 |
 |
 |
| |
MK-8742, an HCV NS5A Inhibitor With a Broad Spectrum of HCV
Genotypic Activity, Demonstrates Potent Antiviral Activity in Genotype-1 and -3 HCV-Infected Patients
|
| |
| |
Reported by Jules Levin
Presented at the 64th Annual Meeting of the American Association for the Study of Liver Diseases, November 1 - 5, 2013, Washington, DC
Wendy W. Yeh,1 Concetta Lipardi,1 Patricia Jumes,1 Inge De Lepeleire,1 Luzelena Caro,1 Xiaobi Huang,1 Eric Mangin,1Robert B. Nachbar,1 Edward Gane,2 Serghei Popa,3 Neli Ghicavii,3 Frank Wagner,4 Joan R. Butterton1
1Merck & Co., Inc, Whitehouse Station, NJ, USA, 2Auckland City Hospital, Auckland, NZ, 3Republican Clinical Hospital, Chisinau, Moldova, 4Charité Research Organisation GmbH, Berlin, Germany


ABSTRACT
Background: MK-8742 is an inhibitor of Hepatitis C Virus (HCV) non-structural protein 5A (NS5A) that is being developed for the treatment of HCV infection. MK-8742 has broad, potent HCV genotypic activity in vitro against viral variants that are resistant to other NS5A inhibitors in development. A Phase 1b, randomized, placebo-controlled study was conducted to assess the safety, pharmacokinetics and antiviral activity of MK-8742 administered as 5 days of monotherapy in patients with genotype (GT) -1 or -3 HCV infection.
