 |
 |
 |
| |
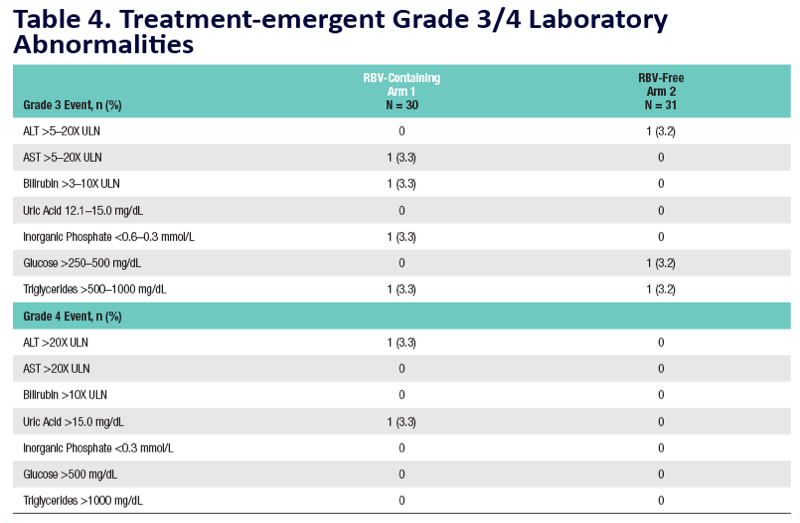
A 12-Week Trial of Interferon-free Regimens Containing ABT-450/r and ABT-267 ± Ribavirin (RBV) in Treatment-na•ve Patients With HCV Genotypes 1-3
|
| |
| |
Reported by Jules Levin
Presented at the 23rd Conference of the Asian Pacifi c Associati on for the Study of the Liver, 6-9 June, 2013, Singapore
Eric Lawitz1, Greg Sullivan2, Maribel Rodriguez-Torres3, Pedram Enayati 4, Michael Bennett 5, Fred Poordad1, Mudra Kapoor6, Andrew Campbell6, Lino Rodrigues-Jr6, Prajakta Badri6, Yiran Hu6, Tami Pilot-Mati as6, Barry Bernstein6
1The Texas Liver Insti tute, University of Texas Health Science Center, San Antonio, Texas, United States; 2Parkway Medical Center, Birmingham, Alabama, United States; 3Fundacion de Investigacion, San Juan, Puerto Rico; 4Cedars-Sinai Medical Center, Los Angeles, California, United States; 5Medical Associates Research Group, San Diego, California, United States; 6AbbVie Inc., North Chicago, Illinois, United States


|
| |
|
 |
 |
|
|