 |
 |
 |
| |
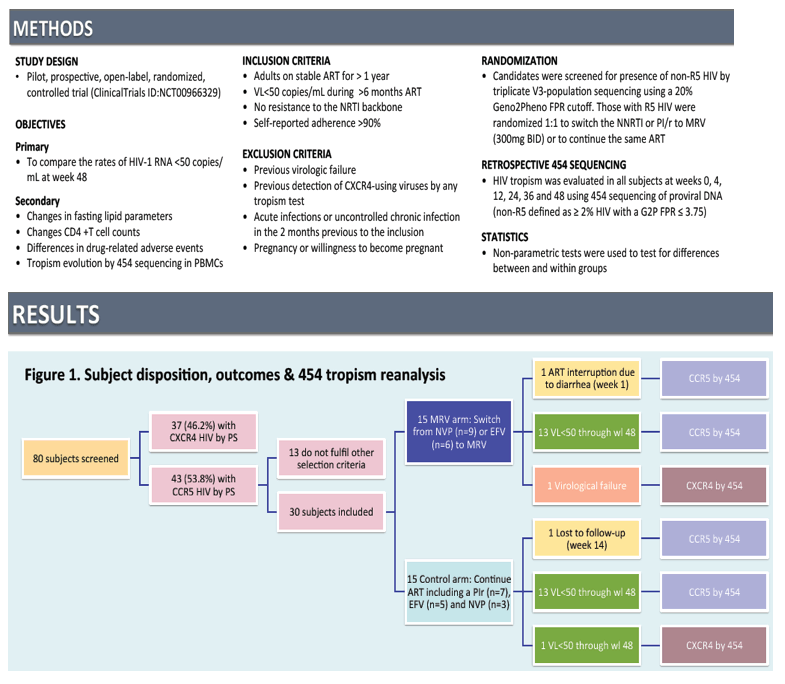
Switching the NNRTI or the PI/r to Maraviroc in Aviremic Subjects Infected with RS HIV by Vs Loop Population Sequencing: 48 week, Prospective, Controlled, Pilot Study
|
| |
| |
Reported by Jules Levin
International Workshop on HIV & Hepatitis Virus Drug Resistance and Curative Strategies; June 5-9, 2012; Sitges, Spain

CONCLUSION
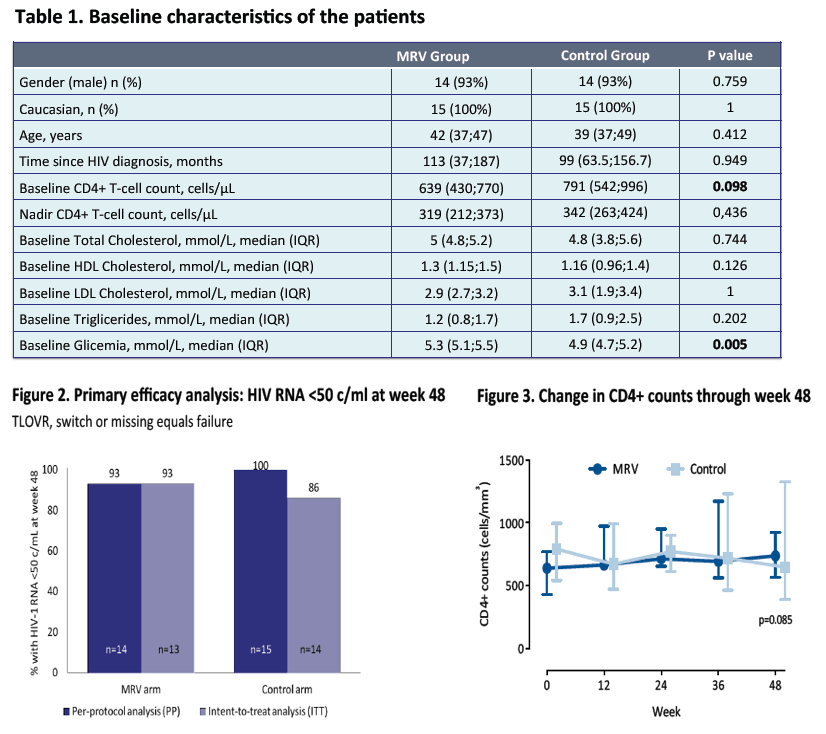
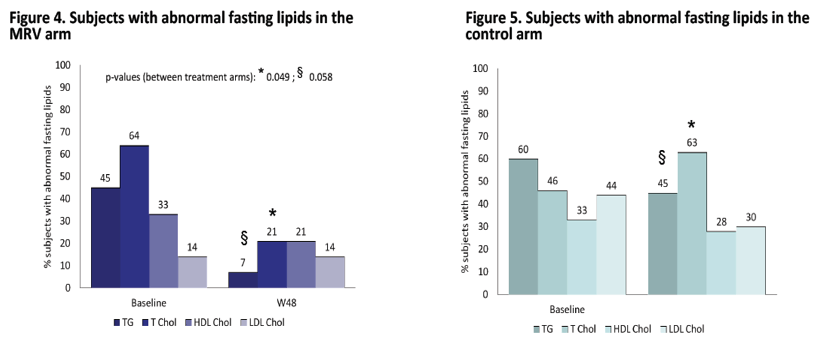
In this small pilot, prospective, randomized clinical trial switching the third drug to maraviroc was safe & overall effective and improved triglycerides & total cholesterol levels.
