 |
 |
 |
| |
Simeprevir (TMC435) with pegylated interferon-a-2a/ribavirin for treatment of chronic HCV genotype 1 infection in patients who relapsed after previous interferon-based therapy: efficacy and safety in patient sub-populations in the PROMISE Phase III trial
|
| |
| |
Reported by Jules Levin
AASLD 2013 Nov 1-4 Wash DC
Xavier Forns,1 Eric Lawitz,2 Stefan Zeuzem,3 Edward J Gane,4 Jean-Pierre Bronowicki,5 Pietro Andreone,6 Andrzej Horban,7 Ashley S Brown,8 Monika Peeters,9 Oliver Lenz,9 Sivi Ouwerkerk-Mahadevan,10
Jane A Scott,11 Ronald Kalmeijer,12 Guy De La Rosa,12 Rekha Sinha,9 Maria Beumont-Mauviel9
1Liver Unit, Hospital Clinic, Institut d'Investigacions Biomèdiques August Pi i Sunyer (IDIBAPS) and Centro de Investigación Biomédica en Red de Enfermedades Hepáticas y Digestivas (CIBEREHD), Barcelona, Spain; 2Texas Liver Institute, University of Texas Health Science Center, San Antonio, TX, USA; 3J.W. Goethe University Hospital, Frankfurt, Germany; 4Auckland Hospital Clinical Studies Unit, Auckland, New Zealand; 5INSERM U954, Universite de Lorraine, Centre Hospitalier Universitaire de Nancy, Vandoeuvre Les Nancy, France; 6Dipartimento di Scienze Mediche e Chirurgiche, University of Bologna, Italy; 7Medical University of Warsaw, Wolska, Warsaw, Poland; 8Imperial College Healthcare NHS Trust, London, United Kingdom; 9Janssen Infectious Diseases BVBA, Beerse, Belgium; 10Janssen Research & Development, Beerse, Belgium; 11Janssen Global Services LLC, High Wycombe, UK; 12Janssen Global Services LLC, Titusville, NJ, USA







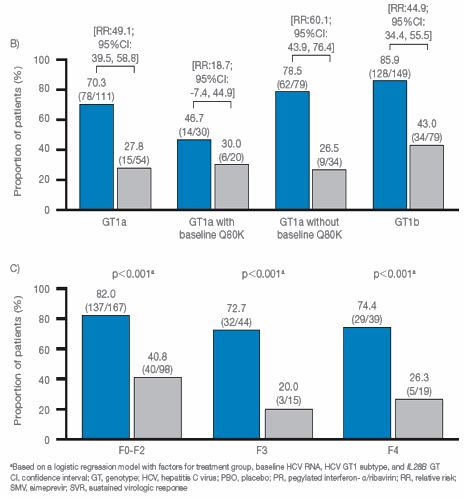
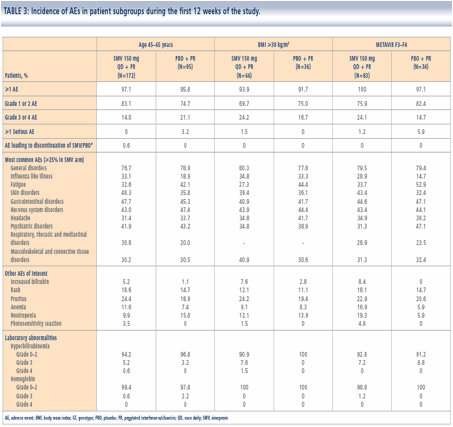
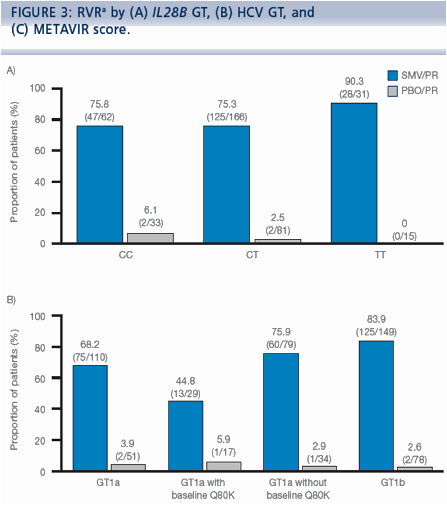
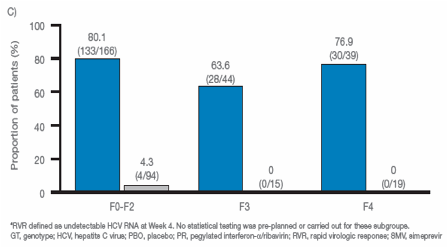

|
| |
|
 |
 |
|
|