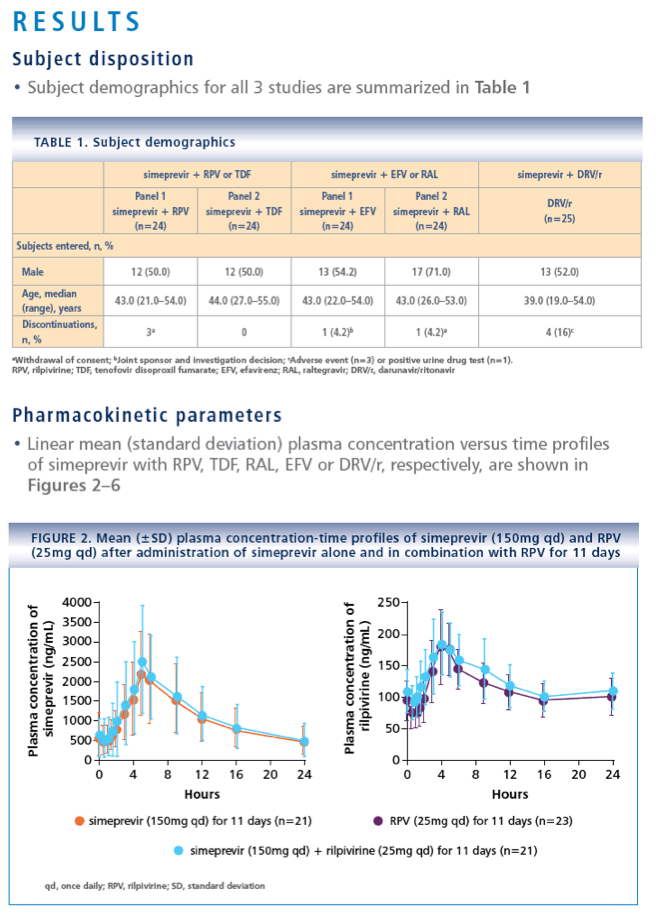
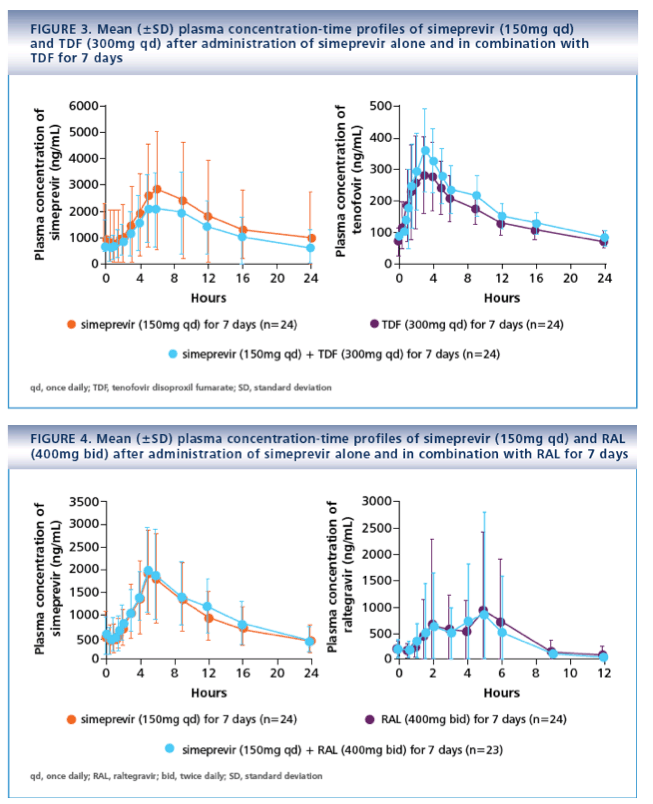
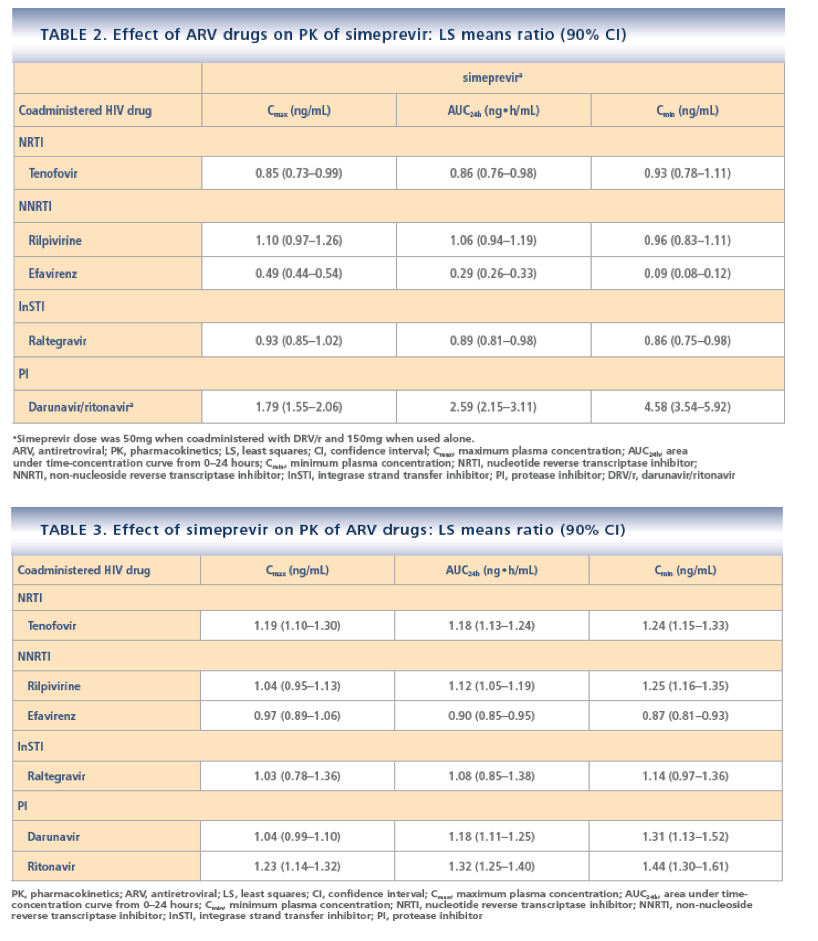
· Results from this PK analysis demonstrated that no dose adjustments are required when combining simeprevir with RPV, RAL or TDF
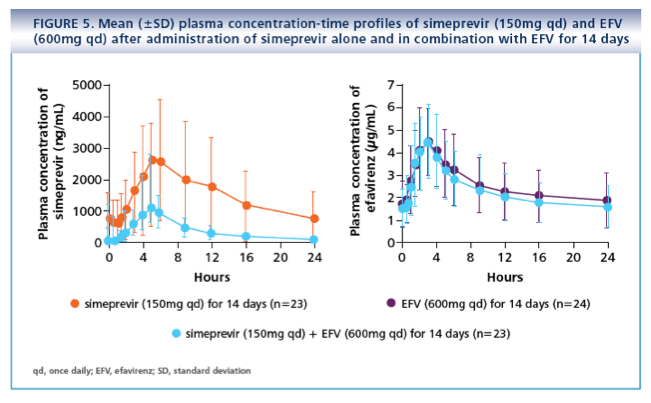
· Coadministration of simeprevir and EFV is not recommended due to a significant decrease in simeprevir plasma concentrations in the presence of EFV
· Coadministration of simeprevir and DRV/r is not recommended due to a significant increase in simeprevir exposure in the presence of DRV/r, even after dose adjustment of simeprevir from 150mg qd to 50mg qd. Similar effects are likely to be seen with other ritonavir-boosted PIs
· All treatments were safe and well tolerated, and no SAEs were reported
· Once available, data from the ongoing Phase III study (NCT01479868) will further characterize the safety, tolerability, efficacy and PK profile of simeprevir in patients with chronic HCV infection who are coinfected with HIV-1
Introduction
· According to the Centers for Disease Control and Prevention, coinfection with hepatitis C virus (HCV) is present in approximately 25% of HIV-positive patients, and approximately 80% of HIV-positive injection drug users are also infected with HCV1
· Persons coinfected with HCV and HIV may experience faster progression of liver fibrosis than those who are HCV mono-infected, and they may experience more liver-related health problems than persons living with HIV alone. Liver disease has become the leading cause of non-AIDS-related death in the HIV/HCV coinfected
population due in part to longer life expectancy after the widespread use of highly active antiretroviral (ARV) therapy in the developed world1
