| |
A Comparison of the Pharmacokinetics of Darunavir,
Atazanavir and Ritonavir during Pregnancy and Post-partum
|
| |
| |
Reported by Jules Levin
CROI 2012 Seattle, WA
Angela Colbers1, Jose Molto2, Jelena Ivanovic3, Carmen Hidalgo Tenori4, Katharina Weizsacker5, Ineke van der Ende6, David
Hawkins7, Graham Taylor8, Tariq Sadiq9, David Burger1, on behalf of the PANNA network
1Department of Pharmacy, Radboud University Nijmegen Medical Centre, Nijmegen, The Netherlands; 2 "Lluita contra la Sida" Foundation, Hospital Universitari Germans Trias i Pujol, Badalona, Spain;
3National Institute for Infectious Diseases "L. Spallanzani", Rome, Italy; 4Hospital Universitario Virgen de las Nieves Granada, Granada, Spain; 5CHARITe Berlin, Berlin, Germany; 6 Erasmus Medical Center
Rotterdam, Rotterdam, The Netherlands; 7Chelsea & Westminster Hospital, London, UK; 8St Mary's Hospital, Imperial College Healthcare NHS Trust, London, UK; 9St George's hospital, London, UK

ABSTRACT (updated)
· Background: It is important to achieve effective concentrations of antiretroviral drugs in the blood to prevent treatment failure and the development of resistance. During pregnancy physiological changes take place influencing the pharmacokinetics (PK) of medicines.
Mostly, the net effect will be a decreased exposure during pregnancy. Very limited data are available on the PK behavior of darunavir (DRV) during pregnancy and placental passage and for atazanavir (ATV) the data have been interpreted inconsistently. In 2008, a European network was established to study the pharmacokinetics of newly developed antiretroviral drugs during pregnancy (PANNA). We present data on 3rd trimester exposure to DRV, ATV and ritonavir (RTV, used as booster).
· Materials & Methods: Patients treated with DRV/RTV (600mg/100mg BID or 800mg/100mg QD) or ATV/RTV (300mg/100mg QD) during pregnancy were screened and a pharmacokinetic curve (t = 0, 0.5, 1, 2, 3, 4, 6, 8, 12, 24h) was taken in the 3rd trimester and at least 2 weeks post-partum. Where possible a cord blood sample and matching maternal blood sample were taken at delivery. Safety and antiviral efficacy were evaluated.
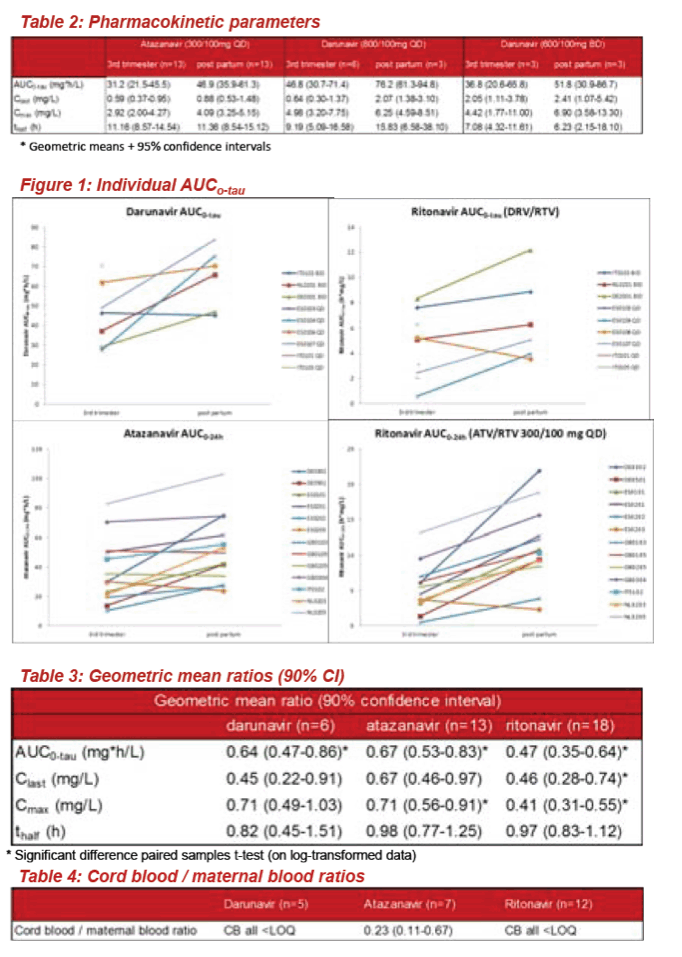
· Results: Paired plasma concentration curves (3rd trimester and post-partum) were available for 6 patients on DRV; 13 patients on ATV and 19 patients using RTV as booster. For DRV, geometric means ratios (GMR) + 90% confidence interval (CI) of PK parameters
3rd trimester/post-partum DRV were: 0.64 (0.47-0.86) for AUCtau; 0.71 (0.49-1.03) for Cmax; 0.45 (0.22-0.91) for Clast. For ATV GMRs (90% CI) were: 0.67 (0.53-0.83) for AUCtau; 0.71 (0.56-0.91) for Cmax; 0.67 (0.46-0.97) for C24h. For RTV GMRs (90% CI) were: 0.47 (0.35-0.64) for AUCtau; 0.41 (0.31-0.55) for Cmax. For DRV 2/9 patients showed concentrations below the target concentration (0.55 mg/L) in the 3rd trimester, compared to none on ATV (target 0.15 mg/L).
The ratio of cord blood/maternal plasma concentrations ranged from 0.11-0.67 for ATV (n=7) and was <0.076 for DRV (n=5); for RTV all cord blood samples were < LOQ (n=12). All children were HIV uninfected, no birth defects were reported.
· Conclusions: Exposure to DRV, ATV and RTV was significantly lower during pregnancy (third trimester) than post-partum, with the most important effect for the boosting agent RTV. This is in line with previously reported exposure to protease inhibitors. Transplacental passage of ATV, DRV and RTV is low. Evidence-based recommendations for ARVs in pregnancy are urgently needed.


| |
| |
| |
|
|
|