 |
 |
 |
| |
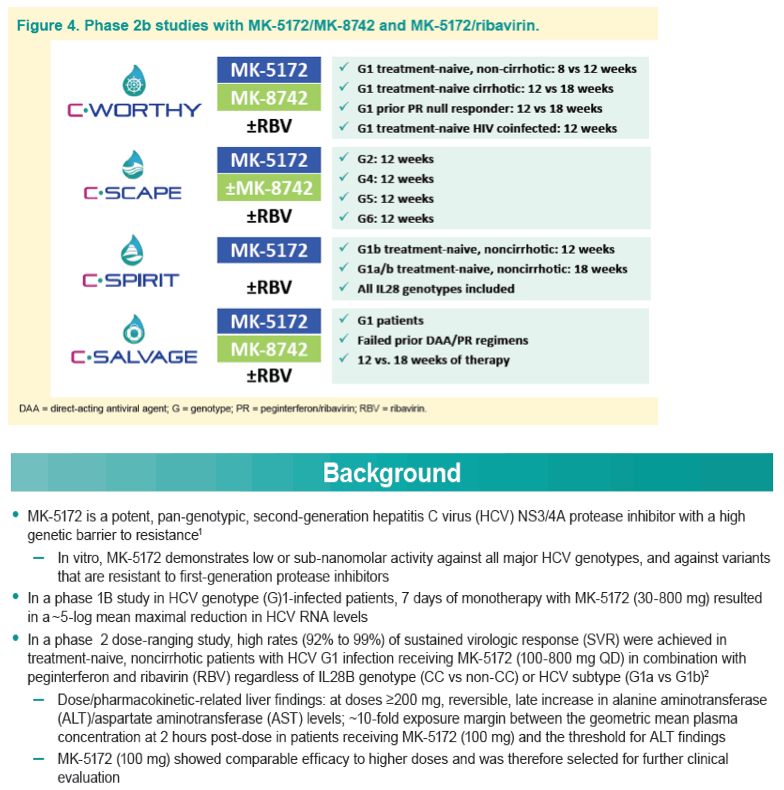
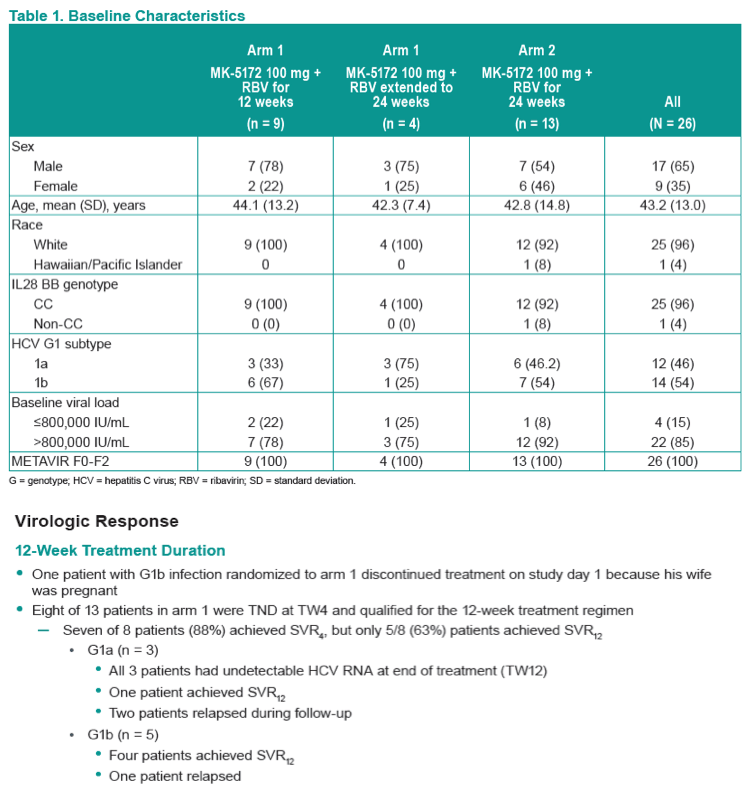
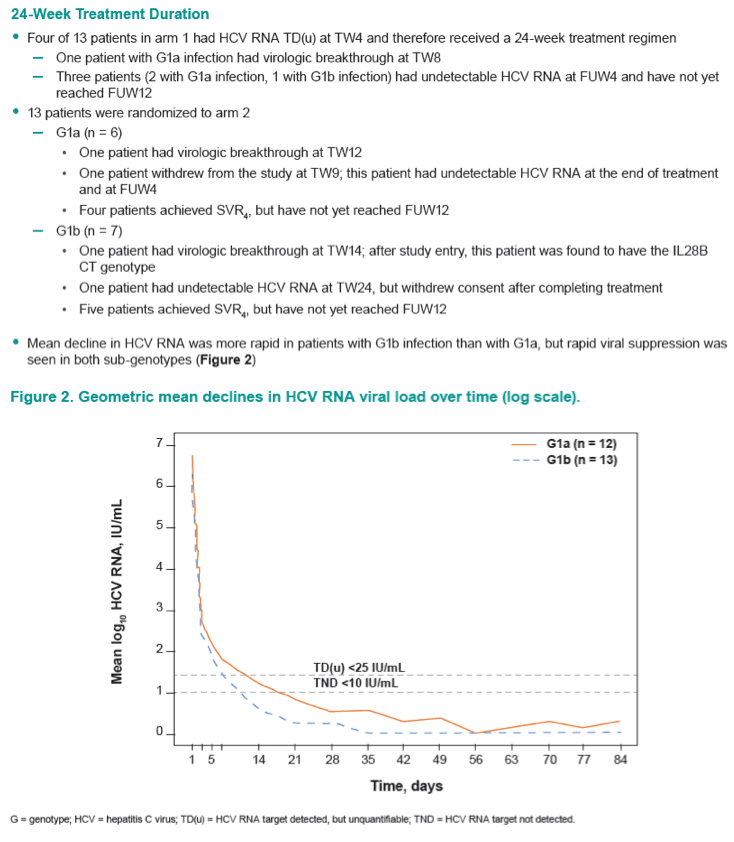
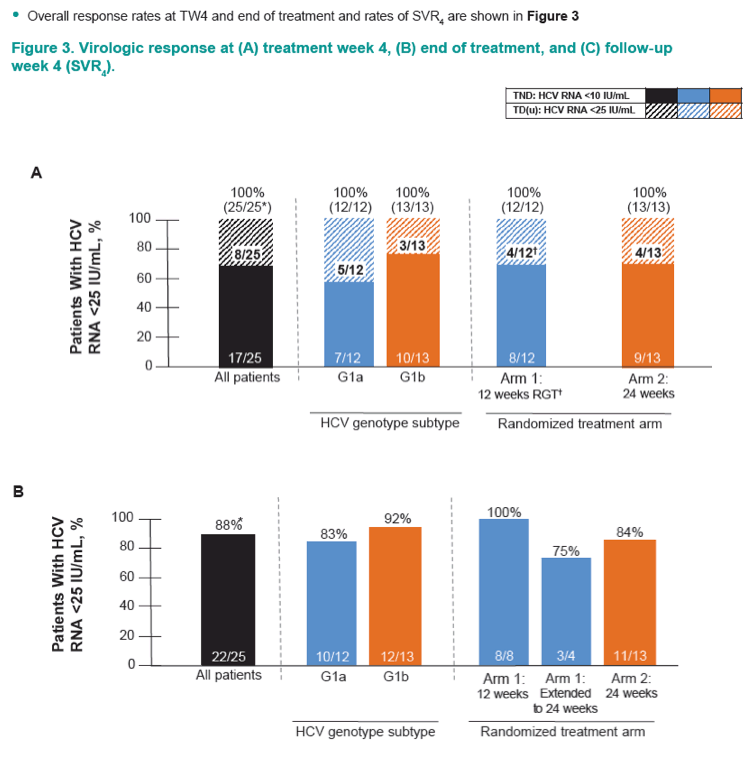
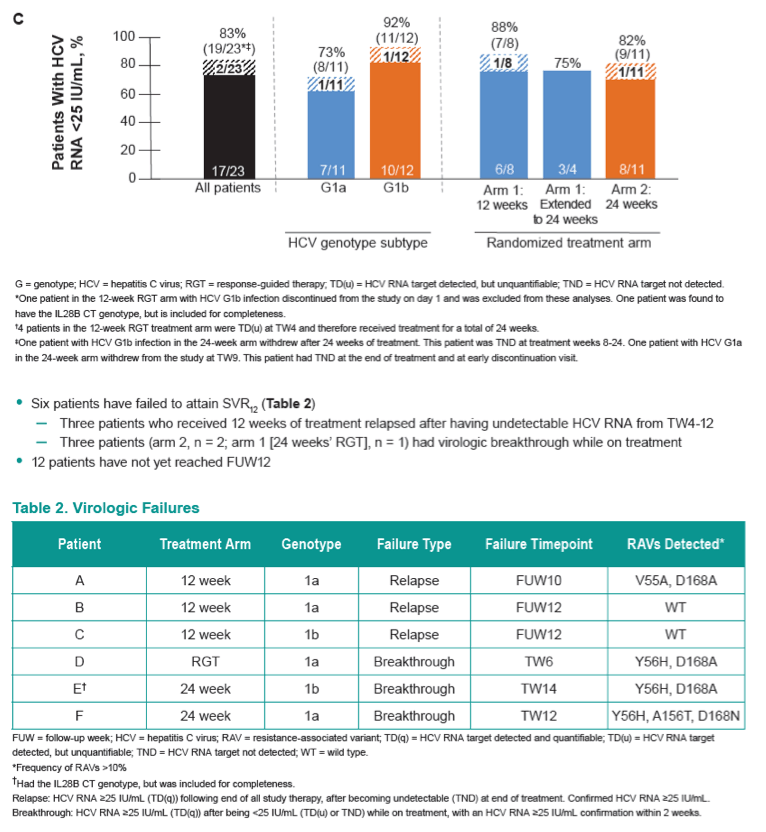
Efficacy and Safety of an Interferon-Free Regimen of MK-5172 + Ribavirin for 12 Weeks or 24 Weeks in Treatment-Naive, Noncirrhotic Subjects With HCV GT1 Infection: The C-SPIRIT Study
|
| |
| |
Reported by Jules Levin
AASLD 2013 Oct 1-4 Wash DC
Edward Gane,1 Ziv Ben Ari,2 Lindsay Mollison,3 Eli Zuckerman,4 Rafael Bruck,5 Yaacov Baruch,6 Janice Wahl,7 Sanhita Bhanja,7 Peggy Hwang,7 Yue Zhao,7 Michael N. Robertson7
1Auckland Clinical Studies, Grafton, Auckland, New Zealand; 2 Liver Disease Center, Sheba Medical Center, Ramat Gan, Israel; 3Fremantle Hepatitis Services, Fremantle Hospital, Fremantle, Australia; 4Liver Unit, Carmel Medical Center, Technion Faculty of Medicine, Haifa, Israel;
5Department of Gastroenterology and Liver Diseases, Tel Aviv Sourasky Medical Center, Tel Aviv, Israel; 6Liver-Unit, Rambam Health Care Campus, Haifa, Israel; 7Merck & Co., Inc, Whitehouse Station, NJ, USA




|
| |
|
 |
 |
|
|