| |
Antiretroviral prophylaxis for HIV infection in injecting drug users in Bangkok, Thailand (the Bangkok Tenofovir Study): a randomised, double-blind, placebo-controlled phase 3 trial
|
| |
| |
Download the PDF here
The Lancet, 15 June 2013
Kachit Choopanya, Michael Martin, Pravan Suntharasamai, Udomsak Sangkum, Philip A Mock, Manoj Leethochawalit, Sithisat Chiamwongpaet,
Praphan Kitisin, Pitinan Natrujirote, Somyot Kittimunkong, Rutt Chuachoowong, Roman J Gvetadze, Janet M McNicholl, Lynn A Paxton,
Marcel E Curlin, Craig W Hendrix, Suphak Vanichseni, for the Bangkok Tenofovir Study Group
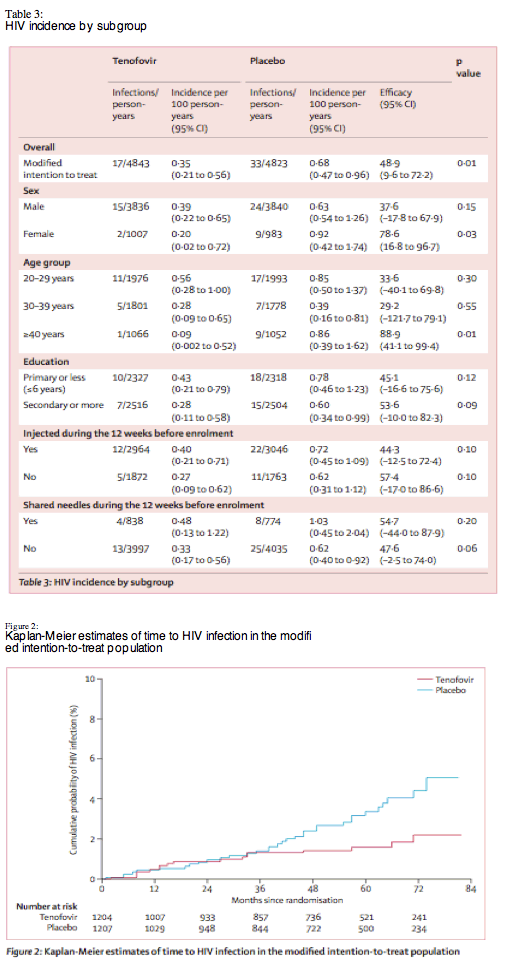
"We confirmed HIV infection in 52 participants (17 [33%] in the tenofovir group, 35 [67%] in the placebo group) indicating a 51·8% reduction in HIV incidence (95% CI 15·3-73·7; p=0·01) in the tenofovir group compared with the placebo group in the intention-to-treat analysis. Excluding the two participants in the placebo group who were HIV-positive at enrolment, HIV incidence was 0·35 per 100 person-years in the tenofovir group and 0·68 per 100 person-years in the placebo group representing a 48·9% reduction in HIV incidence in the modified intention-to-treat analysis (9·6-72·2; p=0·01; table 3). The cumulative probability of HIV infection in the two groups separated consistently after 36 months (figure 2).......Participant reports of injecting drugs and sharing needles decreased during follow-up, consistent with previous trials in people who inject drugs in Bangkok"
"Once-daily oral tenofovir decreased the risk of HIV infection by 48·9% in injecting drug users when provided with other HIV prevention services at drug-treatment clinics in Bangkok. Findings from other pre-exposure prophylaxis trials showed that adherence had an important effect on efficacy.11, 12 In this study, efficacy increased from 46% to 56% in the per-protocol analysis based on observed adherence and to 74% when limited to participants with detectable tenofovir concentrations. Although the trial was not powered to assess efficacy in subgroups, we saw higher efficacy in women (79%) and in participants aged 40 years or older (89%)-two subgroups with high levels of adherence."
"Participants chose either daily directly observed treatment or monthly visits and could switch at monthly visits. Participants received monthly HIV testing and individualised risk-reduction and adherence counselling, blood safety assessments every 3 months, and were offered condoms and methadone treatment. The primary efficacy endpoint was HIV infection, analysed by modified intention-to-treat analysis".
