| |
Development and Pharmacokinetics of a 90-Day Intravaginal Ring for the Sustained Co-Delivery of the Microbicide Tenofovir and Contraceptive Levonorgestrel ........presented Nov 10 2013 at Annual Meeting for the American Ass. Of Pharmaceutical Scientists
|
| |
| |
Slide presentation below
They designed the ring using reservoir-type polyurethane segments that were individually optimized to deliver each drug at the desired dosage-a high flux of tenofovir and low flux of levonorgestrel. The researchers performed in-vitro release testing, and 3-month pharmacokinetic (PK) studies in rabbits and sheep were done in comparison against the tenofovir gel.
The PK studies found that local levels of tenofovir in the target tissue delivered from the IVR are similar or higher than the levels following gel application. In addition, release of the contraceptive agent was consistent with previous levels tested to be efficacious in women.
This multipurpose prevention technology (MPT) can remain in the vagina for up to 90 days and releases the contraceptive levonorgestrel (LNG) and tenofovir (TFV), an antiretroviral that inhibits HIV and HSV replication in susceptible cells.
The CONRAD product development team, in collaboration with Dr. Patrick Kiser at Northwestern University, performed in vitro release testing and 3-month pharmacokinetic (PK) studies of the ring in rabbits and sheep, and compared drug levels to those seen with use of tenofovir gel. The PK studies found that levels of tenofovir in the target tissue delivered from the ring are similar or higher than those obtained after TFV 1% gel application, a product that has proven to be effective in preventing HIV and HSV infections in women. In addition, release of the contraceptive agent was also consistent with previous levels tested to be efficacious in women. Stability studies will continue and lead to Phase I clinical trials in women in 2014, which will test the combination ring, as well as a tenofovir-only ring.
Tenofovir is the first microbicide proven to be efficacious in humans, with the CAPRISA 004 clinical trial showing that women using the gel before and after sex reduced their risk of HIV infection by 39-54%. CAPRISA 004 also showed the gel to be 51% effective in reducing the transmission of HSV-2, making this combination ring potentially triple protective.
"The TFV/LNG ring is the first device to be tested in women that has the potential to offer contraception as well as HIV and herpes prevention," said Dr. Clark. "And so far, tenofovir is the only microbicide that has been proven to be effective in reducing HIV infections when used topically. It's important to develop a variety of delivery mechanisms for tenofovir in order to serve different women's needs."
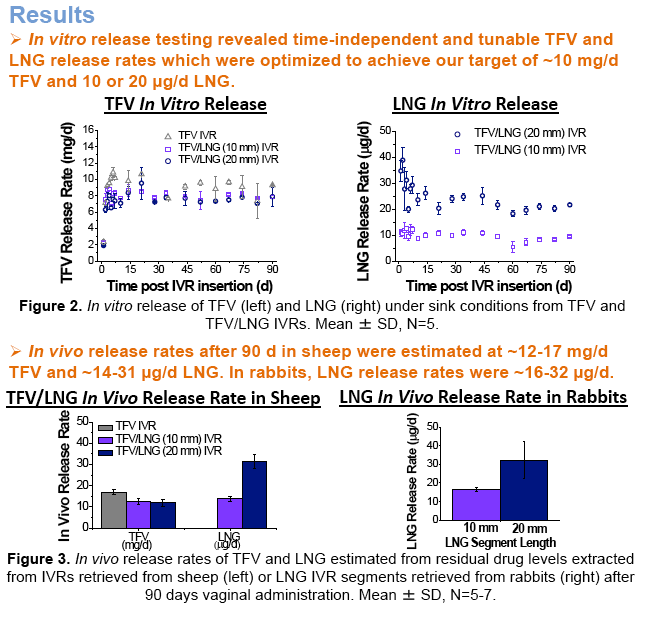
The researchers found that levels of the two drugs were time-independent and tunable, and were optimized to a target of 10mg/day tenofovir and 10 or 20μg/day levonorgestrel. Local levels of tenofovir in the target tissue were similar or higher than the levels following application of a 1% tenofovir gel. The release of levonorgestrel was consistent with levels known to be efficacious in women.
"We developed a unique intravaginal ring technology that met our product objectives of delivering a high flux of a hydrophilic antiretroviral (tenofovir) with a low flux of a hydrophobic contraceptive (levonorgestrel) in a controlled, time-independent manner for 90 days," Kiser and colleagues conclude.
