 |
 |
 |
| |
Long-Term Treatment with Tenofovir Disoproxil Fumarate for Chronic Hepatitis B Infection Is Safe and Well Tolerated and Associated with Durable Virologic Response with No Detectable Resistance: 8 Year Results from Two Phase 3 Trials
|
| |
| |
Reported by Jules Levin
AASLD 2014 Boston Nov 7-11
Patrick Marcellin1, Edward Gane2, Robert Flisiak3, Huy Trinh4, Joerg Petersen5, Selim Gurel6, Kelly Kaita7, Iskren A. Kotzev8, Naoky Tsai9, Raul Aguilar Schall10, John F. Flaherty10, Anuj Gaggar10, Kathryn M. Kitrinos10, G. Mani Subramanian10, John G. McHutchison10, Jacob George11, Harry Janssen12, Maria Buti13
1Hopital Beaujon, Clichy, France; 2Auckland City Hospital, Auckland, New Zealand; 3Medical University of Bialystok, Bialystok, Poland; 4San Jose Gastroenterology, San Jose, CA; 5Asklepios Klinik St. George, University of Hamburg, Hamburg, Germany; 6Uludag Universitesi Tip Fakultesi, Bursa, Gorukle, Turkey; 7University of Manitoba, Winnipeg, Manitoba, Canada; 8University Hospital Sveta Marina, Varna, Bulgaria; 9University of Hawaii at Manoa, Honolulu, HI; 10Gilead Sciences, Inc., Foster City, CA, USA; 11Storr Liver Unit, Westmead Hospital, Sydney, NSW, Australia; 12University of Toronto, Toronto, Ontario, Canada; 13Hospital General Universitari Vall d'Hebron and Ciberehd, Barcelona, Spain

Program abstract:
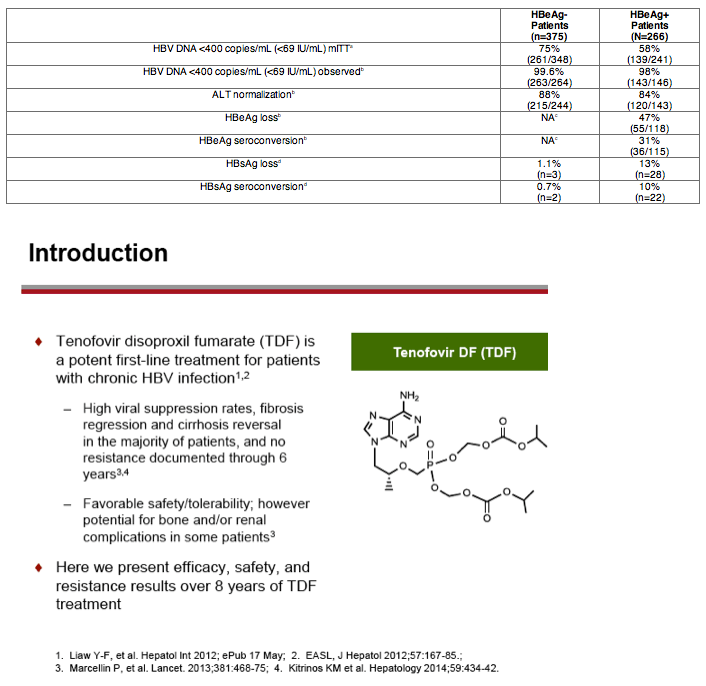
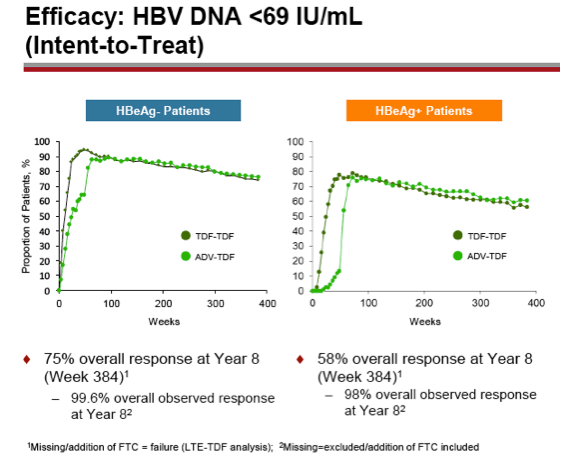
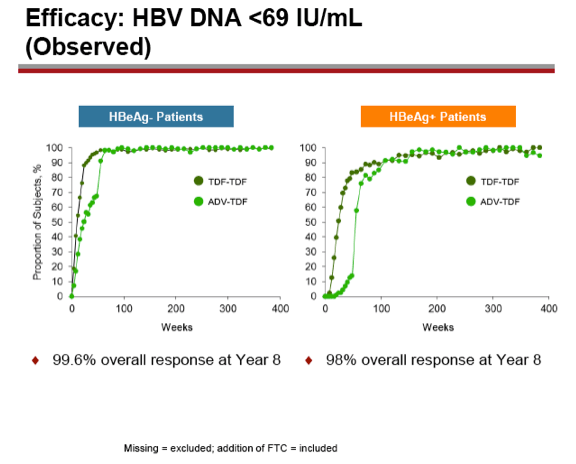
Background: Through 5 years of treatment with tenofovir disoproxil fumarate (TDF) in mostly naïve patients, we reported sustained viral suppression with regression of fibrosis, and reversal of cirrhosis in 74% of patients (Lancet 2013;381:468-75). Further, no evidence of resistance to TDF was seen through Year 6 (J Hepatol. 2014;59:434-42). Here we present Year 8 results, the initially pre-specified end of study period, for two Phase 3 studies in HBeAg- and HBeAg+ chronic hepatitis B patients.