"To our knowledge, this is the first study to show that daily oral pre-exposure prophylaxis with tenofovir, when used in combination with other HIV prevention strategies, reduces the risk of HIV infection in people who inject drugs. Much like findings from other pre-exposure prophylaxis trials, our findings showed that adherence had an important effect on efficacy. On the basis of these findings regulatory and public health authorities can now consider whether pre-exposure prophylaxis with tenofovir should be part of an HIV prevention package to reduce the risk of HIV infection in people who inject drugs."
"Tenofovir and placebo recipients reported similar rates of injecting and sharing needles and similar numbers of sexual partners during follow-up with no interactions between time and treatment group (data not shown). The number of participants reporting injecting drugs during the previous 3 months decreased from 1507 (63%) at enrolment, to 426 (23%) at month 12, and to 117 (18%) at month 72. Reports of sharing needles decreased from 435 (18%) at enrolment to 44 (2%) at month 12, and to eight (1%) at month 72. Sex with more than one partner decreased from 522 (22%) at enrolment to 43 (6%) at month 72 (p<0·0001 for all). Excluding data obtained at enrolment, 1018 (45%) participants reported injecting drugs during the study; 35 (70%) of those who became HIV-positive and 983 (45%) of those who remained HIV-negative during the trial."
"We did not detect tenofovir resistance in HIV-positive participants in this study......As has been reported in other trials,11, 12 participants in the tenofovir group reported more nausea and vomiting in the first couple of months of follow-up than did those in the placebo group. When used for treatment of HIV, tenofovir is associated with small decreases in renal function.26, 27 We did not find higher rates of increased creatinine or renal disease in participants randomly allocated to tenofovir."
Summary
Background
Antiretroviral pre-exposure prophylaxis reduces sexual transmission of HIV. We assessed whether daily oral use of tenofovir disoproxil fumarate (tenofovir), an antiretroviral, can reduce HIV transmission in injecting drug users.
Methods
In this randomised, double-blind, placebo-controlled trial, we enrolled volunteers from 17 drug-treatment clinics in Bangkok, Thailand. Participants were eligible if they were aged 20-60 years, were HIV-negative, and reported injecting drugs during the previous year. We randomly assigned participants (1:1; blocks of four) to either tenofovir or placebo using a computer-generated randomisation sequence. Participants chose either daily directly observed treatment or monthly visits and could switch at monthly visits. Participants received monthly HIV testing and individualised risk-reduction and adherence counselling, blood safety assessments every 3 months, and were offered condoms and methadone treatment. The primary efficacy endpoint was HIV infection, analysed by modified intention-to-treat analysis. This trial is registered with ClinicalTrials.gov, number NCT00119106.
Findings
Between June 9, 2005, and July 22, 2010, we enrolled 2413 participants, assigning 1204 to tenofovir and 1209 to placebo. Two participants had HIV at enrolment and 50 became infected during follow-up: 17 in the tenofovir group (an incidence of 0·35 per 100 person-years) and 33 in the placebo group (0·68 per 100 person-years), indicating a 48·9% reduction in HIV incidence (95% CI 9·6-72·2; p=0·01). The occurrence of serious adverse events was much the same between the two groups (p=0·35). Nausea was more common in participants in the tenofovir group than in the placebo group (p=0·002).
Interpretation
In this study, daily oral tenofovir reduced the risk of HIV infection in people who inject drugs. Pre-exposure prophylaxis with tenofovir can now be considered for use as part of an HIV prevention package for people who inject drugs.
Funding
US Centers for Disease Control and Prevention and the Bangkok Metropolitan Administration.
Introduction
The Joint UN Programme on HIV/AIDS reports that 2·5 million people contracted HIV in 2011.1 One in ten of these new HIV infections was probably caused by injecting drug use; in some countries in eastern Europe and central Asia, more than 80% of all HIV infections are related to drug use.2 In Thailand, HIV spread rapidly in people who inject drugs in the late 1980s3 and HIV prevalence has remained high in this group, ranging from 30% to 50%, through 2009.4 Safe and effective interventions to prevent HIV infection in this population are needed.