· Concurrent treatment of HIV and HCV infection is complicated by pill burden, overlapping drug toxicities and drug interactions, all of which may limit HIV ARV treatment options.1 In addition, low rates of sustained virologic response for HCV genotype 1 with peginterferon and ribavirin underscore the need for other strategies to improve patient outcomes2,3
· Simeprevir (TMC435), an investigational, once daily (qd), oral HCV NS3/4A protease inhibitor (PI)
- Has demonstrated potent antiviral activity in patients infected with HCV genotype 1, with antiviral activity demonstrated against genotypes 2, 4, 5, and 6 isolates4-8
- Is a substrate of cytochrome P450 (CYP) 3A and a mild inhibitor of intestinal (not hepatic) CYP3A and of hepatic CYP1A29
· An ongoing Phase III study (NCT01479868) is evaluating the safety, tolerability, efficacy and pharmacokinetics (PK) of simeprevir in chronically HCV-HIV coinfected patients
· 3 Phase I, randomized, crossover studies were conducted in healthy subjects to evaluate potential PK interactions between simeprevir and the ARV agents rilpivirine (RPV), tenofovir disoproxil fumarate (TDF), efavirenz (EFV), raltegravir (RAL) and darunavir/ritonavir (DRV/r)
-- Data for the PK interactions between simeprevir and DRV/r are new, whereas PK data for simeprevir and RPV, TDF, EFV or RAL were presented earlier this year at the 19th Conference on Retroviruses and Opportunistic Infections in Seattle, WA, March 5-8, 201210
METHODS
Study design
· Studies were conducted to investigate steady-state PK interactions between simeprevir (150mg qd) and the following ARV agents
- RPV (25mg qd for 11 days) or TDF (300mg qd for 7 days; TMC435-C114, NCT01205139)
- EFV (600mg qd for 14 days) or RAL (400mg twice daily [bid] for 7 days; TMC435-C123, NCT01241773)
- DRV/r (800/100mg qd for 7 days; TMC435-C115, NCT01323257)