Several authors are employees of CONRAD, which is contributing to the development of the intravaginal ring.
ABSTRACT
Development and Pharmacokinetics of a 90-Day Intravaginal Ring for the Sustained Co-
Delivery of the Microbicide Tenofovir and Contraceptive Levonorgestrel
P. F. Kiser1, M. R. Clark2, J. T. Clark1, T. J. Johnson1, N. B. Shelke1, J. S. Nebeker1, G. F. Doncel2, D. R. Friend2, T.McCormick21University of Utah,2CONRAD, Eastern Virginia Medical School
Purpose
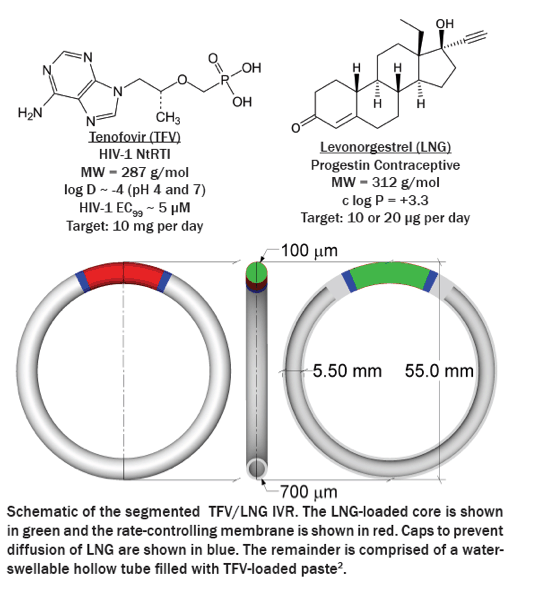
A majority of the world's unintended pregnancies occur within resource-poor regions where the HIV/AIDS pandemic is most prevalent. Tenofovir (TFV) is the only microbicide antiretroviral shown clinically to be effective at reducing the incidence of HIV acquisition when dosed pericoitally from a vaginal gel. There remains a need to 1) develop a topical drug delivery system capable of simultaneously protecting women against both HIV infection and unintended pregnancy; and 2) improve TFV delivery by providing long-lasting and coitally-independent drug levels. Intravaginal rings (IVRs) offer both capabilities; however conventional IVR technologies are unfit for co-delivering these polarity-divergent actives at target release rates that differ 500-1000 fold. Here we report the development and pharmacokinetics (PK) of a segmented, reservoir-type polyurethane (PU) IVR capable of simultaneous zero-order co-delivery of TFV and the progestin contraceptivelevonorgestrel (LNG) for 90 days.