The use of antiretrovirals to prevent HIV infection is a promising new strategy to end the HIV/AIDS epidemic. Several lines of evidence suggest that pre-exposure prophylaxis with tenofovir disoproxil fumarate (tenofovir) can reduce HIV transmission in people who inject drugs. Findings from studies in macaque monkeys show that tenofovir can prevent or delay mucosal and parenteral infection with HIV-like viruses.5-7 Antiretrovirals are also used to reduce mother-to-child transmission8 and the risk that health-care workers will become infected after occupational HIV exposure.9 Additionally, tenofovir is an attractive candidate for use in injecting-drug users because it does not alter the pharmacokinetics or pharmacodynamics of methadone.10 In this context, we did the Bangkok Tenofovir Study, a phase 3, randomised, double-blind, placebo-controlled trial to establish whether pre-exposure prophylaxis with daily oral tenofovir would reduce the risk of HIV infection in people who inject drugs.
Since the study started in 2005, findings from other trials have shown that daily use of the combination antiretroviral tenofovir-emtricitabine can reduce HIV incidence by 44% (95% CI 15-63) in men who have sex with men,11 by 62% (22-83) in heterosexual men and women,12 and by 75% (55-87) in HIV-serodiscordant heterosexual couples,13 and that tenofovir alone can reduce transmission by 67% (44-81) in HIV-serodiscordant heterosexual couples.13 On the basis of these findings, the US Centers for Disease Control and Prevention (CDC) issued guidance on the use of pre-exposure prophylaxis to limit sexual HIV transmission in 2012.14, 15 To our knowledge, this is the first trial to assess HIV pre-exposure prophylaxis in people who inject drugs.
Methods
Participants and trial design
We recruited participants at 17 drug-treatment clinics in densely populated urban communities of Bangkok, Thailand. The clinics offer HIV counselling and testing, risk-reduction counselling, social services, primary medical care, methadone treatment, condoms, and bleach to clean injection equipment, all free of charge. Thailand's narcotics law prohibits the distribution of needles to inject illegal drugs, so needles are not provided in the clinics. However, sterile needles are available without a doctor's prescription at low cost (5-10 Baht; US$0·12-0·25) in pharmacies in Bangkok. Study staff coordinated with Corrections Officials to continue study activities in prisons and participants were able to receive study drug during periods of incarceration.
HIV-negative individuals aged 20-60 years who reported injecting drugs during the previous year were eligible for the study. We excluded people with hepatitis B virus surface antigen and women who were pregnant or breastfeeding. We asked women to use contraception or abstain from sex during the trial. We gave contraceptives (ie, oral, injectable, and condoms) and hepatitis B vaccine to participants.
Volunteers meeting all eligibility criteria could enrol after providing written informed consent (appendix). Participants were compensated for the travel and time required by the study. The amount was determined by reviewing compensation provided by other trials in Thailand and discussions with the community relations committee, comprised of at least one person who injected drugs from each of the 17 drug-treatment clinics, and the chosen amount was acceptable to local ethical review committees.
Ethical Review Committees of the Bangkok Metropolitan Administration (BMA) and the Thailand Ministry of Public Health and the CDC Institutional Review Board approved the study protocol and consent forms. An independent Data and Safety Monitoring Board did annual safety reviews and an interim efficacy analysis. The community relations committee met with investigators every 2 months to provide community input and guidance during protocol preparation and throughout the trial.16 Clinical research organisations assured compliance with good clinical practices.
Randomisation and masking
We randomly assigned participants in a one-to-one ratio to receive daily oral tenofovir 300 mg or placebo in blocks of four using a computer-generated randomisation sequence. The statistician who generated the sequence was not otherwise involved in the conduct of the trial, but was involved in the final analysis. When a participant completed the consent process, study staff assigned them the next sequential randomisation number. Tenofovir and placebo tablets were similar in shape, colour, and taste. Participants and study staff were masked to drug assignment. Data were locked on Jan 18, 2013, at which point PAM and MM were unmasked (other investigators were unmasked individually as needed to help with the analysis).
