 |
 |
 |
| |
Long-term virological outcome in HIV-infected children on ART in the UK/Ireland - Higher Failure Rate With Nevirapine Than Efavirenz or PIs in Children in UK and Ireland
|
| |
| |
7th IAS Conference on HIV Pathogenesis, Treatment and Prevention, June 30-July 3, 2013, Kuala Lumpur
Mark Mascolini
Compared with efavirenz- and boosted protease inhibitor (PI)-based regimens, nevirapine combinations resulted in higher virologic failure rates in a study of almost 1000 children starting antiretroviral therapy (ART) in the United Kingdom and Ireland [1].
Previous trials and cohort studies yielded conflicting data on antiretroviral regimen efficacy in children. Whether nonnucleoside (NNRTI) and PI regimens are equivalent and whether nevirapine regimens are equivalent to efavirenz combinations remained uncertain. To address these questions, Collaborative HIV Paediatric Study (CHIPS) researchers in the UK and Ireland conducted this analysis of children starting their first antiretroviral regimen and (1) reaching a viral load below 400 copies within 12 months and (2) experiencing virologic failure, defined as a confirmed rebound above 400 copies after suppression, an unconfirmed rebound followed by treatment modification, or a viral load above 400 copies after 12 months without previous suppression.
The analysis included children under 18 years old and naive to antiretrovirals when they started a three- or four-drug regimen. The investigators excluded children starting a PI not boosted by ritonavir or a triple-nucleoside (NRTI) combination. They based analyses on an intention-to-continue treatment model, ignoring antiretroviral changes and interruptions.
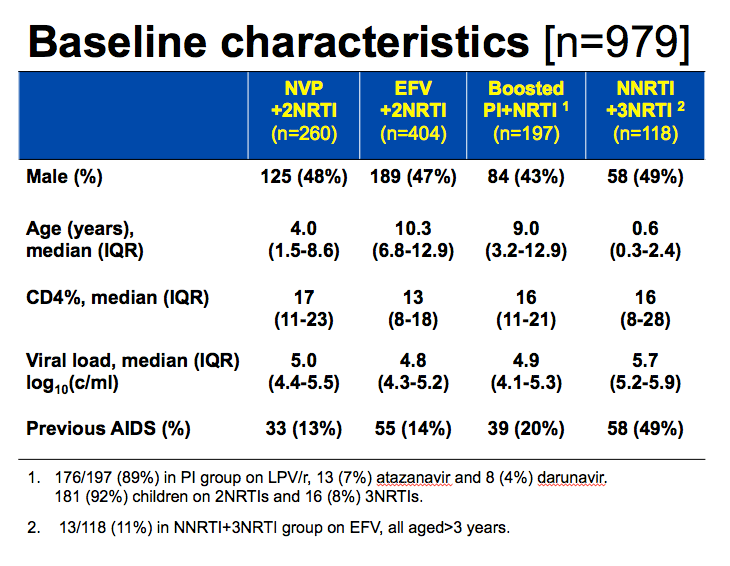
The study group consisted of 260 children starting nevirapine plus two NRTIs, 404 starting efavirenz plus two NRTIs, 197 started a boosted PI, and 118 starting an NNRTI plus three NRTIs. Median age in those four groups stood at 4.0, 10.3, 9.0, and 0.6 years, and slightly more than half of the children in all groups were girls. Principal NRTI backbones were abacavir plus 3TC and zidovudine plus 3TC.
