 |
 |
 |
| |
Dolutegravir Treatment of HIV Subjects With Raltegravir Resistance: Integrase Resistance Evolution in Cohort II of the VIKING Study
|
| |
| |
Reported by Jules Levin
International Workshop on HIV & Hepatitis Virus Drug Resistance and Curative Strategies; June 5-9, 2012; Sitges, Spain
CL Vavro,1 KC Dudas,1 S Hasan,2 JO Huang,3 JM Yeo,4 MR Underwood1
GlaxoSmithKline: 1Research Triangle Park, NC, USA; 2Stevenage, UK; 3Toronto, ON, Canada; 4London, UK

Abstract
Background: The integrase inhibitor (INI) dolutegravir (DTG) has low fold change (FC) in IC50 against HIV with raltegravir (RAL)-resistant Y143 and N155 pathway and Q148H/K/R single mutants; FC generally increases for Q148H/K/R as RAL-associated mutations increase.1 The VIKING study enrolled 2 sequential cohorts of ART-experienced adults harboring RAL-resistant HIV. Subjects received DTG 50 mg once daily (Cohort I) or twice daily (BID; Cohort II). Cohort II also required at least one fully active drug (PSS ≥1) in the optimized background regimen (OBR).
Methods: Integrase genotypic and phenotypic results, minor variant analyses, and plasma HIV-1 RNA were assessed.
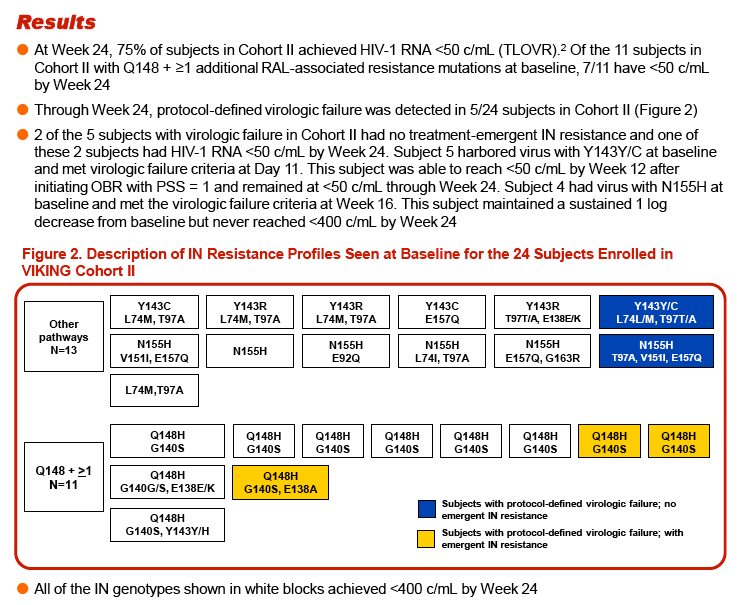
Results: A broad range of RAL-resistant genotypes and DTG FC were observed in the 24 Cohort II subjects. Through Week 24, 75% of subjects achieved <50 c/mL HIV-1 RNA, and 5/24 (21%) subjects experienced VF (Pt5 at Day 11, Pt2 and Pt3 at Week 8, Pt4 and Pt1 at Week 16).
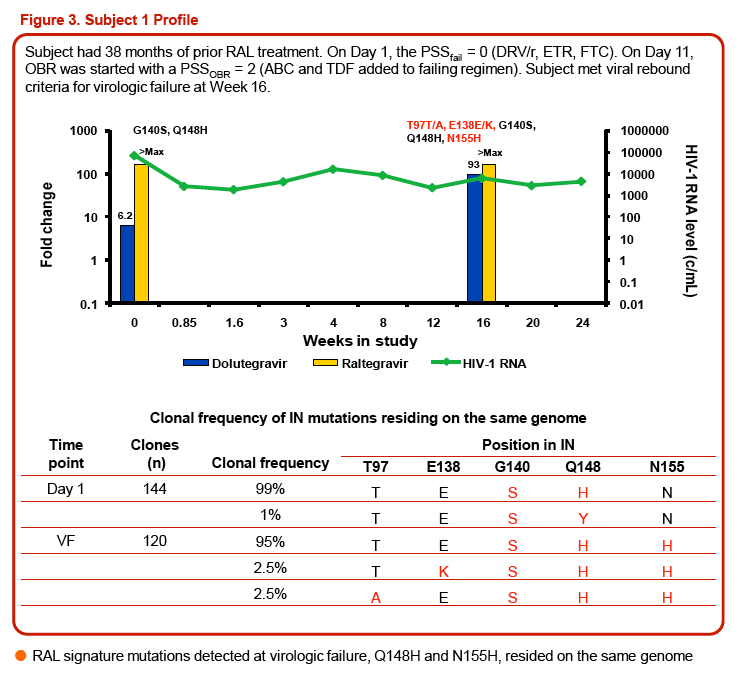
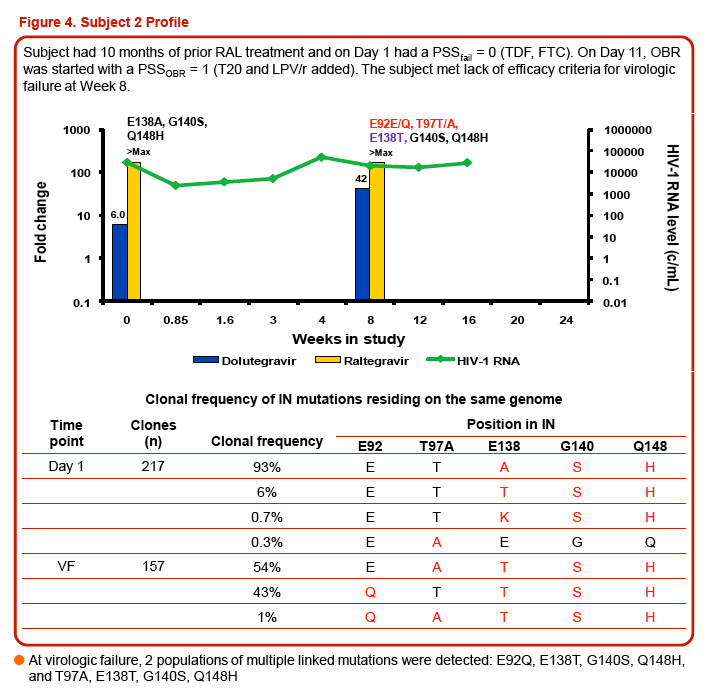
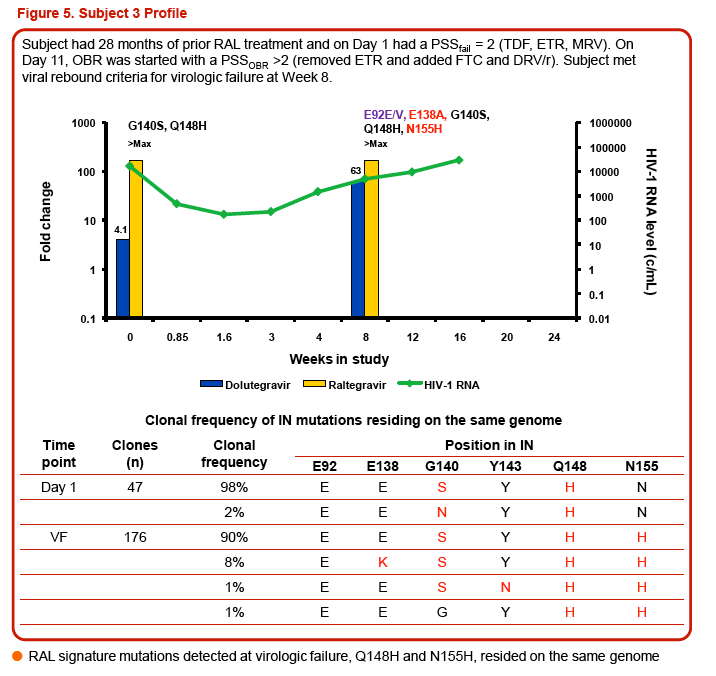