Procedures
At enrolment and monthly visits (every 28 days), participants were assessed for adverse events, individualised adherence and risk-reduction counselling was provided, oral fluid was tested for HIV antibodies (OraQuick Rapid HIV-1/2 Antibody Test; OraSure Technologies Inc, PA, USA), and women had urine pregnancy tests (OneStep urine test; ULTI Med Products, Ahrensburg, Germany). Participants chose daily directly observed therapy (DOT) or monthly visits without DOT and could switch at monthly visits. Adherence was assessed daily at DOT visits and monthly at non-DOT visits using a study drug diary and risk behaviour was assessed every 3 months with an audio computer-assisted self-interview. Staff contacted participants by telephone or did a home visit if participants missed their appointed visit. We obtained blood samples for safety assessment at enrolment, months 1, 2, and 3, and every 3 months thereafter. On Sept 15, 2011, we added 3 monthly enzyme-immunoassay (Genetic Systems HIV-1/HIV-2 Plus O EIA; Bio-Rad, Redmond, WA, USA) blood testing to improve detection of early HIV infection.17, 18 We tested blood samples obtained at the final follow-up visit for HIV with enzyme-immunoassay and nucleic-acid amplification (Aptima HIV-1 RNA Qualitative Assay; Gen-Probe Inc, San Diego, CA, USA; appendix).
Participants with reactive HIV tests discontinued study drug, plasma samples were obtained for antiretroviral resistance testing (TRUGENE; Siemens HealthCare Diagnostics Inc, Tarrytown, NY, USA), and infection was confirmed with enzyme-immunoassay and western blot analysis (Bio-Rad, Redmond, WA, USA). Newly infected individuals were referred for care according to national guidelines19 and received CD4 lymphocyte count20 and plasma HIV RNA level testing (Amplicor Monitor v1.5; Roche Molecular Systems (Branchburg, NJ, USA) every 4 months during the study. We tested stored blood for HIV (COBAS TaqMan, Roche Molecular Systems) to define the last HIV-negative and first HIV-positive specimens.
We measured plasma tenofovir concentrations using a validated ultra-high-performance liquid chromatography-tandem mass spectrometry method with a lower limit of quantitation of 0·31 ng/mL.21 Specimens were obtained from HIV-positive participants the day infection was detected and from HIV-negative participants at four of the 17 clinics at study exit. We could not collect specimens matched by time on study because of logistic constraints.
Statistical analysis
We used HIV-incidence results from the 1999-2003 AIDSVAX B/E HIV vaccine trial,22 which was done in the same clinics, to estimate sample size requirements and aimed to accrue at least 40 incident HIV infections, providing 80% power, assuming 67% tenofovir efficacy, to show 10% or higher efficacy with a one-sided α of 0·025.16, 23
The primary efficacy endpoint was HIV infection, analysed by modified intention-to-treat analysis, including all randomly allocated participants apart from those with HIV at enrolment. We estimated efficacy using the hazard ratio from Cox regression and Kaplan-Meier method to estimate the cumulative probability of HIV infection.
The data and safety monitoring board did an interim efficacy analysis when 24 HIV infections accrued. We used the Lan-DeMets alpha-spending function with O'Brien-Fleming boundaries for stopping rules in which the overall type-1 error rate of 2·5% (one-tail) could be spent in a flexible manner using EAST (version 5.3 software).23, 24
We did a secondary per-protocol adherence-defined analysis, restricted to DOT participants who took the study drug at least 71% of days (to approximate 5 days per week) with no more than 2 consecutive days off study drug (appendix). We also did an unmatched case-control analysis in participants receiving tenofovir to investigate the risk of HIV infection associated with quantifiable plasma tenofovir concentrations. HIV infection was rare, allowing us to use the odds ratio (OR) to approximate the relative risk to estimate efficacy.
To assess the effect of tenofovir prophylaxis on HIV disease progression, we compared plasma HIV RNA concentrations and CD4 lymphocyte counts by treatment group at the first visit after a participant was identified as being HIV-positive and each follow-up visit, and used time-to-event analysis to compare time to two consecutive plasma HIV RNA concentration tests of greater than 30 000 copies per mL and CD4 lymphocyte count of less than 500 cells per mL and less than 350 cells per mL by group.
All participants were included in the safety analysis. We coded adverse events using the International Classification of Diseases (tenth revision, Thai Modification)25 and grouped codes to summarise events. We compared rates of adverse events and graded laboratory results by group using a Poisson model with robust SE (appendix).
We used generalised estimating equations logistic regression and its multinomial extension to model the trend of participant reports of use of injecting drugs, needle sharing, and the number of sexual partners during follow-up, and to test for a between-group difference. Data were censored at the last HIV test. Unless otherwise specified, we used SAS (version 9.3) for statistical analyses. This trial is registered with ClinicalTrials.gov, number NCT00119106.