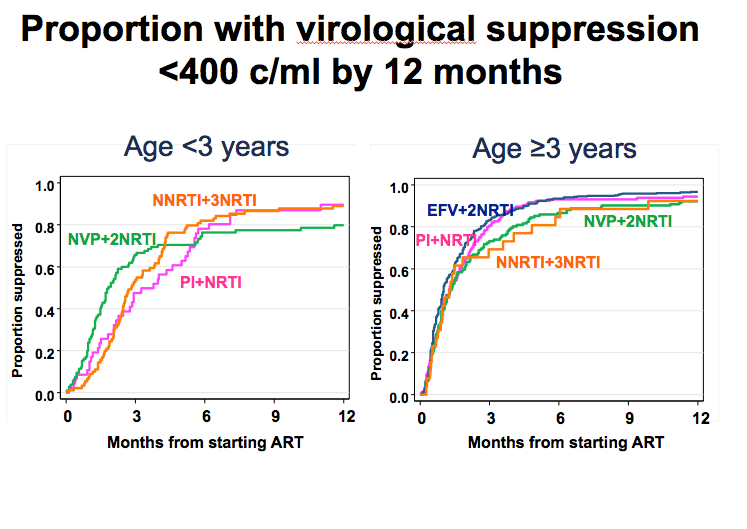
An estimated 92% of children reached a viral load below 400 copies within the first 12 months of therapy (95% confidence interval [CI] 91% to 94%). Compared with nevirapine plus two NRTIs, efavirenz plus two NRTIs was associated with an 18% better chance of virologic suppression within 12 months, but this difference stopped short of statistical significance (adjusted rate ratio [aRR] 1.18, 95% CI 0.97 to 1.44). There was no evidence that boosted PI regimens or an NNRTI plus three NRTIs yielded a better or worse 12-month response than nevirapine plus two NRTIs. These trends held true when the CHIPS team divided children into those younger than 3 years old and those 3 or older.
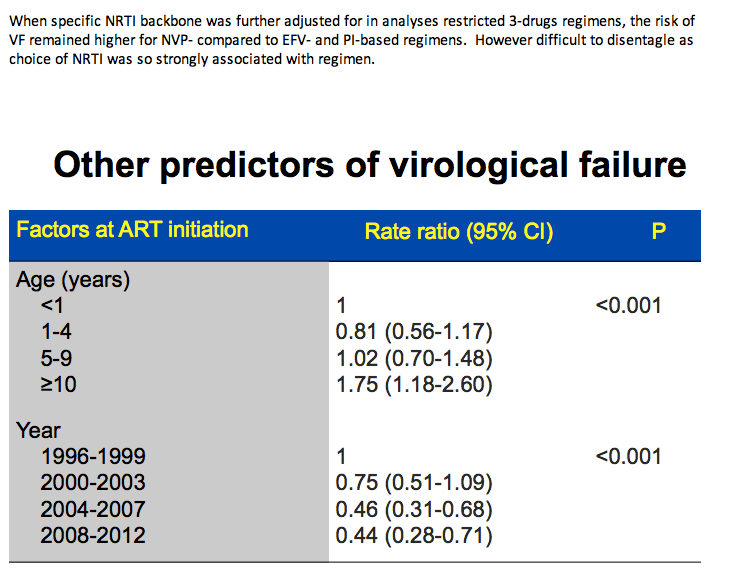
Older age, a lower pretreatment viral load, and starting ART in later calendar years were all associated with significantly better chances of virologic suppression in 12 months. Every 10-fold higher pretreatment viral load cut chances of virologic response 12% (aRR 0.88, 95% CI 0.82 to 0.95, P = 0.001). Compared with children younger than 1 year old, those 1 to 4, 5 to 9, and 10 or older had 31%, 60%, and 68% better chances of 12-month virologic suppression. Compared with children starting ART in 1996-1999, those starting in 2000-2003, 2004-2007, and 2008-2012 had 21%, 46%, and 68% better chances of suppression.
Of 979 children analyzed, 324 (33%) experienced virologic failure, including 256 with a confirmed rebound above 400 copies a median of 20.1 months after first reaching a sub-400 load. Compared with children starting nevirapine plus two NRTIs, the three other treatment groups had significantly lower chances of virologic failure (P < 0.001):
-- Efavirenz plus 2 NRTIs: aRR 0.54, 95% CI 0.40 to 0.73
-- Boosted PI plus 2 NRTIs: aRR 0.66, 95% CI 0.46 to 0.93
-- NNRTI plus 3 NRTIs: aRR 0.59, 95% CI 0.39 to 0.90
These associations held true for children 3 years old or older but not for children younger than 3. With further statistical adjustment for NRTI backbone, risk of virologic failure remained higher with nevirapine and two NRTIs than with efavirenz or a boosted PI plus other NRTIs.
Compared with children under 1 year old, those 10 or older had a 75% higher risk of virologic failure (aRR 1.75, 95% CI 1.18 to 2.60, P < 0.001). Compared with children starting ART in 1996-1999, those starting in 2004-2007 had a 54% lower risk of failure and those starting in 2008-2012 had a 56% lower risk.
