 |
 |
 |
| |
The Safety and Efficacy of Entecavir and Tenofovir Combination Therapy for Chronic Hepatitis B in Patients with Previous Nucleos(t)ide Treatment Failure: Week 96 Results of the ENTEBE study
|
| |
| |
Reported by Jules Levin
AASLD 2014 Nov 7-11 Boston
Fabien Zoulim,1 Maciej S Jablkowski,2 Mircea Diculescu,3
Jörg Petersen,4 Patrick Marcellin,5 Soumaya Bendahmane,6 Aleksandra Kedzierska,7 Krzysztof Simon,8 Harry LA Janssen9,10
1Hepatology Department, Hospices Civils de Lyon, INSERM U1052, Lyon University, Lyon, France;
2. Department of Infectious and Liver Diseases, Medical University of Lodz, Poland; 3. Centre of Gastroenterology and Hepatology, Fundeni Clinical Institute, Bucharest, Romania; 4. IFI Institute for Interdisciplinary Medicine, Asklepios Klinik St Georg, Hamburg, Germany; 5. Hôpital Beaujon, Assistance Publique Hôpitaux de Paris, University of Paris 7, and INSERM Unité 773, Centre de Recherches Claude Bernard sur les Hepatites Virales, Clichy, France; 6. Bristol-Myers Squibb, Braine-I'Alleud, Belgium; 7. Bristol-Myers Squibb, Paris, France; 8. Division of Infectious Diseases and Hepatology, Wroclaw University of Medicine, Wroclaw, Poland; 9. Department of Gastroenterology & Hepatology, Erasmus Medical Center, University Medical Center Rotterdam, the Netherlands; 10. Liver Clinic, Toronto Western & General Hospital, University Health Network, Toronto, Canada.



Program abstract:
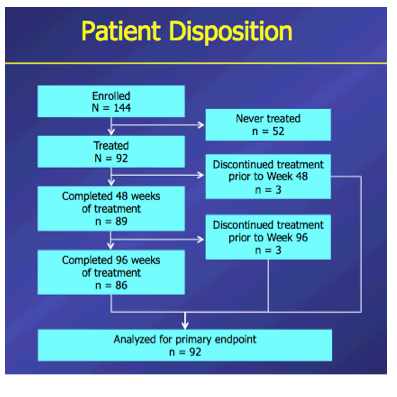
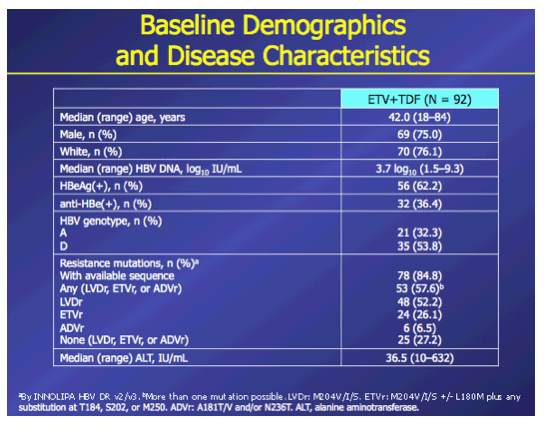
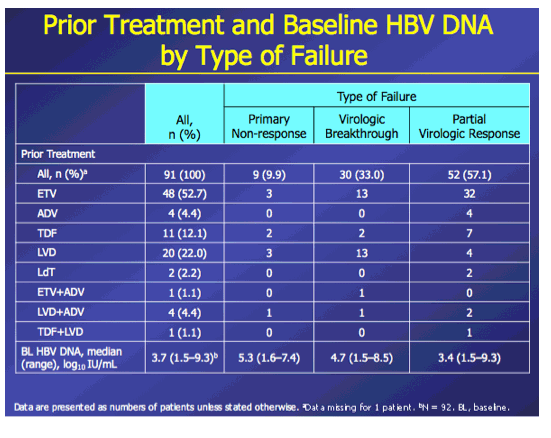
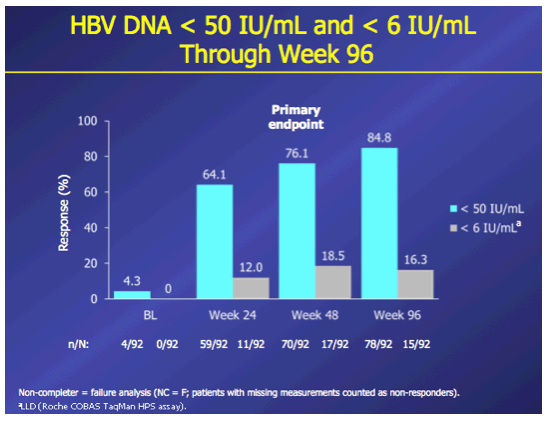
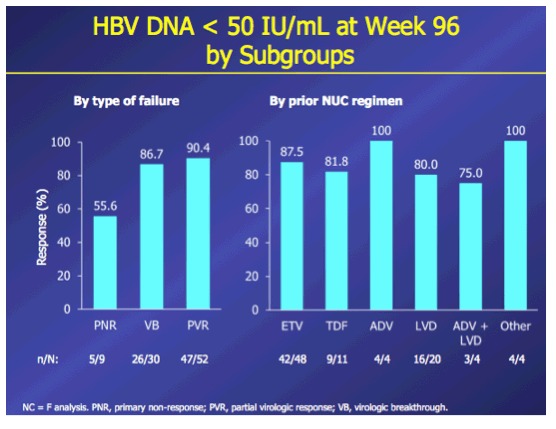
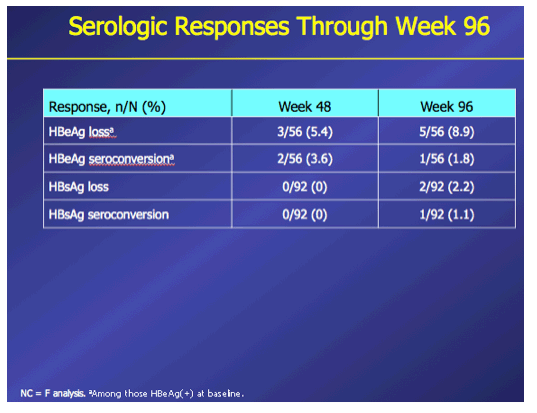
Introduction: In patients with chronic hepatitis B (CHB) who failed on prior nucleos(t)ide (NUC) therapy, rescue therapy should involve an effective antiviral regimen that is active against any existing drug-resistant hepatitis B virus (HBV) variants. Combination therapy with entecavir (ETV) and tenofovir disoproxil fumarate (TDF), two