 |
 |
 |
| |
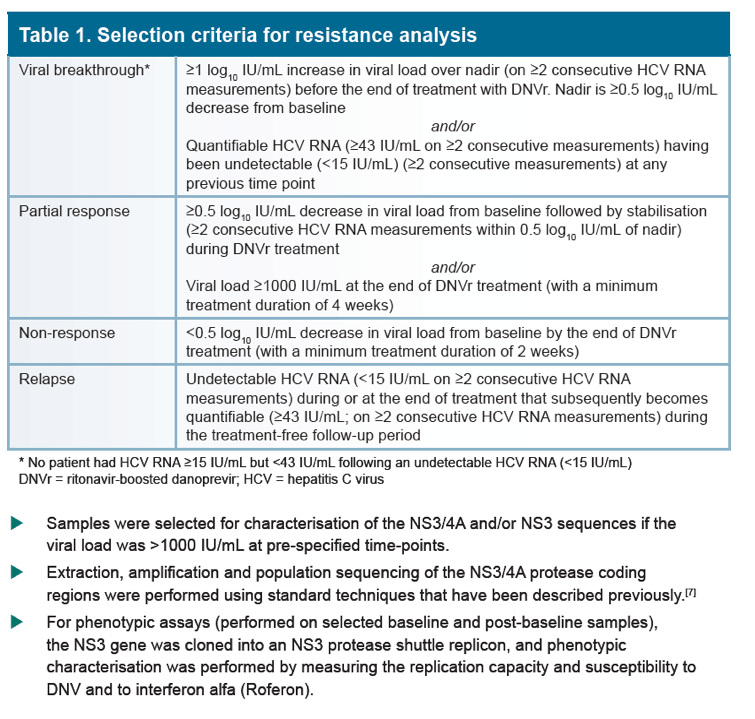
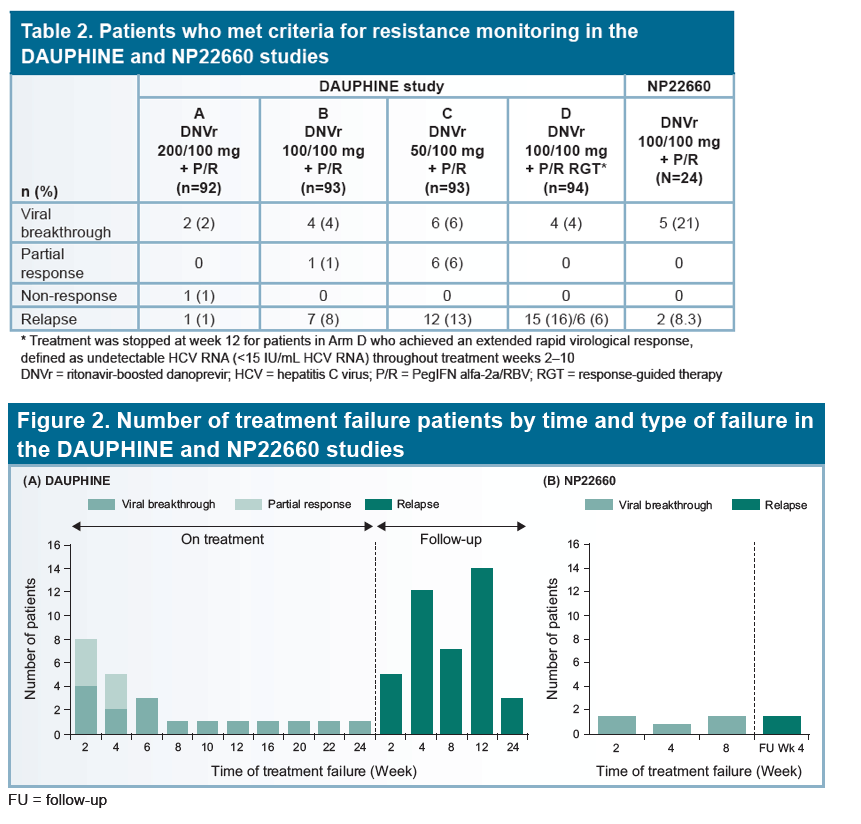
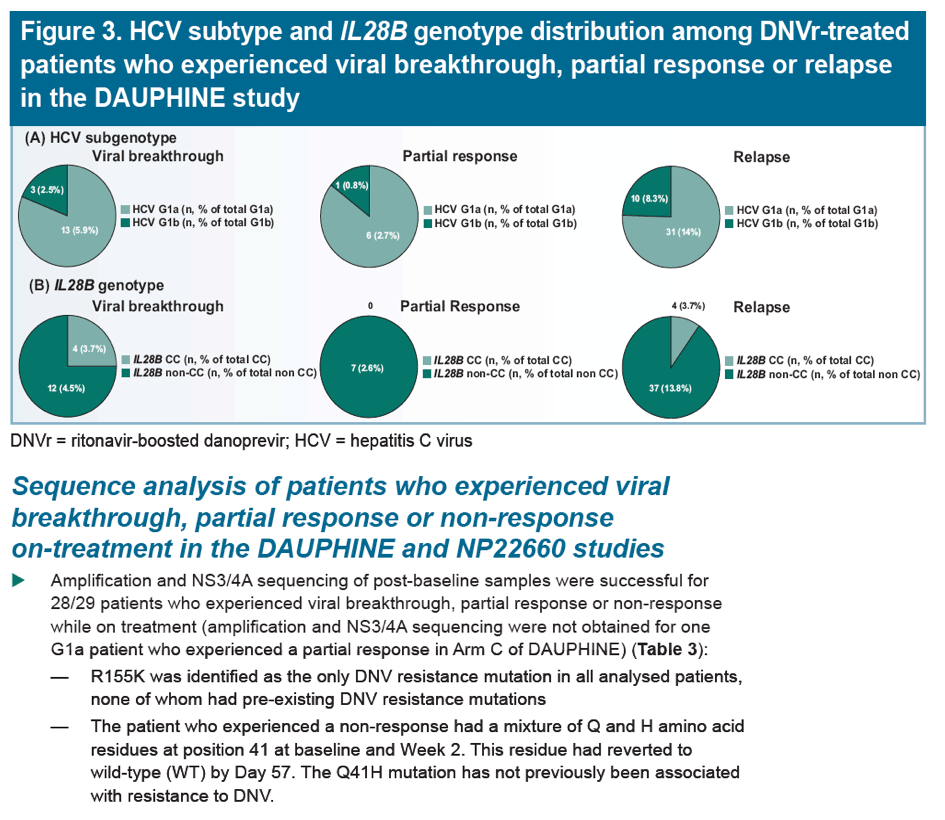
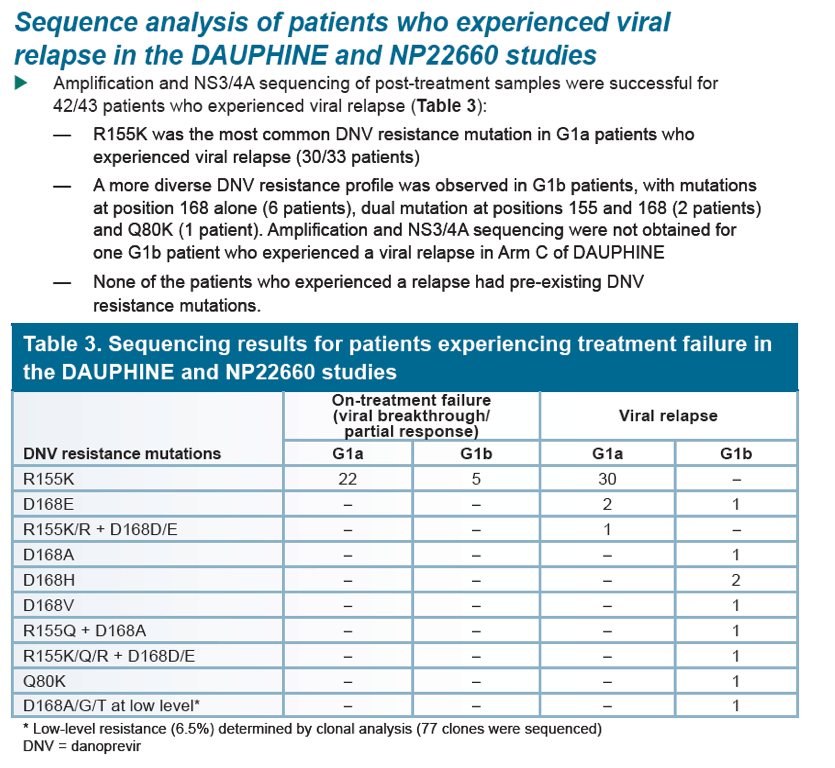
Low rate of danoprevir resistance with ritonavir-boosted danoprevir (DNVr) plus PegIFN alfa-2a/ribavirin (P/R) in HCV G1b-infected treatment-naive and treatment-experienced patients
|
| |
| |
Reported by Jules Levin