Time to discontinuation for toxicity or adverse events was faster with nevirapine regimens than with efavirenz regimens. The researchers suggested three other reasons for the higher failure rate with nevirapine than efavirenz: (1) choice of NRTI backbone, (2) nevirapine underdosing, and (3) nevirapine dose-escalation strategy.
The CHIPS team concluded that in these children three-drug efavirenz regimens and four-drug NNRTI regimens "appeared at least as effective as three-drug PI-based regimens in maintaining virological suppression."
Reference
1. Duong T, Judd A, Doerholt K, et al. Long-term virological outcome in HIV-infected children on ART in the UK/Ireland. 7th IAS Conference on HIV Pathogenesis, Treatment and Prevention, June 30-July 3, 2013, Kuala Lumpur. Abstract MOAB0105.

Choice of NRTi backbone so strongly associated with regimen difficult to disentagle. NVP under dosing reported / documented in earlier calendar years.
***The difference between EFV+2NRTIs and NVP+2NRTIs regimens appeared greater during the first 2 years of starting ART (RR 0.46, 0.32-0.66) compared to after (0.78, 0.51-1.19, p for interaction=0.05).
(i) type of regimen was compared with specific NRTI back bone further adjusted, based on children on 3-drugs regimens only (ii) the limit of >1000 copies/ml (instead of >400 copies/ml) was used for viral load rebound in the definition of virological failure (iii) children who had ever been on once-daily NVP after the lead-in dosing period were excluded and (iv) in the analysis of virological failure, follow-up was censored at treatment interruption with viral load <400 copies/ml in order to exclude viral load rebound occurring while off-treatment.




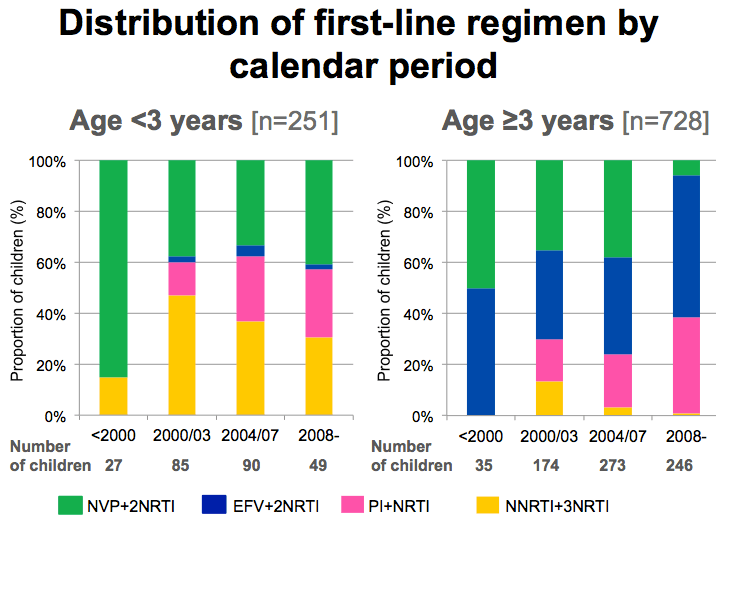
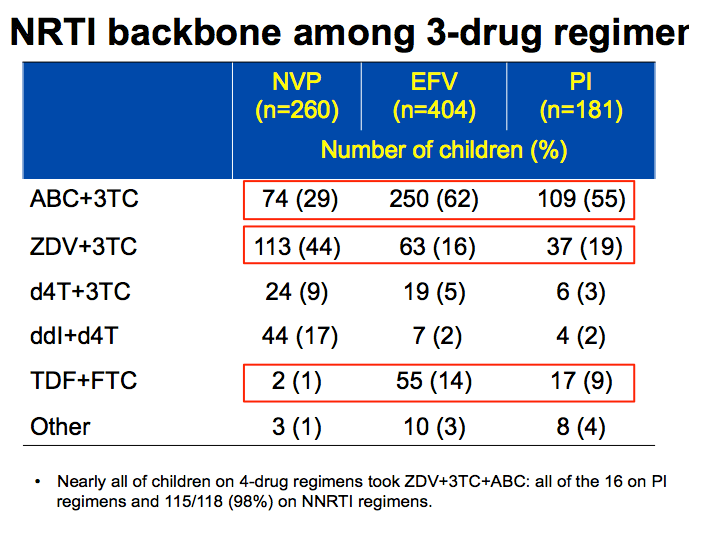
Important to look at backbone NRTI as this varies significantly with regimen.
