 |
 |
 |
| |
Pharmacokinetic (PK) interactions between Boceprevir (BOC) and Atazanavir/r (ATV/r) or Raltegravir (RAL) in HIV/HCV coinfected patients (pts)
|
| |
| |
Reported by Jules Levin
Clinical Pharmacology Workshop April 22-24 2013 Amsterdam
You can read comments on this study in Courtney Fletcher's NATAP Report from this workshop, scroll down to section on Hepatitis C Coinfection:
14th Intrntnl Wrkshp Clinical Pharm HIV Therapy Clinical Pharmacology at the 14th Workshop on Clinical Pharmacology of HIV Therapy and HCV - Courtney V. Fletcher, Pharm.D. Dean and Professor College of Pharmacy University of Nebraska Medical Center - (05/20/13)
PROGRAM ABSTRACT-
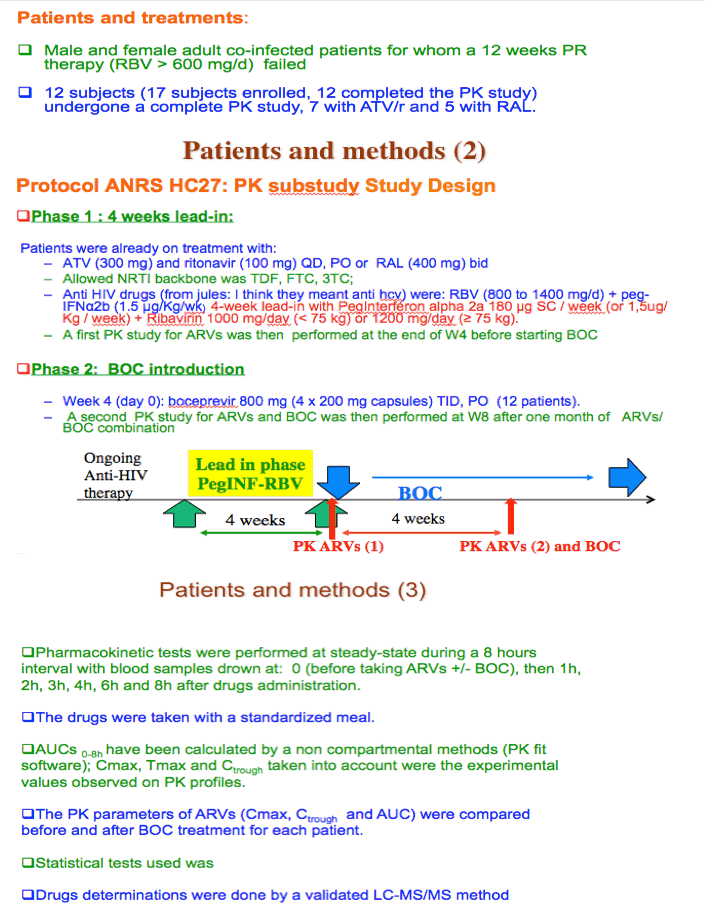
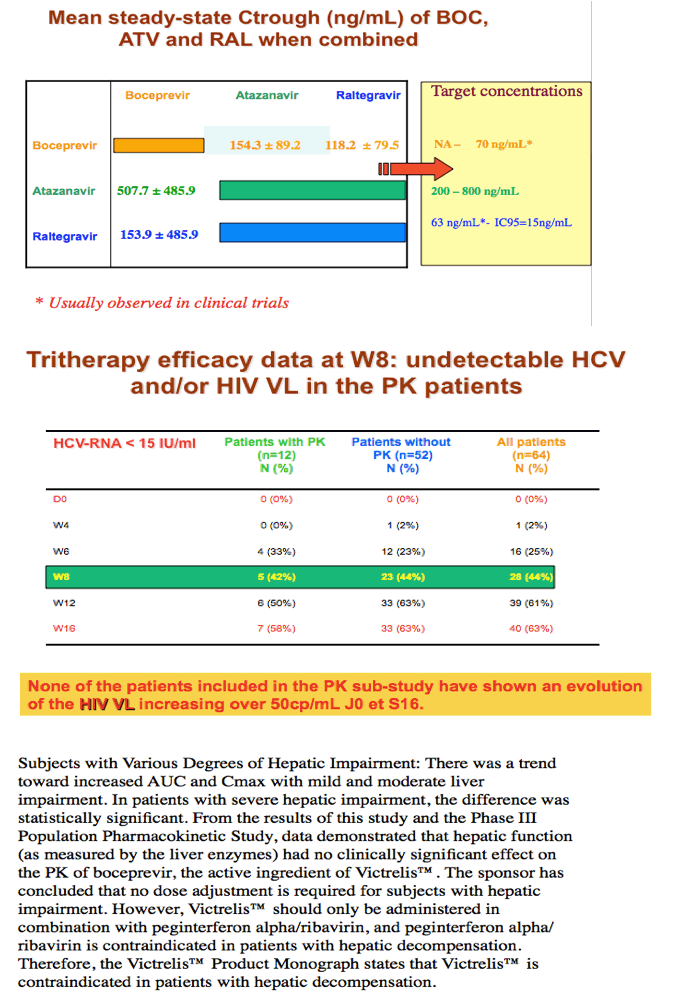
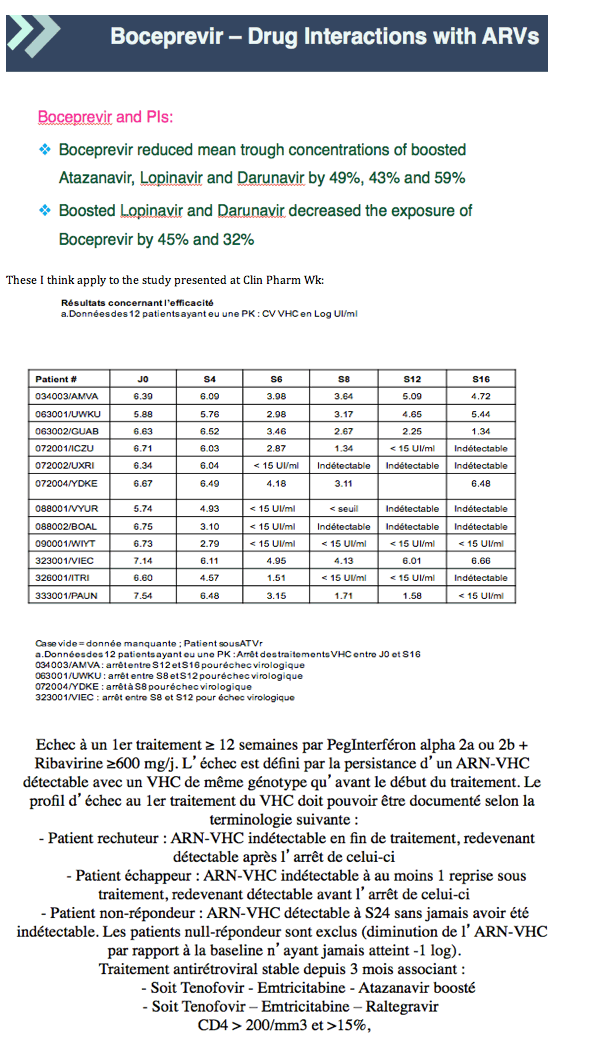
Background: There are PK interactions between BOC and HIV PIs in healthy individuals. The aim of this ANRS HC27 substudy was to investigate the impact of BOC on ATV/r and RAL PK in HIV/HCV co-infected pts.
