 |
 |
 |
| |
Safety and Efficacy of BMS-791325, a Non-Nucleoside NS5B Polymerase Inhibitor, Combined With
Peginterferon Alfa-2a and Ribavirin in Treatment-Naive Patients Infected with Hepatitis C Virus Genotype 1
|
| |
| |
Reported by Jules Levin
AASLD 2013 Nov 1-4 Wash DC
Tatum H,1 Thuluvath PJ,2 Lawitz E,3 Martorell C,4 Cohen SM,5 Rustgi V,6 Ravendhran N,7 Ghalib R,8 Hanson J,9 Zamparo J,10 Zhao J,11 Treitel M,12 Hughes E12
1Options Health Research, Tulsa, OK, USA; 2Mercy Medical Center, Baltimore, MD, USA; 3The Texas Liver Institute, University of Texas Health Science Center, San Antonio, TX, USA; 4The Research Institute, Springfield, MA, USA;
5University Hospital/Case Western Medical Center, Cleveland, OH, USA; 6Metropolitan Research, Arlington, VA, USA; 7Digestive Disease Associates, Baltimore, MD, USA; 8Texas Clinical Research Institute, Arlington, TX, USA;
9Charlotte Gastroenterology and Hepatology, Charlotte, NC, USA; 10Bristol-Myers Squibb, Wallingford, CT, USA; 11Bristol-Myers Squibb, Hopewell, NJ, USA; 12Bristol-Myers Squibb, Princeton, NJ, USA