It must be that, although the inclusion criteria did not restrict for previous NNRTI or PI use, all subjects switched to maraviroc in this study were previously receiving PIs.
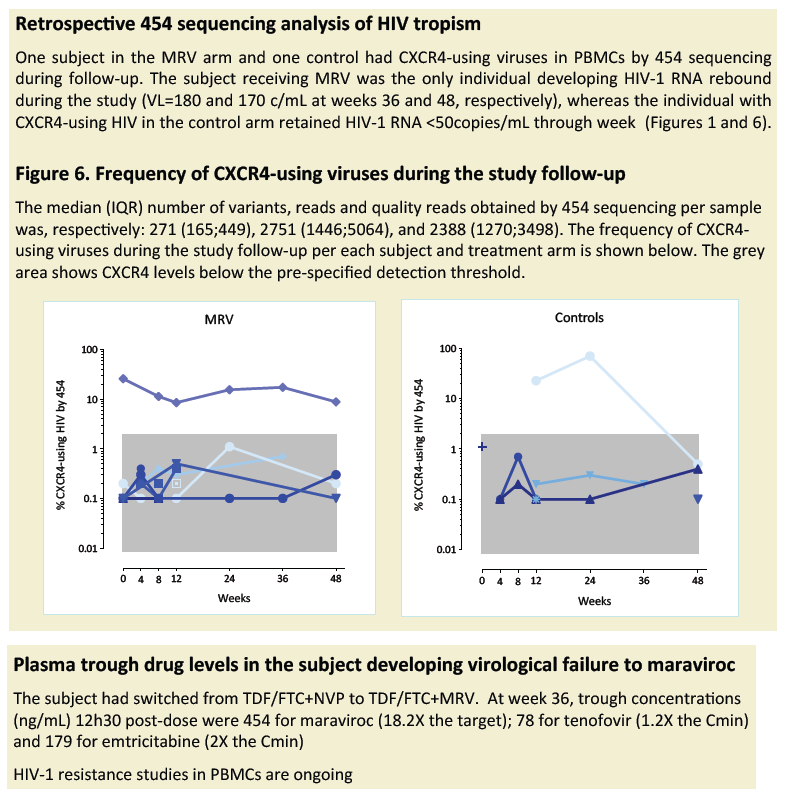
The only subject developing low-level viral-load rebound under maraviroc had non-R5 HIV missed by population sequencing. However, CXCR4-levels remained stable
in this subject under maraviroc treatment selective pressure. Plasma trough levels of TDF, FTC and MRV were adequate. This study should inform larger clinical trials
to validate the clinical utility of MRV switching in subjects with suppressed HIV-1 RNA levels.

|
| |
|
 |
 |
|
|