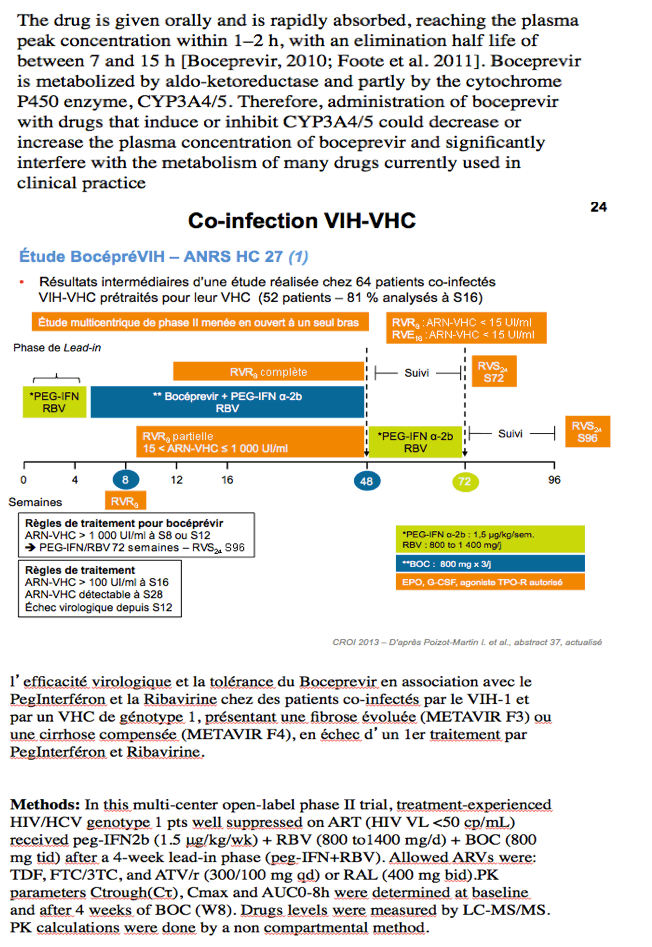
Methods: In this multi-center open-label phase II trial, treatment-experienced HIV/HCV genotype 1 pts well suppressed on ART (HIV VL <50 cp/mL) received peg-IFNα2b (1.5 μg/kg/wk) + RBV (800 to1400 mg/d) + BOC (800 mg tid) after a 4-week lead-in phase (peg-IFN+RBV). Allowed ARVs were: TDF, FTC/3TC, and ATV/r (300/100 mg qd) or RAL (400 mg bid).PK parameters Ctrough(Cτ), Cmax and AUC0-8h were determined at baseline and after 4 weeks of BOC (W8). Drugs levels were measured by LC-MS/MS. PK calculations were done by a non-compartmental method.
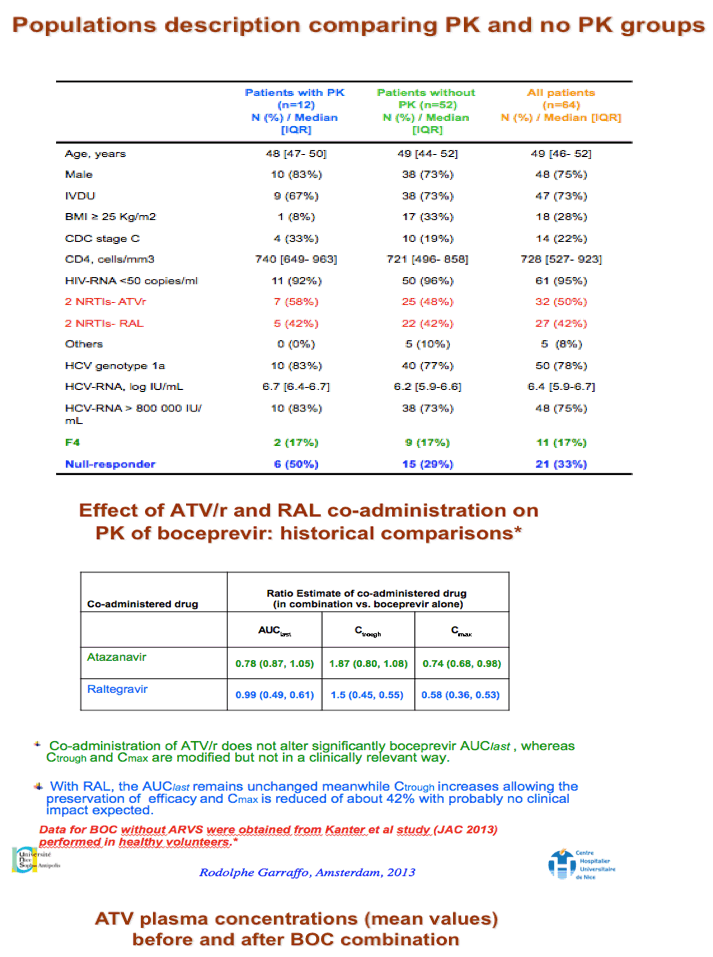
Results: 12 pts completed the PK study, 7 with ATV/r and 5 with RAL. See table for results.