Phase 3 Study:
http://www.clinicaltrials.gov/ct2/show/NCT01973049?term=bms-791325&rank=2v
AASLD: Phase 2b Study of the Interferon-Free and Ribavirin-Free Combination of Daclatasvir, Asunaprevir, and BMS-791325 for 12 Weeks in Treatment-Naive Patients With Chronic HCV Genotype 1 Infection - (11/05/13)


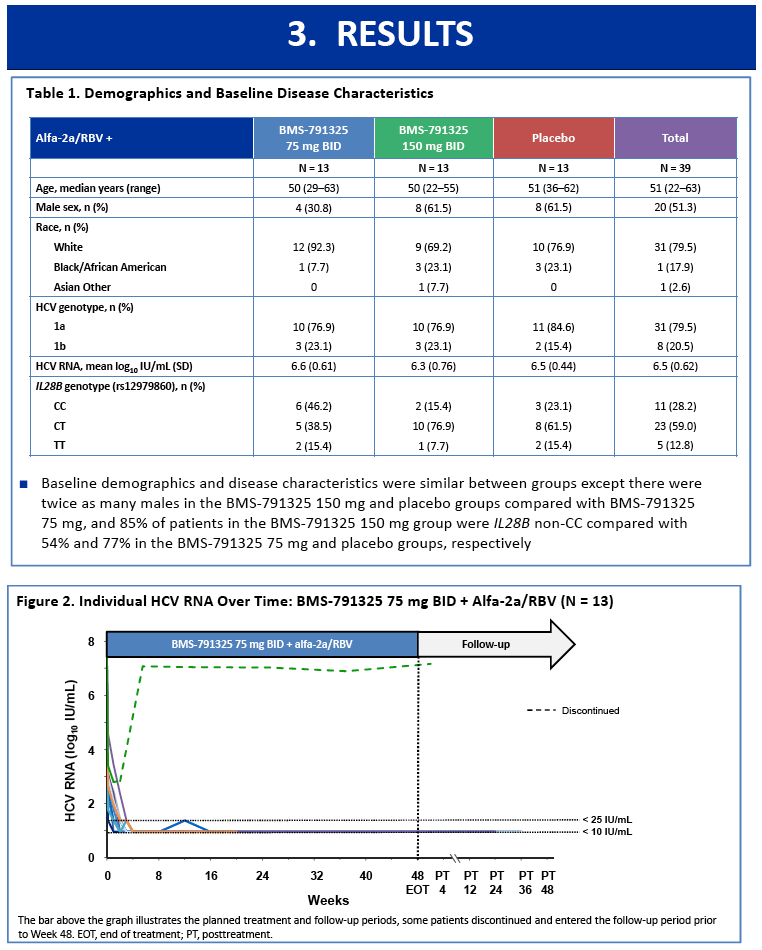
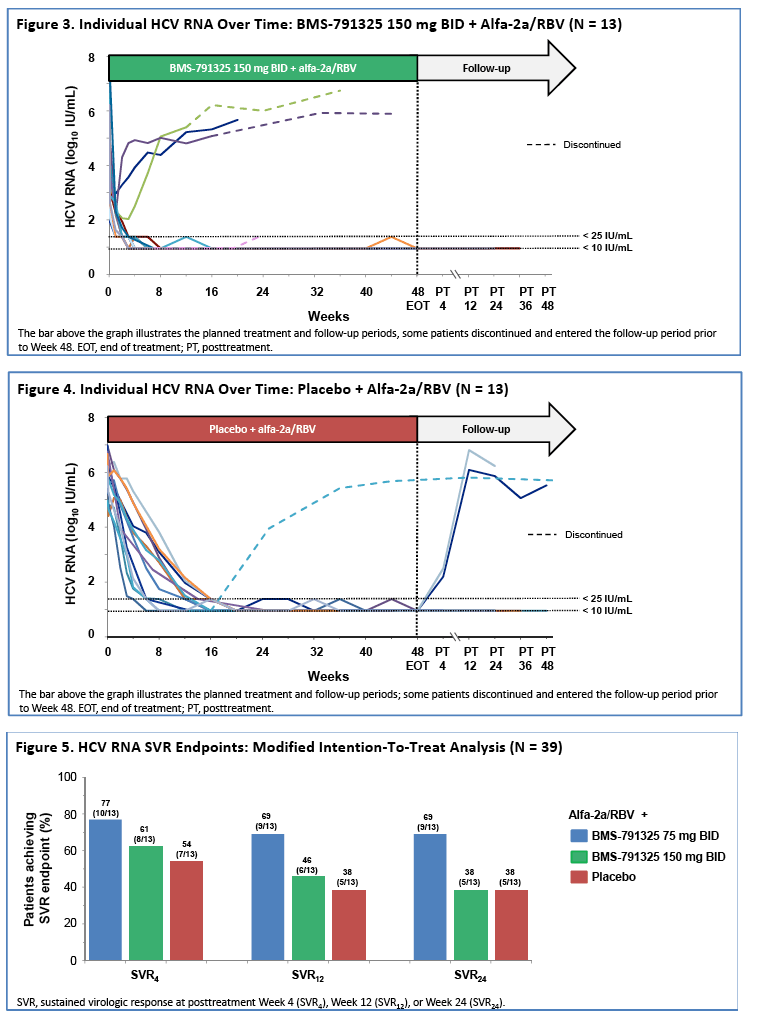
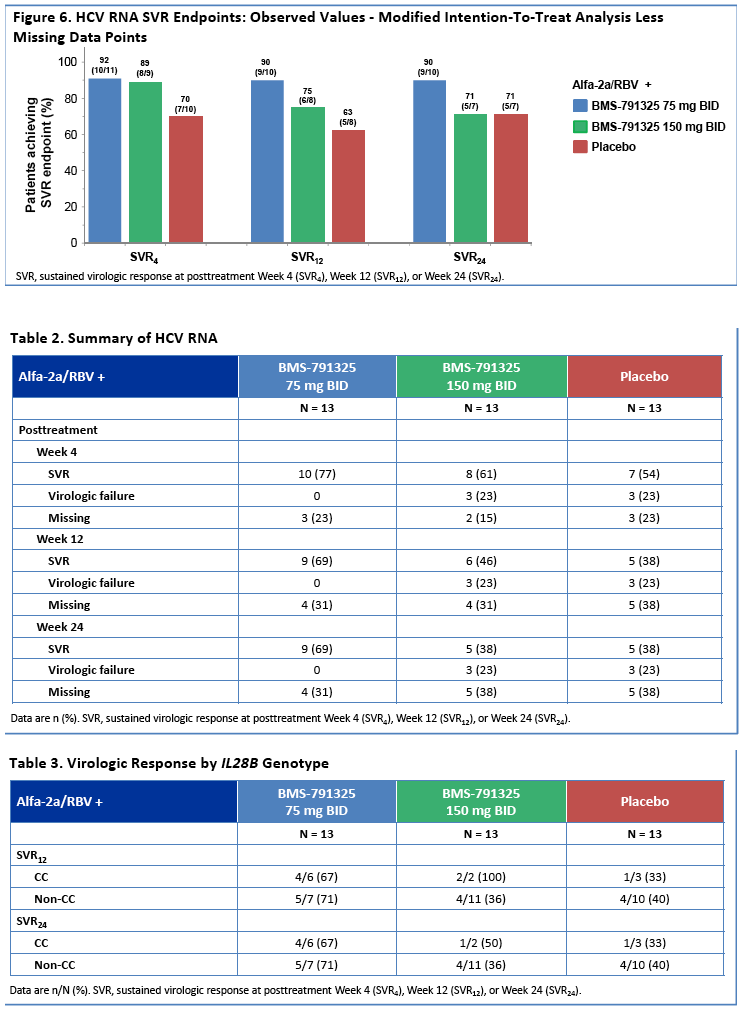
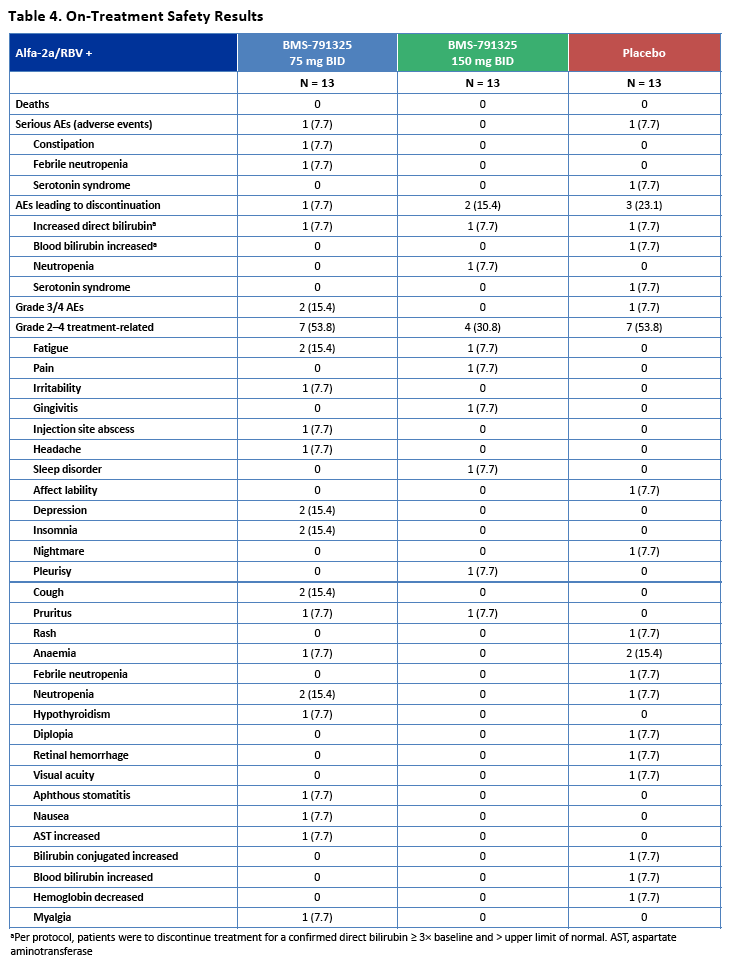
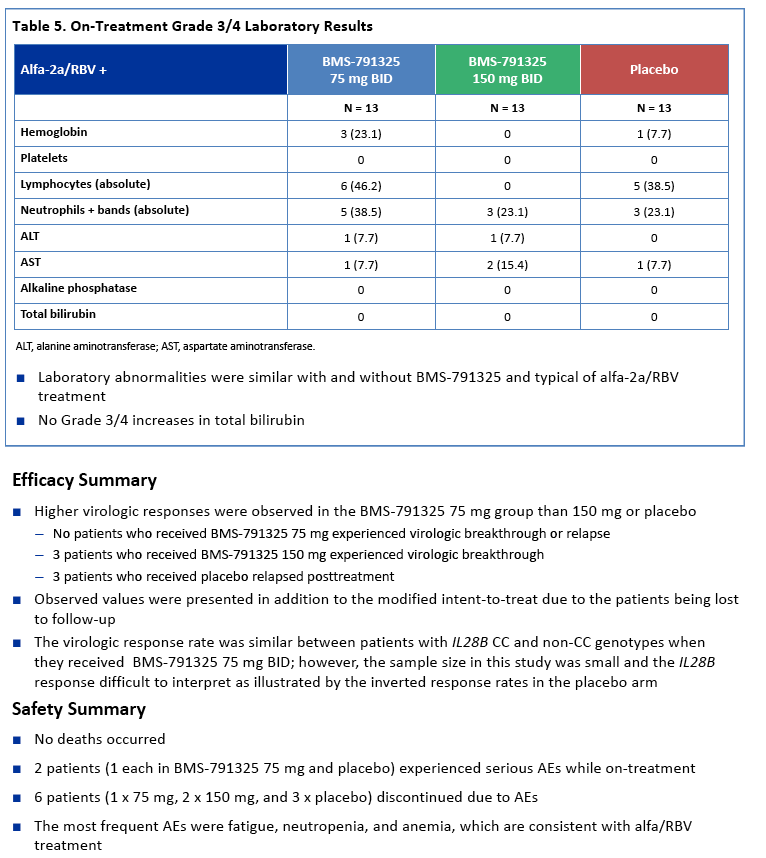

|
| |
|
 |
 |
|
|