 |
 |
 |
| |
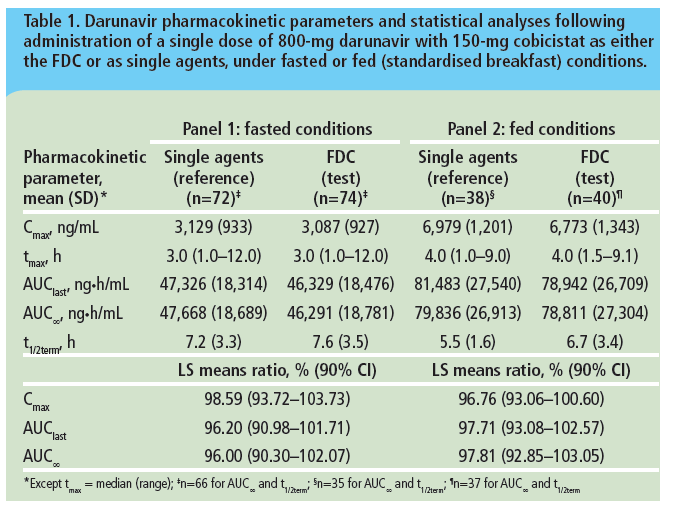
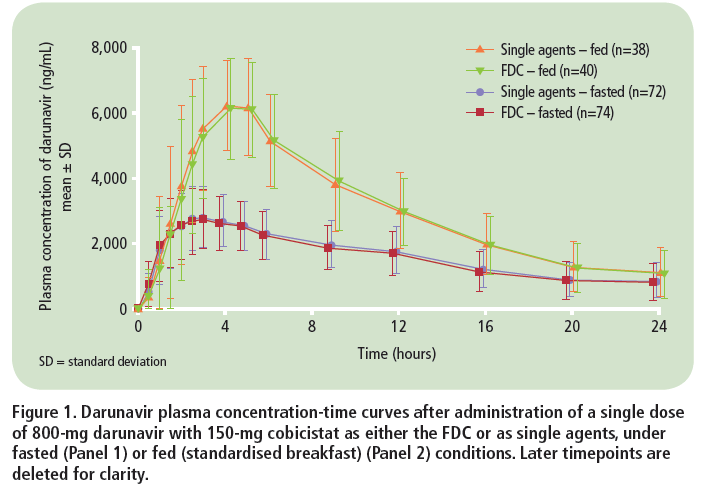
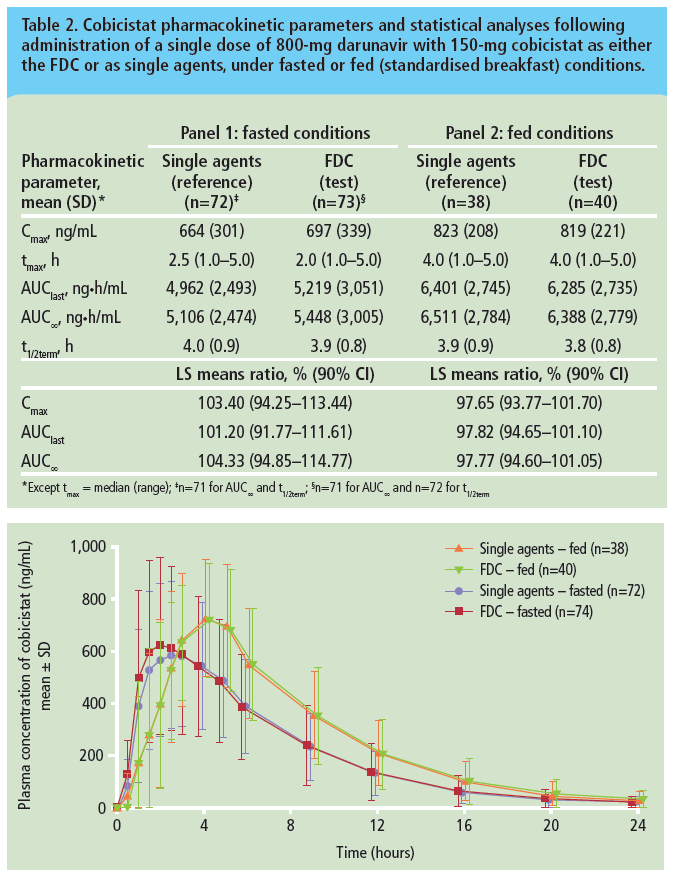
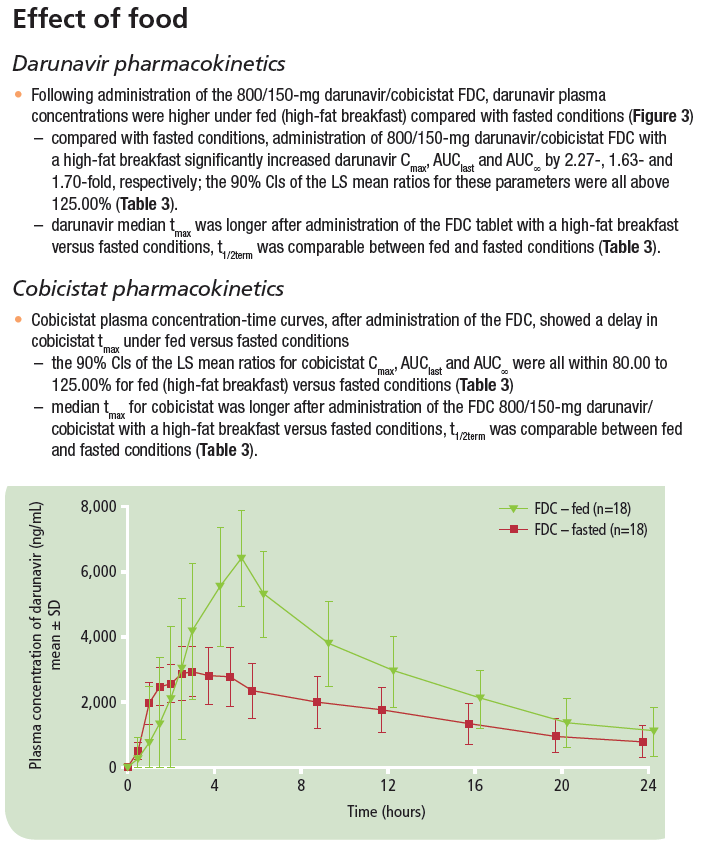
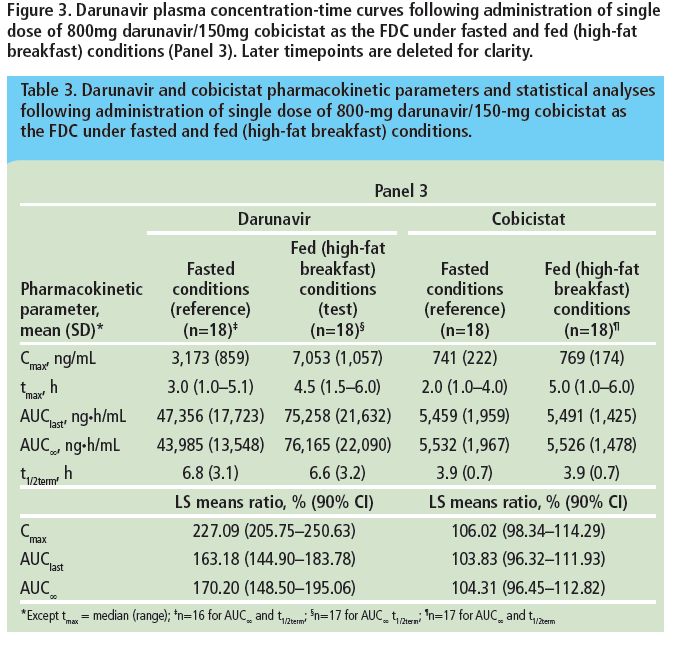
Bioequivalence of darunavir (800mg) co-administered with cobicistat (150mg) as either a fixed-dose combination tablet or as single agents under fasted and fed conditions in healthy volunteers
|
| |
| |
Reported by Jules Levin
IAS 2013 Kuala Lumpur, Malaysia June 30-July 3
Thomas N Kakuda,1 Tom Van De Casteele,2 Romana Petrovic,2 Magda Opsomer,2 Frank Tomaka,1 Joseph Mrus,3 Richard MW Hoetelmans2
1Janssen Research & Development LLC, Titusville, NJ, USA; 2Janssen Infectious Diseases BVBA, Beerse, Belgium; 3Janssen Global Services, Titusville, NJ, USA






|
| |
|
 |
 |
|
|