· In anticipation of an interaction, the dose of simeprevir was reduced to 50mg qd when combined with DRV/r
· Each study consisted of 2 panels: each panel evaluated simeprevir and an ARV except in TMC435-C115, in which Panel 1 assessed the PK of simeprevir in combination with erythromycin (results are not included in this report)
-- Per panel, each subject received 3 treatments during 3 consecutive sessions according to a classic 6-sequence, 3-period Williams design (Figure 1)
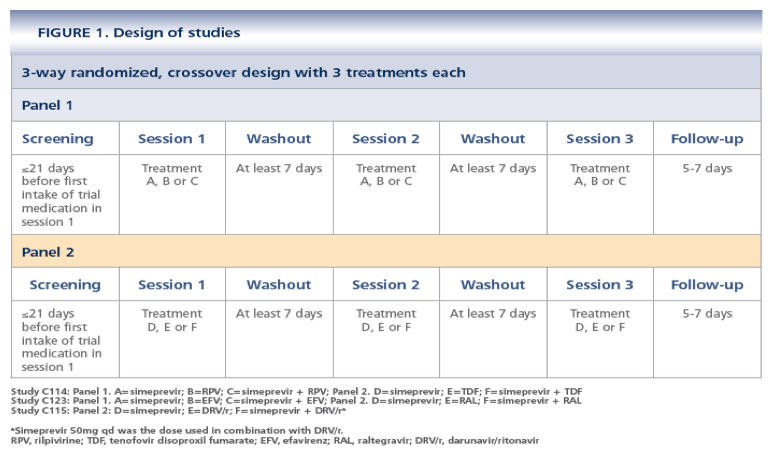
· Eligible subjects were healthy men and women aged 18 to 55 years with a body mass index of 18.0 to 30.0 kg/m2 who had been nonsmokers for at least 3 months before screening. Women of childbearing potential had to have a negative pregnancy test at screening and had to use highly effective methods of birth
control before, during and up to 30 days after the last dose of study medication
· Subjects with HIV-1, HIV-2 or hepatitis A, B or C infection at screening were excluded
· All medication had to be discontinued at least 14 days before the first intake of study drug (Day 1 of Session 1), except for paracetamol (acetaminophen)
or ibuprofen. Subjects were not allowed to use any medication other than the study drug(s)-except for paracetamol or ibuprofen-until the follow-up
visit, which was scheduled between 5 and 7 days after the last intake of study medication, or until after dropout
· The calculated sample size was 22 evaluable subjects within each panel in all 3 studies; therefore, a total of 24 subjects were enrolled in each panel to allow for discontinuations
· All treatments given alone and combined were administered under fed conditions, except for EFV, which was administered 2 hours after food
Study end points
· PK parameters of simeprevir and of ARVs at steady state, when given alone and as the combination