Role of the funding source
The study sponsor participated in study design, data collection, analysis, and interpretation of the results. The corresponding author had full access to all the data in the study and had final responsibility for the decision to submit for publication.
Results
Enrolment and baseline characteristics are described elsewhere.16 Briefly, from June 9, 2005, to July 22, 2010, we screened 4094 volunteers, randomly assigning 2413 to either tenofovir or placebo (figure 1). Baseline characteristics were much the same between groups (except for sexual intercourse with a casual partner in the past 12 weeks and men having sex with men in the past 12 weeks, which both seemed more common in the placebo group): the median age of participants was 31 years (mean 32·4 years, SD 8·4; range 20-59), most were men, and nearly half had primary school education or less (table 1). The study endpoint target was reached on Nov 30, 2011, exit visits were completed on June 8, 2012, and the database was locked on Jan 18, 2013.
We followed-up participants for 9665 person-years (mean 4·0 years, SD 2·1; maximum 6·9 years). There were no differences in follow-up time, withdrawal, or loss to follow-up between treatment groups (figure 1, appendix).
On the basis of participants' study drug diaries, participants took the study drug an average (mean) of 83·8% of days (SD 23·0, median 94·1%, IQR 79·2-98·7) and adherence did not differ by treatment group (p=0·16) or by time on study (p=0·22). Adherence was better in participants aged 40 years and older (median 98·2%, 93·5-99·5) than it was in younger participants (92·3%, 75·5-98·2; p<0·0001) and, controlling for age, better in women (95·6%, 81·1-98·9) than men (93·8%, 78·8-98·7%; p=0·04). Participants were on DOT an average of 86·9% of the time (SD 24·7); median adherence on DOT was 94·8% (IQR 80·3-98·8) and on non-DOT was 100% (91·6-100).
193 (8%) participants reported sharing their study drug: 109 (5%) shared their pills, 121 (5%) took pills from others, and 158 (82%) reported sharing only once. Total doses shared were less than 0·1% of all doses taken. Sharing did not differ by treatment group (p=0·10).
Tenofovir and placebo recipients reported similar rates of injecting and sharing needles and similar numbers of sexual partners during follow-up with no interactions between time and treatment group (data not shown). The number of participants reporting injecting drugs during the previous 3 months decreased from 1507 (63%) at enrolment, to 426 (23%) at month 12, and to 117 (18%) at month 72. Reports of sharing needles decreased from 435 (18%) at enrolment to 44 (2%) at month 12, and to eight (1%) at month 72. Sex with more than one partner decreased from 522 (22%) at enrolment to 43 (6%) at month 72 (p<0·0001 for all). Excluding data obtained at enrolment, 1018 (45%) participants reported injecting drugs during the study; 35 (70%) of those who became HIV-positive and 983 (45%) of those who remained HIV-negative during the trial.
The frequency of deaths, serious adverse events, grade 3 and 4 laboratory results, and increased creatinine concentrations were much the same between the two groups (table 2). During the trial, 107 participants died: 24 (22%) from a drug overdose, 13 (12%) from traffic accidents, and 12 (11%) from sepsis (appendix). The number of deaths was much the same between groups (p=0·34), as were the causes of death (appendix).
Reports of nausea or vomiting were higher in the tenofovir group than the placebo group (table 2); the between-group difference resolved by the second month of follow-up (appendix). Grade 1 or 2 increases of alanine aminotransferase concentrations were more common in the tenofovir group than in the placebo group (table 2). The median difference at monthly visits was 1-5 U/L and did not increase with time in the study. The number and severity of other adverse events reported were similar in participants in the two groups (appendix).
We confirmed HIV infection in 52 participants (17 [33%] in the tenofovir group, 35 [67%] in the placebo group) indicating a 51·8% reduction in HIV incidence (95% CI 15·3-73·7; p=0·01) in the tenofovir group compared with the placebo group in the intention-to-treat analysis. Excluding the two participants in the placebo group who were HIV-positive at enrolment, HIV incidence was 0·35 per 100 person-years in the tenofovir group and 0·68 per 100 person-years in the placebo group representing a 48·9% reduction in HIV incidence in the modified intention-to-treat analysis (9·6-72·2; p=0·01; table 3). The cumulative probability of HIV infection in the two groups separated consistently after 36 months (figure 2).
