 |
 |
 |
| |
C-SWIFT: Grazoprevir (MK-5172) + Elbasvir (MK-8742) + Sofosbuvir in Treatment-Naive Patients With Hepatitis C Virus Genotype 1 Infection, With and Without Cirrhosis, for Durations of 4, 6, or 8 Weeks (Interim Results)
|
| |
| |
Reported by Jules Levin
AASLD Boston 2014 Nov 8-11
from Jules: WHAT DO THESE STUDY FINDINGS MEAN: This study needs to be considered in perspective. this was simply an initial small preliminary proof-of-concept study looking at 4, 6 & 8 weeks therapy with the 2 current Merck HCV drugs, their protease MK5172 + their NS5A MK8742 in combination with Sofosbuvir. First, the numbers of patients in all the arms of this study were relatively small. The 8 weeks duration therapy had good SVR rates with 93-100% SVR rates and the numbers of patients in this group were small. The 4 weeks duration group had a low SVR rate of 35-60% & the 8 weeks duration group did better but not that great either with 81-88% SVR compared to what we see in other studies of 12 weeks duration, but these results are based on a study with THESE DRUGS looked at in this study, obviously. Merck purchased Idenix a few months ago to acquire their nukes, there are at least 2, and Merck has a next generation NS5A inhibitor for which their preclinical data shows it to be perhaps more potent & active against potentially pre-existing NS5A mutations compared with the current generation of NS5A inhibitors including Merck's current NS5A inhibitor MK8742 they used in this study. Of course the preclinical findings will be explored in a study in patients to confirm what they see in preclinical study. As well, the dosing of the new nuke they acquired from Idenix may be such that additional potency may be able to be attained with it. SO, the point is that with more potent drugs in a regimen, perhaps 4 or 6 weeks duration might be successful in achieving high SVR rates. I am sure Merck will do these studies of 4, 6, 8 & 12 weeks with these new drugs in a new regimen including their new HCV drugs to see if 4 or 6 weeks duration is possible.
Eric Lawitz,1 Fred Poordad,1 Julio A. Gutierrez,1 Barbara Evans,2 Peggy Hwang,2 Anita Howe,2 Hwa-Ping Feng,2 Michael Robertson,2 Janice Wahl,2 Eliav Barr,2 Barbara Haber2
1The Texas Liver Institute, University of Texas Health Science Center, San Antonio TX, USA; 2Merck & Co., Inc., Whitehouse Station, NJ, USA


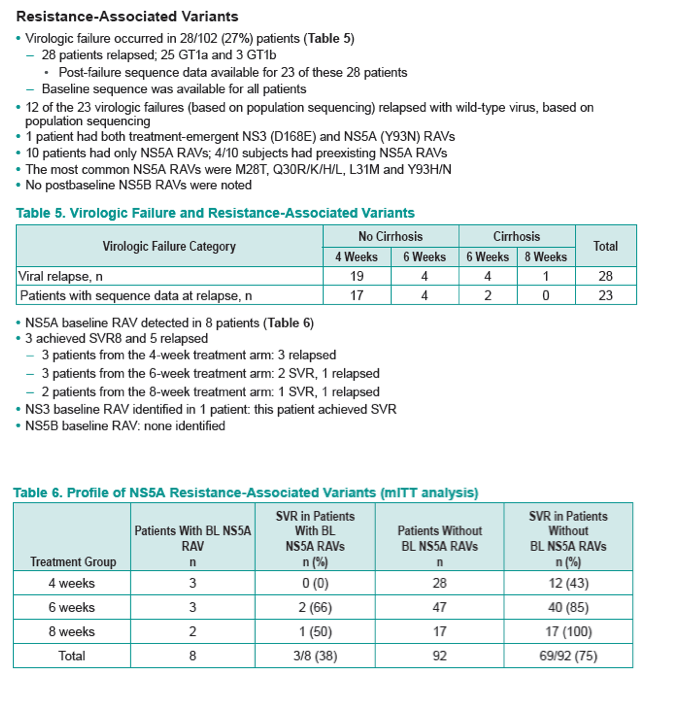
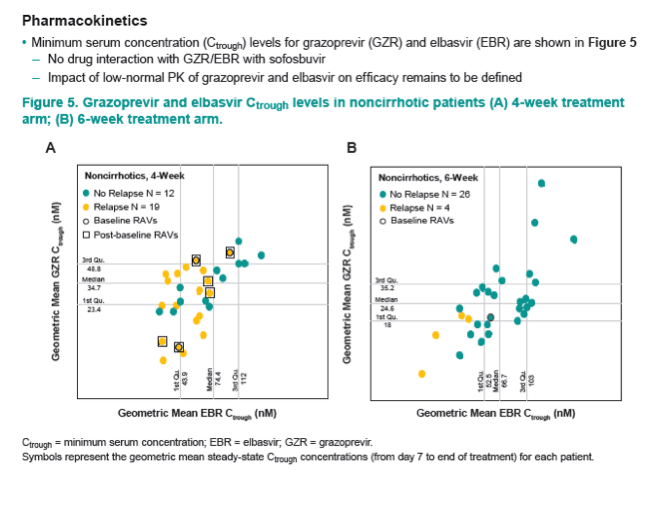
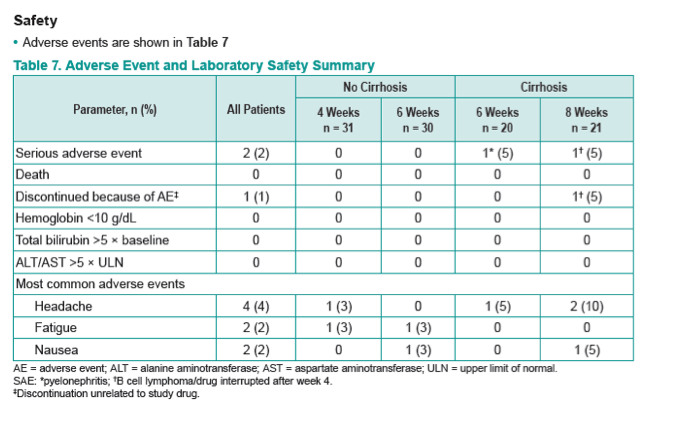
|
| |
|
 |
 |
|
|