Methods
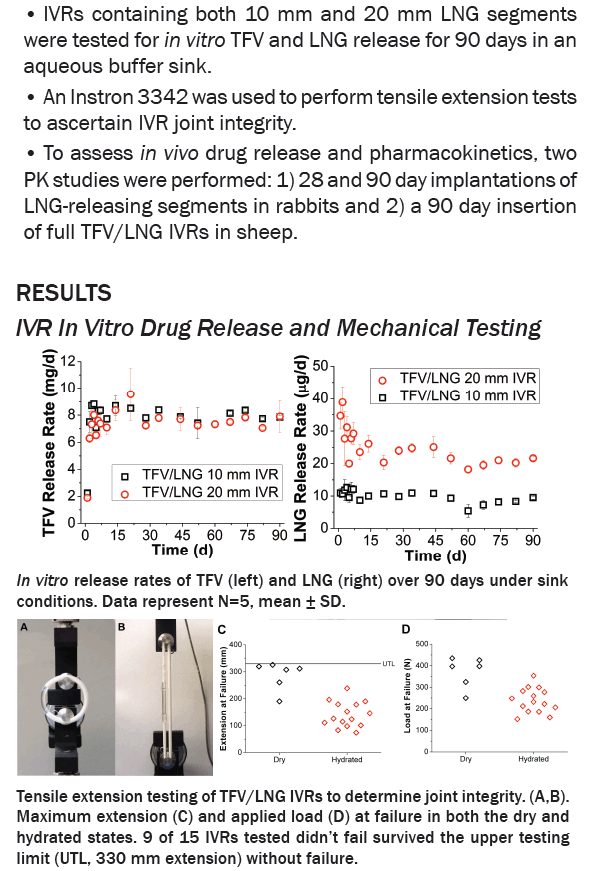
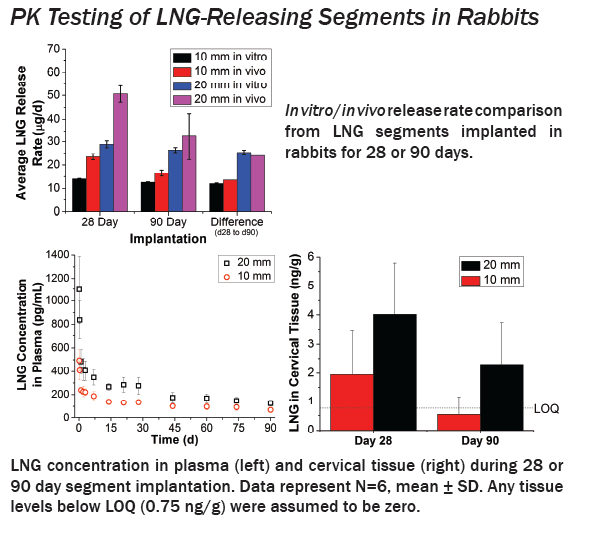
IVRs were designed using reservoir-type polyurethane segments which were individually optimized to deliver each drug. TFV segments comprised water-swellable PU tubing filled with a high density, semisolid TFV paste. LNG segments comprised a LNG-loaded non-swellable PU core co-extruded with a rate controlling PU membrane. LNG segments were cut to either 1 or 2 cm lengths to obtain 2-fold different release rates. In vitro release testing (IVRT) and 3-month PK studies in rabbits and sheep evaluated device performance.
Results
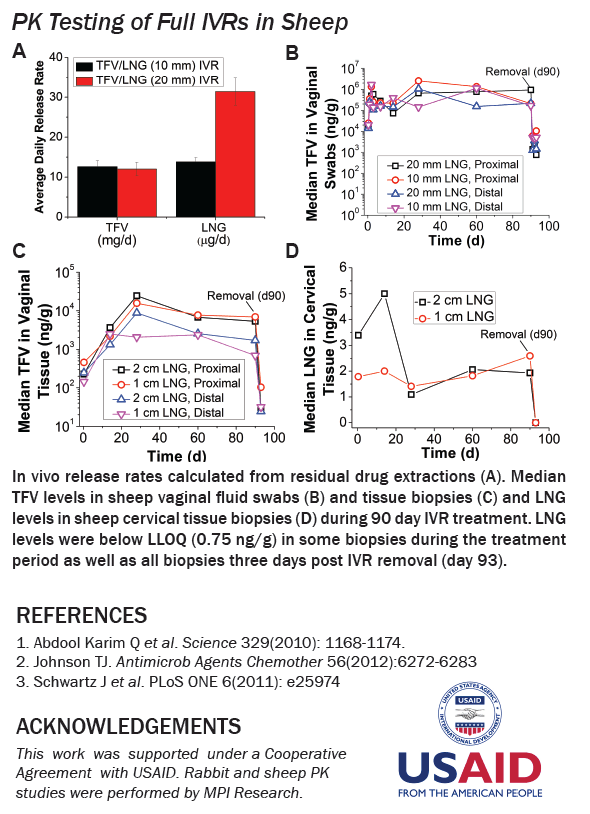
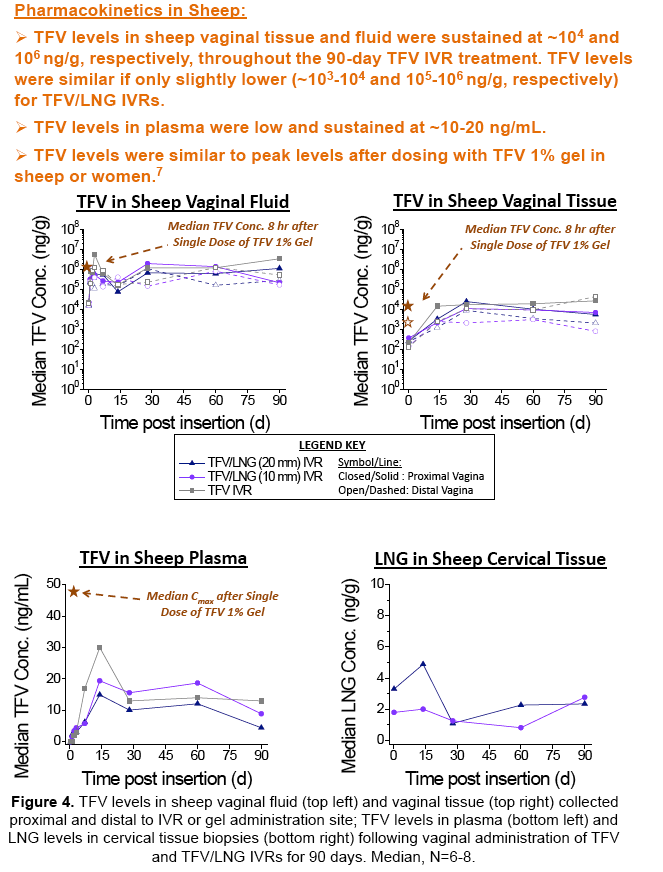
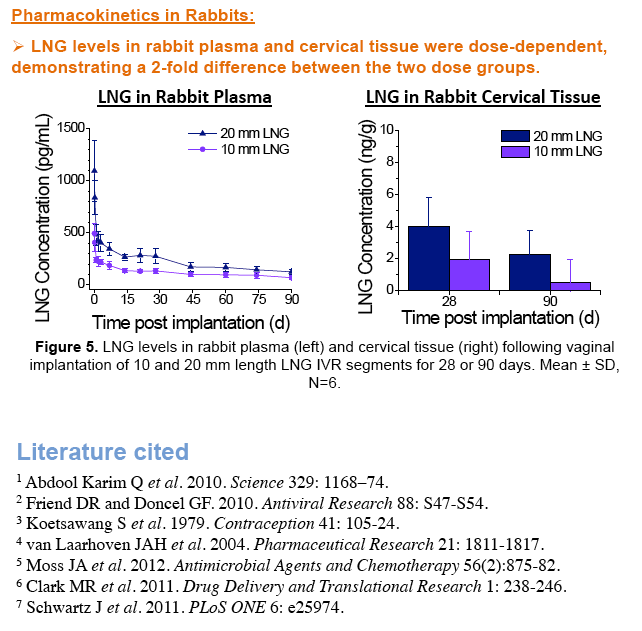
IVRT revealed time-independent and tunable TFV and LNG release rates which were optimized to achieve our target 10 mg/d TFV and 10 or 20 μg/d LNG. In sheep, TFV and LNG release rates were estimated at ~12-17 mg/d and ~14-31 μg/d, respectively. TFV vaginal tissue and fluid levels were ~104 and 106 ng/g, respectively, similar to maximal levels reported after clinical dosing of TFV 1% gel. In rabbits, LNG PK demonstrated 2-fold differences in plasma and cervical tissue concentrations between the two dose groups, as predicted by IVRT.
Conclusion