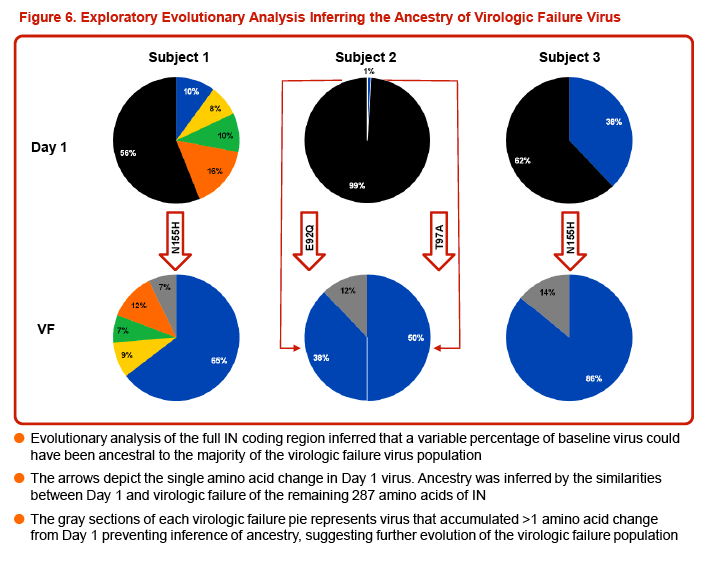
Three of the 24 subjects, all with virus having Q148 + ≥1 additional integrase resistance-associated mutations at Day 1, had treatment-emergent integrase resistance-associated mutations and reduced DTG susceptibility at the time of VF. All 3 subjects had initial HIV-1 RNA declines of 1-2 log10 c/mL with subsequent viral rebound. Virus from Pt1 added T97T/A, E138E/K, and N155H, with DTG FC increase from 6.23 to 93 (Week 16). Virus from Pt2 added E92E/Q and T97T/A, with DTG FC increase from 6.04 to 42 (Week 8). Virus from Pt3 added E138E/K and N155H, with DTG increase from 4.11 to 63 (Week 8). Minority species and evolutionary analyses revealed that small Day 1 cluster(s) acquired additional linked integrase resistance-associated mutations and expanded as a majority of the VF population.
Conclusion: Few ART-experienced subjects with RAL resistance in VIKING Cohort II experience VF while receiving DTG 50 mg BID and OBR that includes at least one active drug. Treatment-emergent IN resistance with an increase in DTG FC was detected in only 3 subjects, all harboring virus with Q148 + ≥3 additional integrase resistance-associated mutations at VF. These results support DTG 50 mg BID use for a larger ongoing phase III study (VIKING-3) in this patient population.

References
1. Kobayashi et al. Antimicrob Agents Chemother. 2011;55:813-821. 2. Soriano et al. EAC 2011; Belgrade, Serbia. Abstract PS1/2. 3. Underwood et al. IDRW 2011; Los Cabos, Mexico. Abstract 2. 4. Fransen et al. J Virol. 2009;83:11440-11446.
|
| |
|
 |
 |
|
|