· Short-term safety and tolerability of simeprevir administered alone or in combination with ARVs
Assessments
· Plasma concentrations of simeprevir and ARVs were assessed at steady state; selected trough samples were also obtained
· Blood and urine samples for safety analyses were collected, and electrocardiograms and vital signs were taken at screening, in each treatment session (RPV-TDF study: Days 1, 7, 8, 11 and 12; EFV-RAL study: Days 1, 7, 8, 14 and 15; DRV/r study: Days 1, 7 and 8), at follow-up and at discontinuation
· Physical examinations were performed at screening; in each treatment session on Day -1 and Day 12 (for RPV), Day -1 and Day 15 (for EFV) and Day -1 and Day 8 (for TDF, RAL and DRV/r); as well as at follow-up and at discontinuation
· Adverse events (AEs) were monitored throughout the trial
Statistical methods
· Statistical methods included descriptive statistics, frequency tabulations, intent-to-treat analysis, linear mixed-effects modeling and nonparametric tests (for time to reach the maximum plasma concentration)
· PK and statistical analyses were performed using WinNonlin Professional® (version 4.1; Pharsight Corporation, Mountain View, CA, USA).
Noncompartmental analysis model 200 (extravascular input, plasma data) was applied for the PK analysis
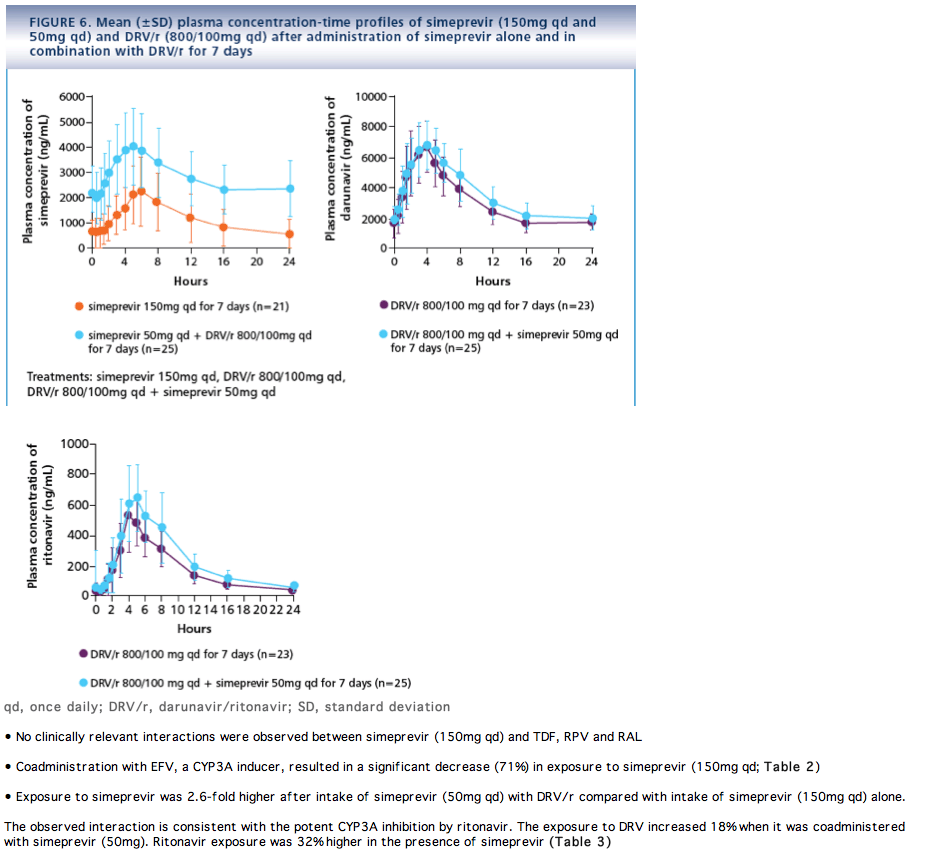
SAFETY
Simeprevir and RPV or TDF
· No AE-related treatment discontinuations or serious AEs (SAEs) were reported with simeprevir and RPV (25mg qd) or TDF (300mg qd)
- No relevant differences in the incidence of AEs were observed when simeprevir and RPV or TDF were coadministered, compared with when simeprevir or either HIV ARV was administered alone
Simeprevir and EFV or RAL