-More potent backbones of ABC+3TC and TDF+FTC were more frequently used in EFV based regimens, accounting for over 75% of children, and 64% in those on PI as compared to 30% in NVP.
-Backbones with more risk of toxicity inc. ZDV+3TC, ddi+d4T were more frequently used with NVP and less with other regimens.
-Of note, 4 drug cocktail more likely be ABC based.
-children on EFV+2NRTIs and PI-based regimens were more likely to receive lamivudine+abacavir (29%, 62% and 55%, respectively) and tenofovir+emtricitabine backbones (1%, 14% and 9%, respectively), and less likely to receive zidovudine+lamivudine (44%, 16% and 19%, respectively)
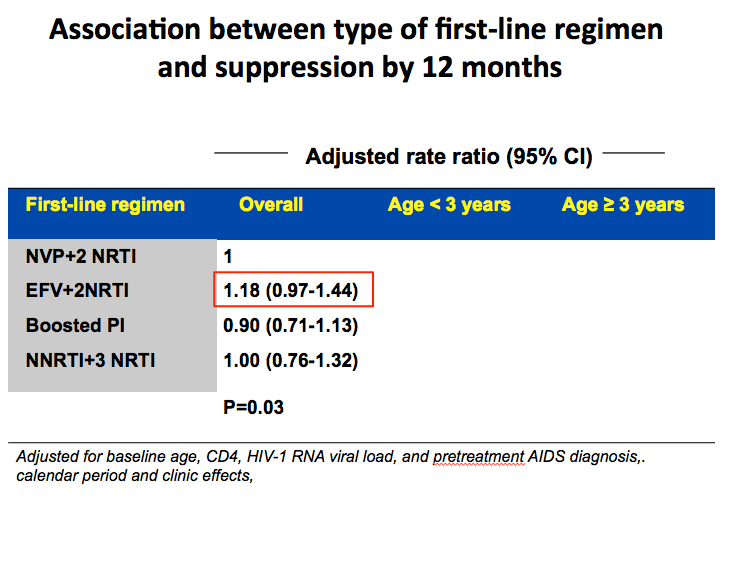
