 |
 |
 |
| |
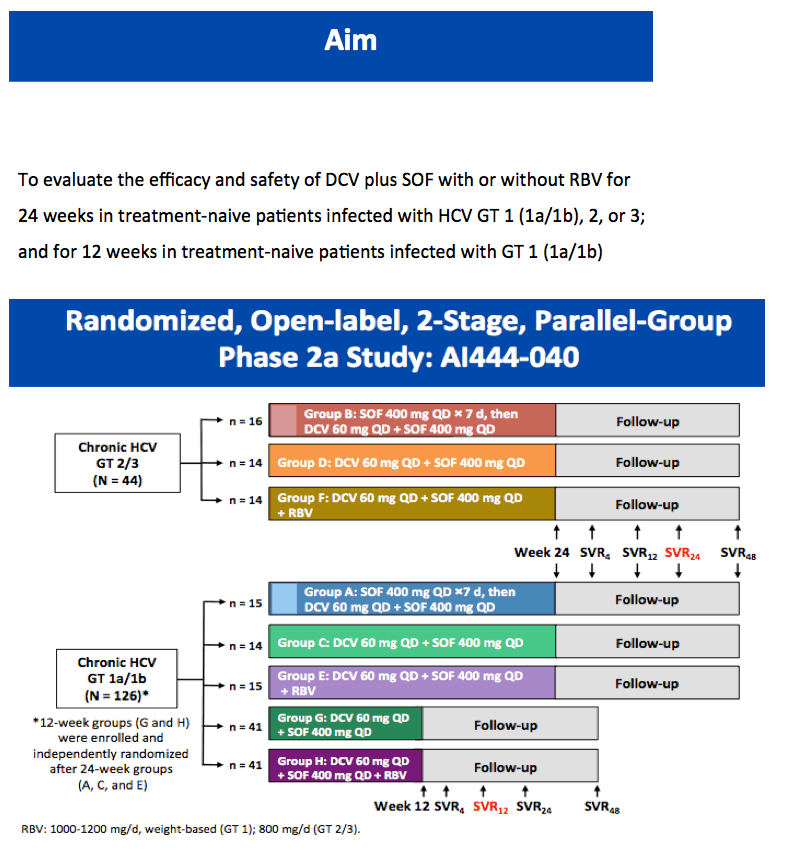
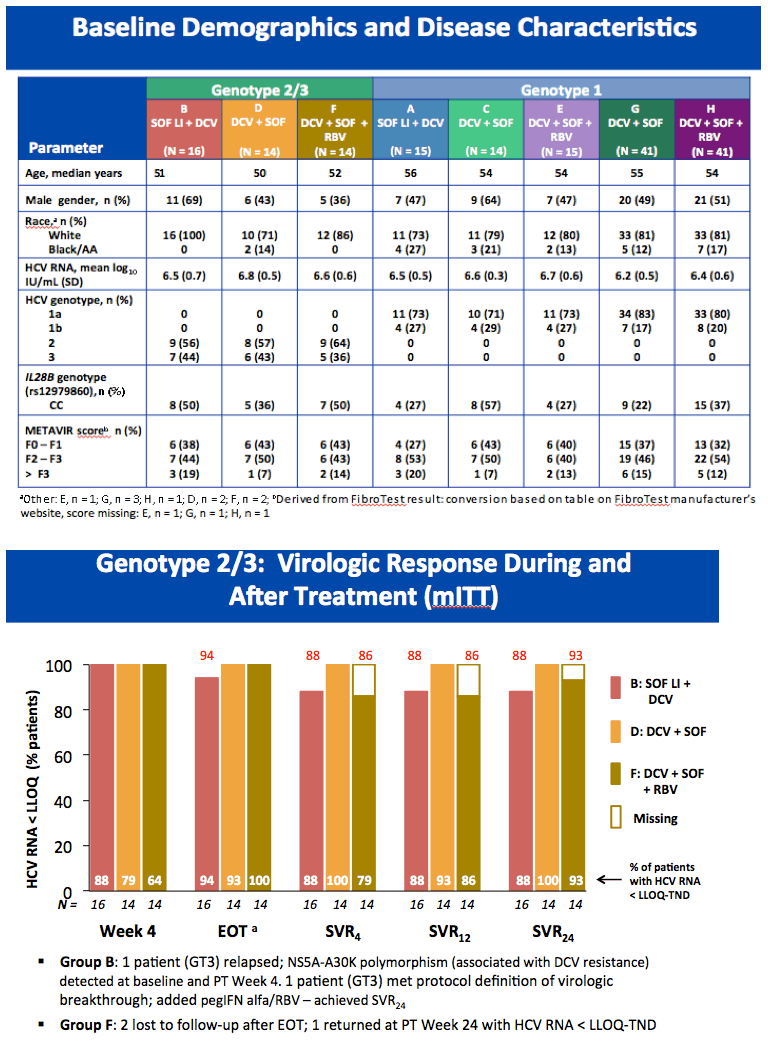
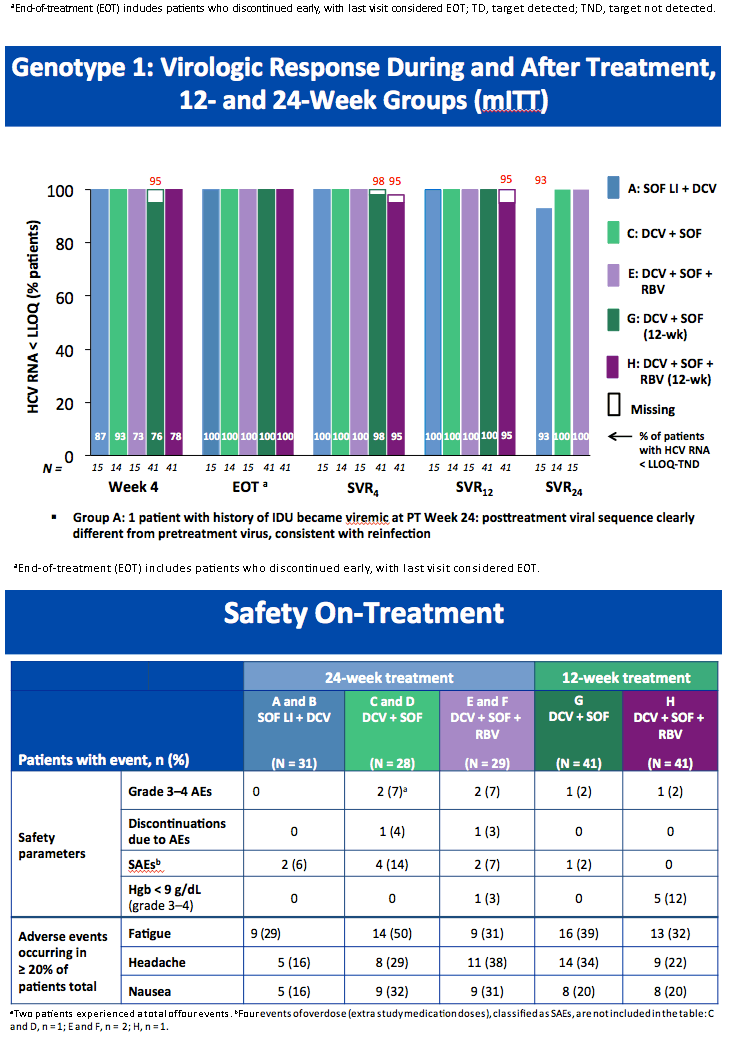
High Rate of Sustained Virologic Response With the All-Oral Combination of Daclatasvir (NS5A Inhibitor) Plus Sofosbuvir (Nucleotide NS5B Inhibitor), With or Without Ribavirin, in Treatment-Naive Patients Chronically Infected With HCV GT 1, 2, or 3 (updated SVR12 reported, previously at AASLD/2012 SVR4 was reported for 12-week regimen)
|
| |
| |
Reported by Jules Levin
APASL Liver Week 2013: The 2013 Annual Meeting of the
Asian Pacific Association for the Study of the Liver
Singapore, June 6-10, 2013
AASLD/2012: High Rate of Sustained Virologic Response With the All-Oral Combination of Daclatasvir (NS5A Inhibitor) Plus Sofosbuvir (Nucleotide NS5B Inhibitor), With or Without Ribavirin, in Treatment-Naive Patients Chronically Infected With HCV GT 1, 2, or 3.......http://www.natap.org/2012/AASLD/AASLD_06.htm
Sulkowski MS, Gardiner DF, Rodriguez-Torres M, Reddy KR, Hassanein T, Jacobson IM, Lawitz E, Lok AS, Hinestrosa F, Thuluvath PJ, Schwartz H, Nelson DR, Eley T,
Wind-Rotolo M, Huang S-P, Gao M, McPhee F, Sherman D, Hindes R, Symonds W, Pasquinelli C, and Grasela DM for the AI444040 Study Group



|
| |
|
 |
 |
|
|