APASL 2013 Singapore June 6-10
S. Le Pogam,1 L. Li,1 M.T. Navarro,2 J. Zhou,3 A. Voulgari,4 N.S. Shulman,2 K. Klumpp,1 I. Najera1
1Hoffmann-La Roche Inc, Nutley, NJ, USA; 2Genentech, South San Francisco, CA, USA; 3Roche Product Development - Asia Pacific, Shanghai, China; 4Roche Products Ltd, Welwyn, UK
APASL: High SVR24 rates with ritonavir-boosted danoprevir (DNVr) plus PegIFN alfa-2a/ribavirin (P/R) in HCV genotype (G)1/4-infected treatment-naive patients: the DAUPHINE study - (06/09/13)
3 & 4 Oral IFN-Free Roche HCV Regimens Studies, Treatment Naïve or Null Responders: setrobuvir+mericitabine+danoprevir/r+rbv. ANNAPURNA Study
http://www.natap.org/2012/HCV/090412_01.htm



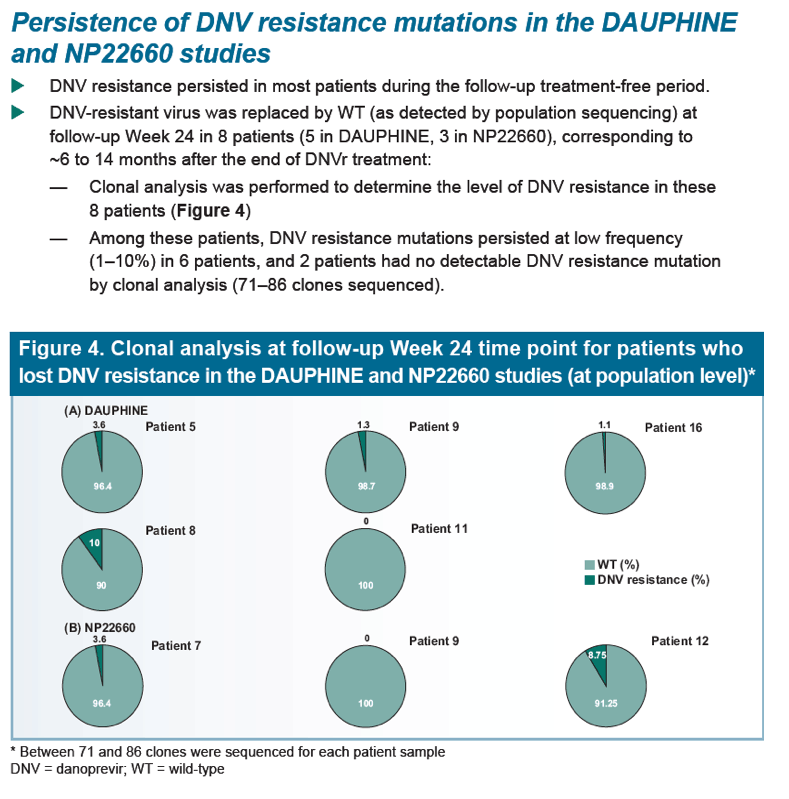
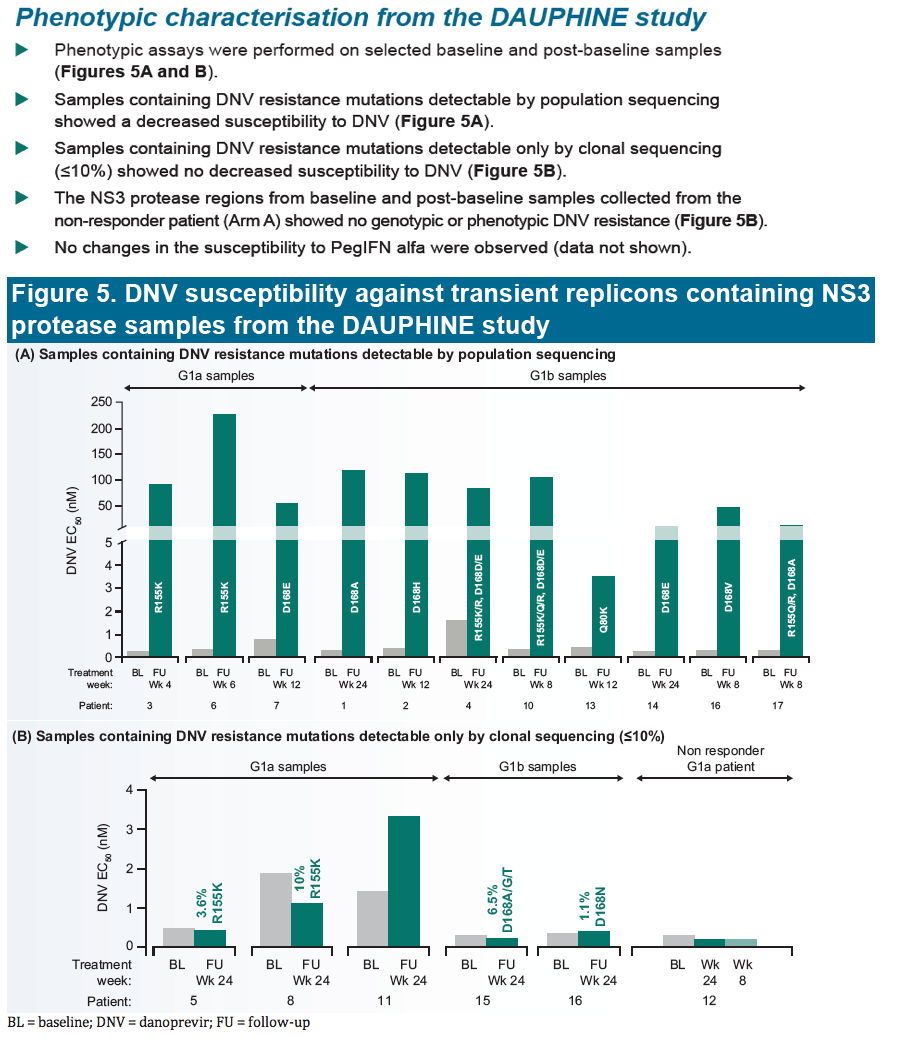

|
| |
|
 |
 |
|
|