potent agents with non-overlapping resistance profiles, may provide a single regimen suitable for all patients who failed on other NUC regimens. Here we present Week 96 results of the ENTEBE study assessing ETV+TDF for patients with prior failure on NUC therapy. Methods: In this single-arm, open-label, multicenter study, CHB patients with prior non-response, partial response, or virologic breakthrough on NUC therapy were treated with ETV (1 mg) plus TDF (300 mg) for 96 weeks. The primary endpoint was the proportion of patients with HBV DNA <50 IU/mL (Roche COBAS TaqMan-HPS Assay) at Week 48 (non-completer=failure). Secondary endpoints included proportions of patients with antiviral responses at Week 96, safety, and resistance to ETV or adefovir (ADV). Results: Overall, 92 patients were treated; 6 patients discontinued prior to Week 96. At baseline, 65% of patients were HBeAg(+), median HBV DNA was 3.7 log10 IU/mL. Prior NUC treatment included monotherapy with ETV (53%), lamivudine (LVD; 22%), TDF (12%), (ADV; 4%), or telbivudine (LdT; 2%), or combinations of these agents (7%); 58% of patients had evidence of single- or multidrug resistance mutations (LVD: 52%, ETV: 26%; ADV: 7%). At Week 48, 76% (70/92) of patients achieved the primary endpoint (HBV DNA <50 IU/mL). By Week 96, 85% (78/92) of patients had HBV DNA <50 IU/mL, including 80% (16/20) with prior failure on LVD, 100% (4/4) on ADV, 88% (42/48) on ETV, 82% (9/11) on TDF, 100% (2/2) on LdT, and 83% (5/6) on combination therapy. No treatment-emergent resistance to ETV or ADV was observed. Six patients had on-treatment serious adverse events, none of which were considered related to study treatment. One patient died from hepatocellular carcinoma. Conclusions: In patients who failed prior NUC therapy, 96 weeks of ETV+TDF combination therapy was well tolerated and achieved virologic suppression in the majority (85%) of patients, irrespective of the type of prior NUC, with no new resistance development.




|
| |
|
 |
 |
|
|