Methods: 48 adult males, with HCV RNA >105 IU/mL and GT1 or GT3 HCV infection without clinical evidence of cirrhosis, were randomized to receive placebo or MK-8742 from 5 to 50 mg (GT1) or 10 to 100 mg (GT3) once daily for 5 days (MK-8742: placebo ratio of 5:1/panel). Safety and tolerability were evaluated using laboratory values, ECGs, and evaluation of adverse experiences (AEs). Antiviral efficacy was assessed using the Roche Cobas TaqMAN 2.0 plasma HCV RNA assay (lower limit of quantitation = 25 IU/mL).
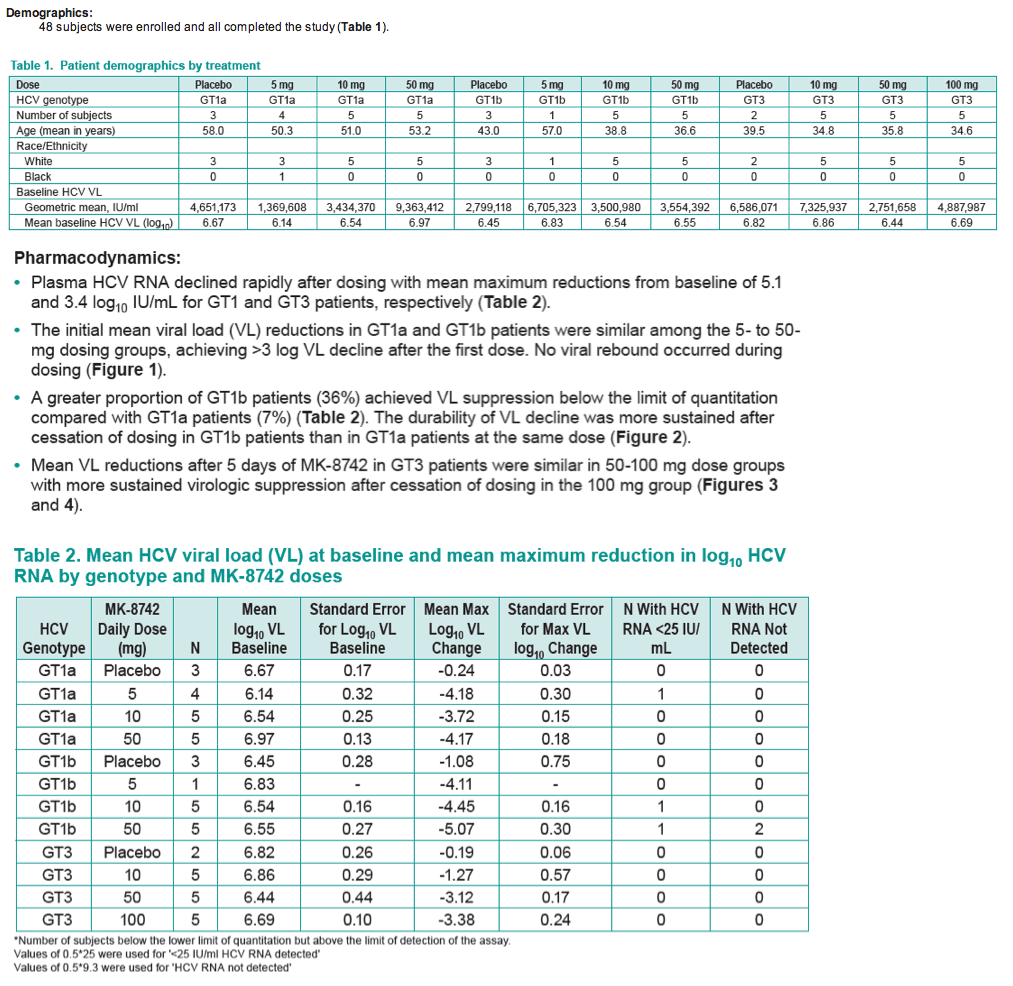
Results: Plasma HCV RNA declined rapidly after dosing with mean maximum reductions from baseline of 5.1 and 3.4 log10 IU/mL for GT1 and GT3 patients, respectively.
The initial mean viral load (VL) reductions in GT1a and GT1b patients were similar among the 5- to 50-mg dosing groups, achieving >3 log VL decline after the first dose.
No viral rebound occurred during dosing.
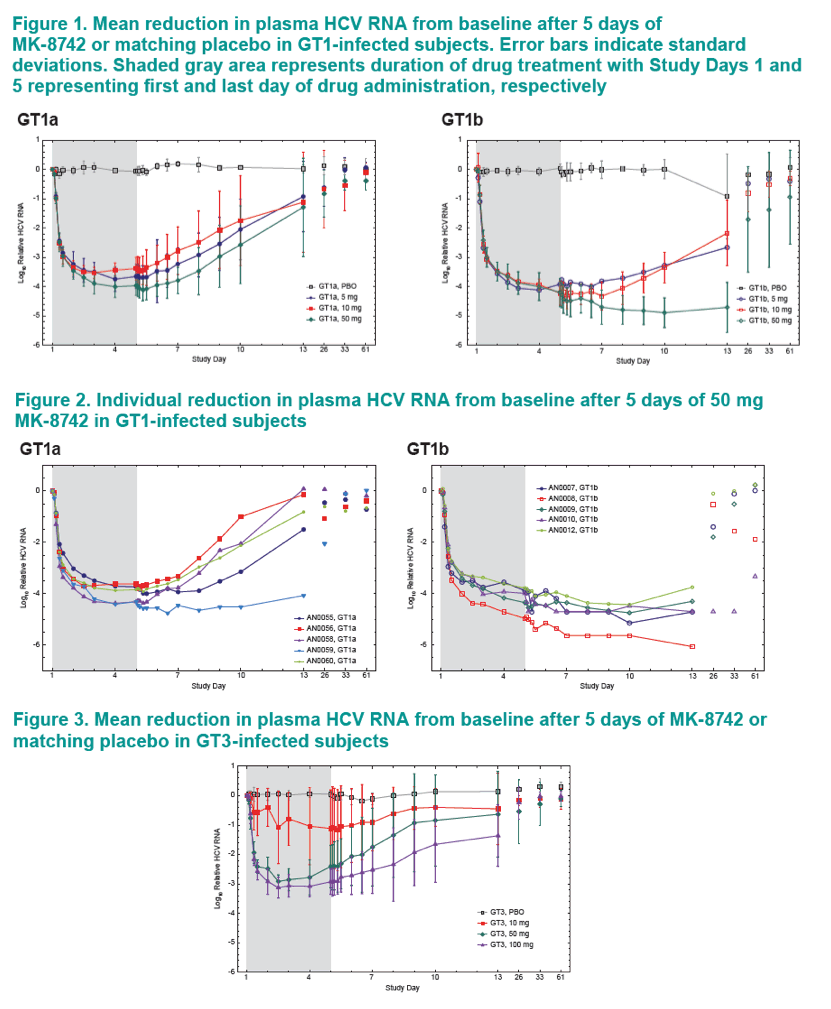
A greater proportion of GT1b patients achieved VL suppression below the limit of quantitation compared to G1a patients.