Two of the 50 participants with incident HIV infection were excluded from the per-protocol adherence-defined analysis because their time on study before HIV infection did not reach the time required (ie, 28 days before the last negative HIV test result; appendix). The tenofovir efficacy estimate based on the 48 eligible participants was 45·7% (3·1-70·6; p=0·04); 17 met the adherent criteria (ie, took the study drug for 71% or more of days and did not miss more than 2 consecutive days of study drug)-five in the tenofovir group and 12 in the placebo group, yielding an efficacy estimate of 55·9% (95% CI, -18·8 to 86·0; p=0·11). Drug concentration testing showed that two of the five participants in the tenofovir group did not have detectable tenofovir in their plasma. We did a post-hoc analysis, removing these two participants, and recorded a modified tenofovir efficacy estimate of 73·5% (16·6-94·0; p=0·03).
The trial was not powered to assess efficacy by subgroup, but tenofovir showed statistically significant efficacy in women (p=0·03) and participants aged 40 years and older (p=0·01; table 3).
We obtained plasma samples from 46 participants with incident HIV infections the day infection was detected, and from 282 HIV-negative participants to test for the presence of tenofovir. Tenofovir was detected in one (1%) of 177 participants in the placebo group and 100 (66%) of 151 participants in the tenofovir group. In the case-control analysis in participants assigned to tenofovir, we detected tenofovir in the plasma of five (39%) of 13 HIV-positive participants and 93 (67%) of 138 HIV-negative participants (appendix). Compared with participants without detectable tenofovir, the odds of HIV infection was three times lower (OR 0·30; 95% CI 0·09-0·98; p=0·04) in participants with detectable concentrations of tenofovir, which corresponds to a reduction in risk of 70% (95% CI 2·3-90·6; p=0·04).
We were able to amplify viral RNA in specimens from 49 of the 52 HIV-positive participants for molecular genotyping; 43 (88%) were consistent with CRF01_AE (29 in the placebo group, 14 in the tenofovir group), five (10%) with subtype B' (four in placebo, one in tenofovir), and one (2%) with CRF01_AE and subtype B' recombinant (in placebo). We detected no tenofovir-associated resistance mutations (ie, K65R and K70E).
CD4 lymphocyte counts were much the same in HIV-positive participants in the two groups. Plasma HIV RNA concentrations were lower in the tenofovir group at the visit when HIV infection was detected and study drug stopped (p=0·01), but there was no difference between the groups at month 4 through to month 24 after infection or in longitudinal analysis (p=0·10) or in time to plasma HIV RNA concentrations being higher than 30 000 copies per mL (p=0·22).
Discussion
Once-daily oral tenofovir decreased the risk of HIV infection by 48·9% in injecting drug users when provided with other HIV prevention services at drug-treatment clinics in Bangkok. Findings from other pre-exposure prophylaxis trials showed that adherence had an important effect on efficacy.11, 12 In this study, efficacy increased from 46% to 56% in the per-protocol analysis based on observed adherence and to 74% when limited to participants with detectable tenofovir concentrations. Although the trial was not powered to assess efficacy in subgroups, we saw higher efficacy in women (79%) and in participants aged 40 years or older (89%)-two subgroups with high levels of adherence. The modified intention-to-treat efficacy result did not rule out tenofovir efficacy at less than 10% as specified in the protocol.
We do not know why HIV incidence in the two groups did not differ consistently until after 36 months (figure 2). Low levels of adherence or low risk behaviour during the first 36 months could have masked the effect of tenofovir, but adherence did not change by time on study and risk behaviour decreased during follow-up. The low HIV incidence and slow accrual of infections might be why no between-group difference was seen before 36 months. At 36 months, there were 27 infections and, assuming 49% efficacy, the distribution should have been nine with tenofovir and 18 with placebo. However, there were 13 with tenofovir and 14 with placebo, a difference of only four events.
As has been reported in other trials,11, 12 participants in the tenofovir group reported more nausea and vomiting in the first couple of months of follow-up than did those in the placebo group. When used for treatment of HIV, tenofovir is associated with small decreases in renal function.26, 27 We did not find higher rates of increased creatinine or renal disease in participants randomly allocated to tenofovir.