· No AE-related treatment discontinuations or SAEs were reported with simeprevir and EFV (600mg qd) or RAL (400mg bid). One subject discontinued participation
in the study after treatment with EFV 600mg qd alone due to AEs including rash, depressed mood and nightmares. All of these AEs were grade 1-2 and were
considered unrelated to simeprevir
· No relevant differences in the incidence of AEs were seen when simeprevir and RAL were coadministered compared with when simeprevir or RAL was administered alone
Simeprevir and DRV/r
· No SAEs were reported
- In total, 3 (12.0%) subjects discontinued study medication due to an AE: one subject with grade 2 rash permanently discontinued simeprevir and DRV/r, one subject with grade 1 rash permanently discontinued simeprevir and one subject with grade 1 genital rash permanently discontinued simeprevir and DRV/r (discontinuation of study medication due to grade 1 rashes was at investigator's discretion)
- Except for fatigue, no differences in the incidence of AEs were observed between administration of simeprevir alone and coadministration of simeprevir and DRV/r
REFERENCES
1. HIV and viral hepatitis. Centers for Disease Control and Prevention Web site. November
2011. http://www.cdc.gov/hiv/resources/factsheets/PDF/HIV-Viral-Hepatitisacc.pdf.
Accessed September 2012.
2. Chung RT, et al. N Engl J Med. 2004;351:451-459.
3. Torriani FJ, et al. N Engl J Med. 2004;351:438-450.
4. Lin TI, et al. Antimicrob Agents Chemother. 2009;53:1377-1385.
5. Reesink HW, et al. Gastroenterology. 2010;138:913-921.
6. Moreno C, et al. J Hepatol. 2012;56:1247-1253.
7. Fried MW, et al. Oral presentation at AASLD 2011.
8. Zeuzem S, et al. Poster LB-2998 presented at EASL 2011.
9. Sekar V, et al. Poster 1076 presented at EASL 2010.
10. Ouwerkerk-Mahadevan S, et al. Paper 49 presented at CROI 2012.
|
| |
|
 |
 |
|
|