The durability of VL decline was more sustained after cessation of dosing in GT1b patients than in GT1a patients at the same dose.
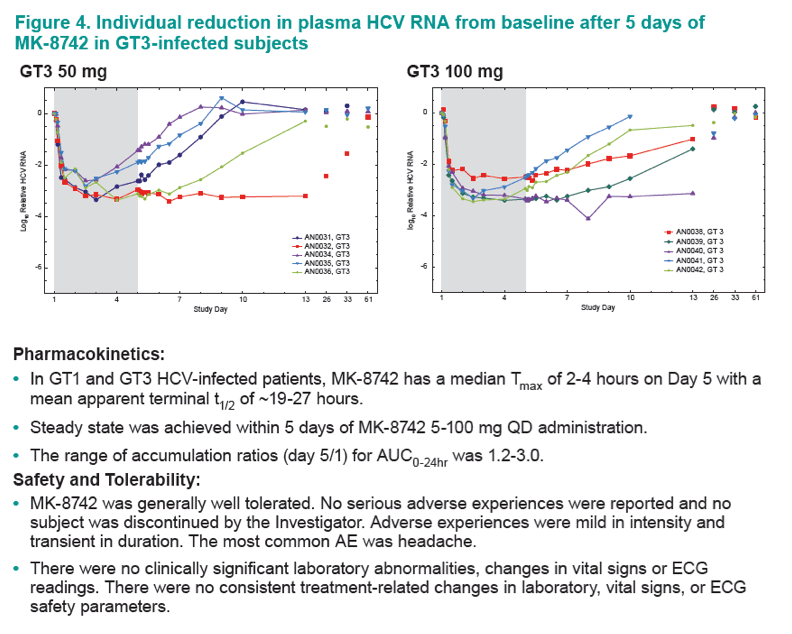
Mean VL reductions after 5 days of MK-8742 in GT3 patients were similar in 50-100 mg dose groups with more sustained virologic suppression after cessation of dosing in the 100 mg group. MK-8742 has a median Tmax of 2-4 hours on Day 5 with a mean apparent terminal t1/2 of ~19-27 hours.
Steady state was achieved within 5 days of MK-8742 5-100 mg QD administration.
The range of accumulation ratios (day 5/1) for AUC0-24hr was 1.2-3.0.
MK-8742 was generally well tolerated, with all AEs transient and mild in intensity. The most common AE was headache. There were no clinically significant laboratory abnormalities, changes in vital signs or ECG readings.
Conclusions: MK-8742 exhibits potent antiviral activity during 5 days of monotherapy in patients with GT1 and GT3 chronic HCV infection. The safety, pharmacokinetics, and antiviral data support the continued clinical investigation of MK-8742 as a once-daily component of an all-oral, interferon-free regimen for the treatment of chronic HCV infection.

|
| |
|
 |
 |
|
|