Other pre-exposure prophylaxis trials have described antiretroviral-resistance mutations in HIV-positive participants, especially in those with unrecognised HIV infection at enrolment.11-13 We did not detect tenofovir resistance in HIV-positive participants in this study. The two participants with unrecognised HIV infection at enrolment were randomly allocated to placebo, limiting the possibility that acquired resistance would occur.
Participant reports of injecting drugs and sharing needles decreased during follow-up, consistent with previous trials in people who inject drugs in Bangkok.28, 29 The HIV incidence in placebo recipients in our study was 0·68 per 100 person-years. This incidence compares with an incidence of 5·8 per 100 person-years in a preparatory trial done in the same clinics in 1995-99 and of 3·4 per 100 person-years during the 1999-2003 AIDSVAX B/E HIV vaccine trial.22, 28 This decrease over time is probably due to many factors, including monthly HIV risk-reduction counselling, decreased needle sharing, and monthly HIV testing speeding up the diagnosis of HIV and limiting the number of people with unrecognised acute HIV infection able to transmit HIV to others.
Our study had several limitations. Participants could have under-reported stigmatised and illegal behaviours such as injecting drugs.30 However, the illegality and stigma attached to these activities did not change during the trial, meaning that rates of under-reporting should have remained constant. The study aimed to establish whether tenofovir would reduce parenteral HIV transmission, but participants might have become infected sexually. Previous studies in people who inject drugs in the same clinics in Bangkok have shown strong associations between injecting drugs and HIV infection, but no association between sexual activity and HIV infection.28, 29 In this study, although reports of injecting drug use decreased, 1018 (45%) participants reported injecting drugs during follow-up, including 35 (70%) of those who contracted HIV during the course of the study. Furthermore, similar to the previous studies in the drug-treatment clinics, drug overdose, traffic accidents, and sepsis were the most common causes of death, and participants were frequently incarcerated. Together these data suggest that participants were actively injecting drugs and that parenteral HIV transmission, not sex, was the primary route of HIV infection. Additional risk behaviour analyses are underway. The study was done in drug-treatment clinics offering a package of HIV prevention interventions and DOT; tenofovir effectiveness might differ in other settings.
Findings from three randomised, placebo-controlled trials have shown that a daily dose of tenofovir or tenofovir-emtricitabine can reduce sexual HIV transmission.11-13 Findings from two other studies showed that tenofovir and tenofovir-emtricitabine did not reduce sexual HIV transmission.31, 32 Adherence seems to be the key factor determining efficacy.33 These trials draw attention to the need for methods to help people using pre-exposure prophylaxis achieve effective levels of adherence.
To our knowledge, this study is the first to show that daily oral pre-exposure prophylaxis with tenofovir, when used in combination with other HIV prevention strategies, reduces the risk of HIV infection among people who inject drugs (panel). The US Food and Drug Administration has approved the use of tenofovir-emtricitabine to prevent sexual acquisition of HIV in high-risk individuals.34 On the basis of the results of this study, regulatory and public health authorities can now consider whether pre-exposure prophylaxis with tenofovir should be part of an HIV prevention package to reduce the risk of HIV infection in people who inject drugs.
Panel
Research in context
Systematic review
We searched PubMed for phase 1, 2, and 3 randomised clinical trials in human beings assessing tenofovir for the treatment of HIV infection and animal trials using tenofovir to prevent HIV infection. We used the search terms "HIV", "tenofovir", "treatment", "prevention", and "clinical trials", restricting our search to studies published in English through December, 2004. The study was launched in 2005 and, at the time, no phase 3 clinical trials using tenofovir in human beings for HIV pre-exposure prophylaxis had published results.
Interpretation
To our knowledge, this is the first study to show that daily oral pre-exposure prophylaxis with tenofovir, when used in combination with other HIV prevention strategies, reduces the risk of HIV infection in people who inject drugs. Much like findings from other pre-exposure prophylaxis trials, our findings showed that adherence had an important effect on efficacy. On the basis of these findings regulatory and public health authorities can now consider whether pre-exposure prophylaxis with tenofovir should be part of an HIV prevention package to reduce the risk of HIV infection in people who inject drugs.
| |
| |
| |
|
|
|