We developed a unique IVR technology that met our product objectives of delivering a high flux of a hydrophilic antiretroviral (TFV) with a low flux of a hydrophobic contraceptive (LNG) in a controlled, time-independent manner for 90 days.
--------------------------------------------------
SLIDE PRESENTATION
Development and Pharmacokinetics of a 90-Day Intravaginal Ring for the Sustained Co-Delivery of the Microbicide Tenofovir and Contraceptive Levonorgestrel
Reported by Jules Levin
2013 Annual meeting of the American Association of Pharmaceutical Scientists, held from Nov. 10-14 in San Antonio, Texas.
Meredith R. Clark1, Justin T. Clark2, Todd J. Johnson2, Namdev B.Shelke2, Joel S. Nebeker2, Gustavo F. Doncel1, David R. Friend1, Patrick F. Kiser2
1 CONRAD, Eastern Virginia Medical School, Arlington, Virginia, USA, 2Department of Bioengineering, University of Utah, Salt Lake City, Utah, USA



Development of a new intravaginal ring technology for the extended delivery of the microbicide tenofovir with and without the contraceptive levonorgestrel
Reported by Jules Levin
Presented at AIDS 2012 - Washington, D.C., USA
Todd Johnson1, Meredith Clark2, Justin Clark1, Namdev Shelke1, Gustavo Doncel2, David Friend2, Patrick Kiser1 1Department of Bioengineering, University of Utah, USA 2CONRAD, Eastern Virginia Medical School, Virginia, USA Corresponding author: mclark@conrad.org


|
|
| |
| |
|
|
|