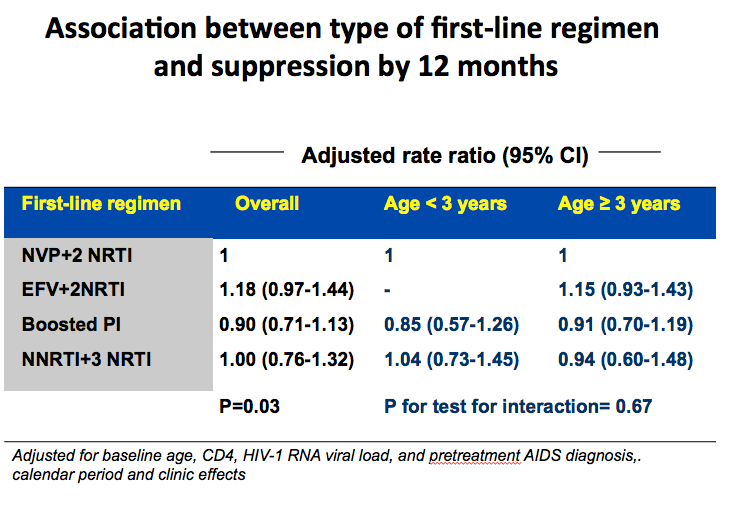
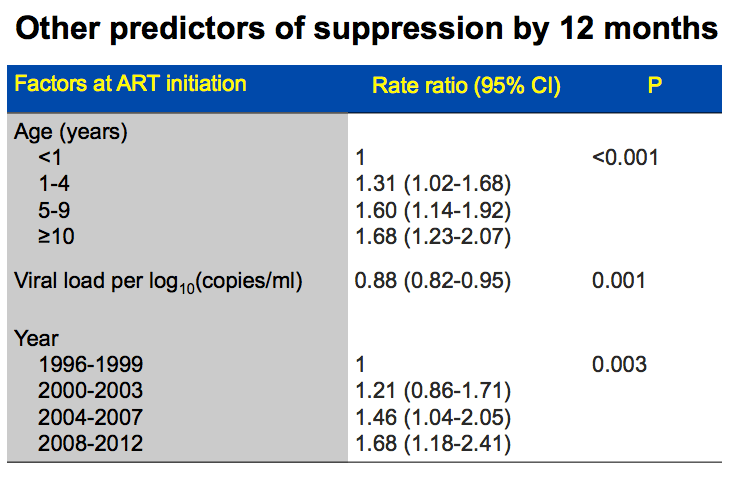
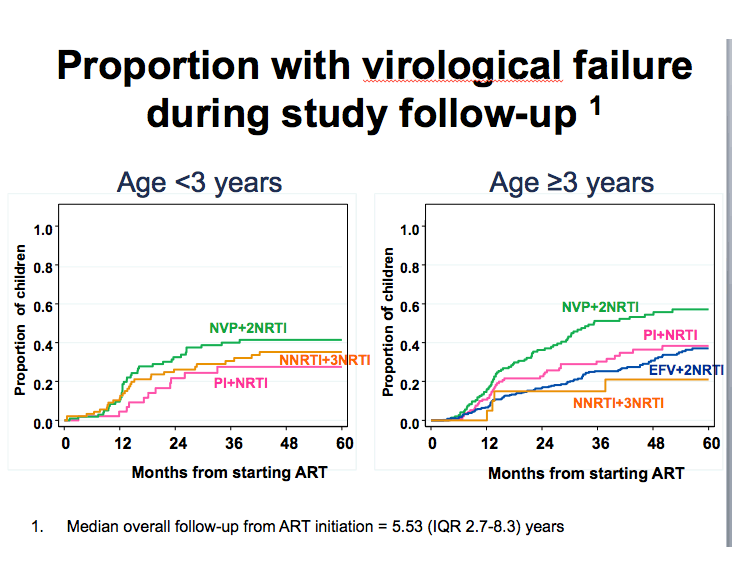


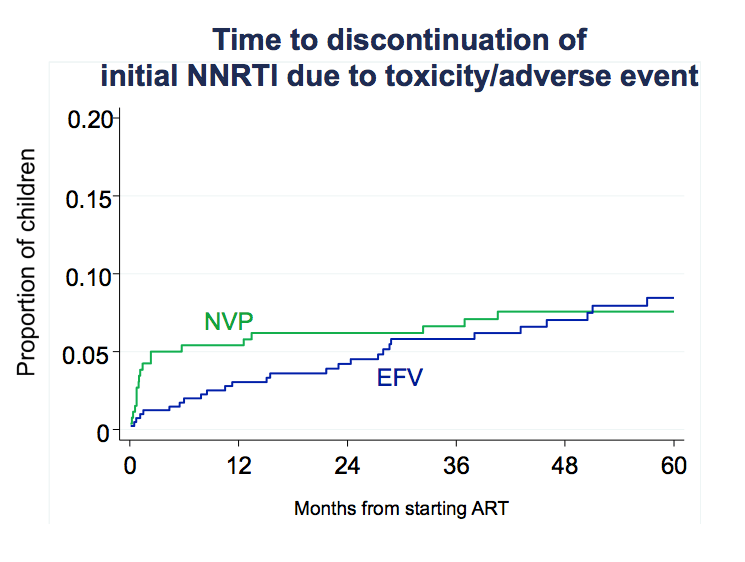
We conducted various analyses to explore reasons for the difference in response between the NVP vs EFV arms. One was time to treatment discontinuation for toxicity. This graph shows that NVP higher risk in first months of treatment for discontinuation, while EFV had steady increase and in long term converged and had similar overall rate. This may contribute to why NVP had more VF earlier on on therapy. Interestingly risk of VF in NVP was greater in the first 2 years of ART as compared to after.
|
| |
|
 |
 |
|
|