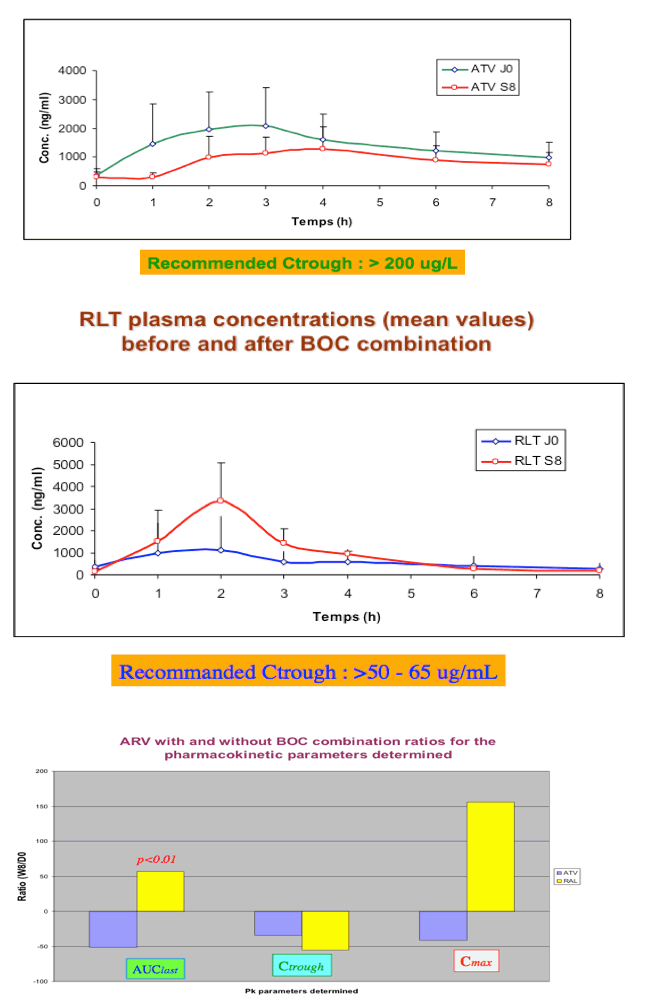
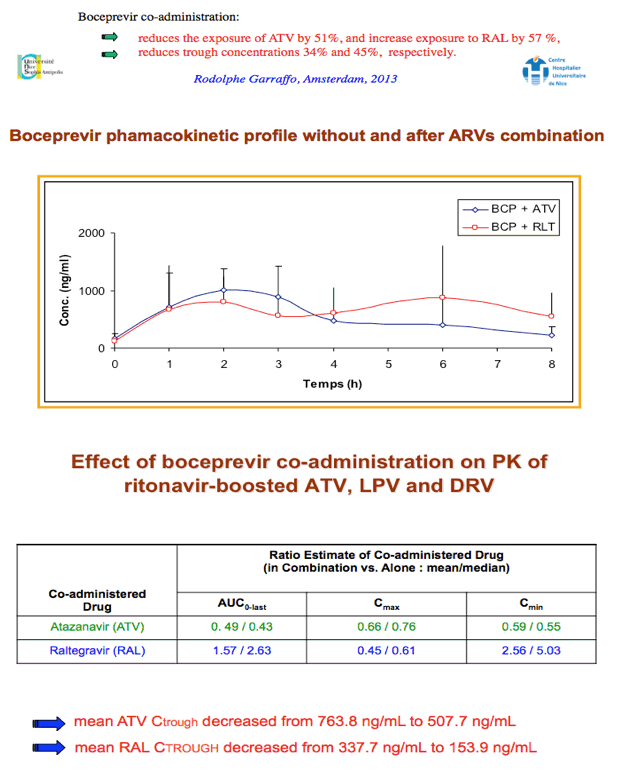
Conclusions: With the limitations of this small study, there was a trend towards lower ATV PK parameters when ATV/r was combined with BOC that was significant only for AUC. There was substantial variability in RAL PK parameters with a trend towards higher RAL AUC0-8h and Cmax and lower RAL Cτ which were not statistically significant. BOC PK was unaffected. Pending more data, carefully monitoring of HIV replication in pts under ATV/r or RAL-based regimens receiving BOC would be appropriate.
ANRS Clinical Study in patients presented at CROI-
CROI: ANRS-HC27 BocepreVIH Interim Analysis: High EVR with Boceprevir+peg-IFN+ RBV in HCV/HIV-co-infected Patients with Previous Failure to peg-IFN+RBV - (03/08/13)
Rodolphe Garraffo, Isabelle Poizot-Martin, Lionel Piroth, Thibaud Lavrut Stanislas Pol, Eric Bellissant, Elina Teicher, Caroline Solas,, Marc Bourliere, Jean-Michel Molina and ANRS HC27 BOCEPREVIH Study Group



|
| |
|
 |
 |
|
|