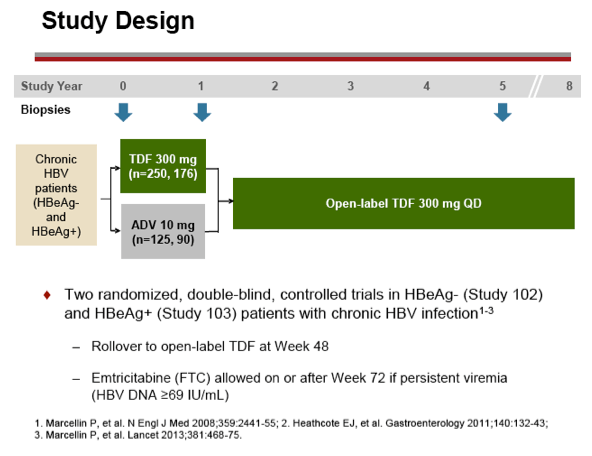
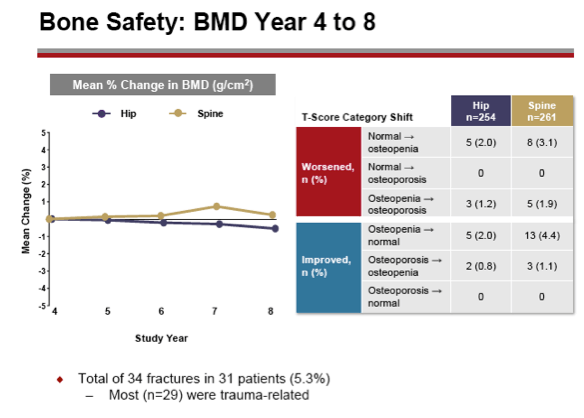
Methods: After 48 weeks of double-blind comparison of TDF to adefovir dipivoxil, all patients were eligible to continue open-label TDF. Patients were assessed every 3 months for efficacy and safety; resistance surveillance was performed annually, and annual bone mineral density (BMD) assessments by DXA were included starting at Year 4.
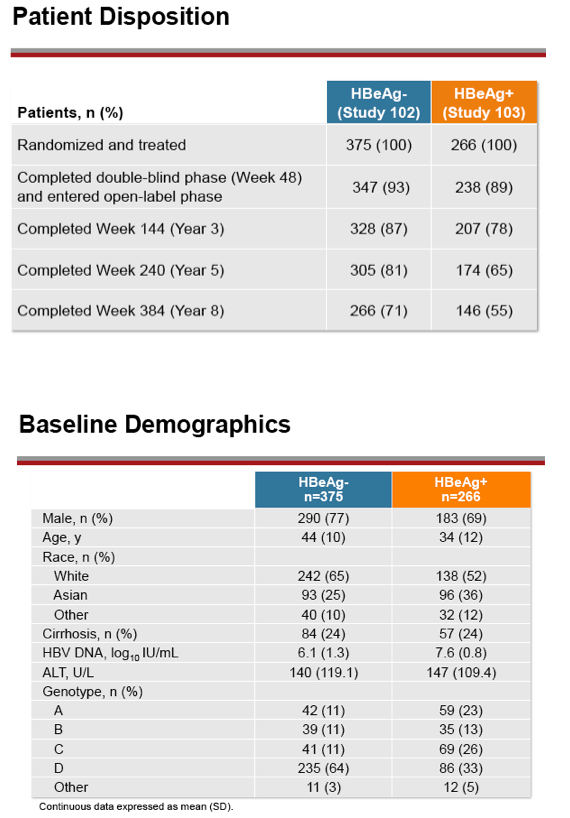
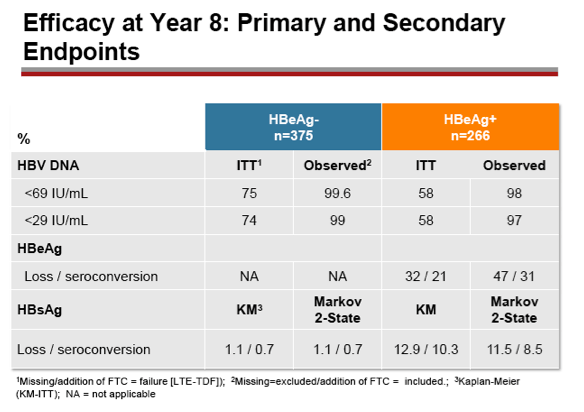
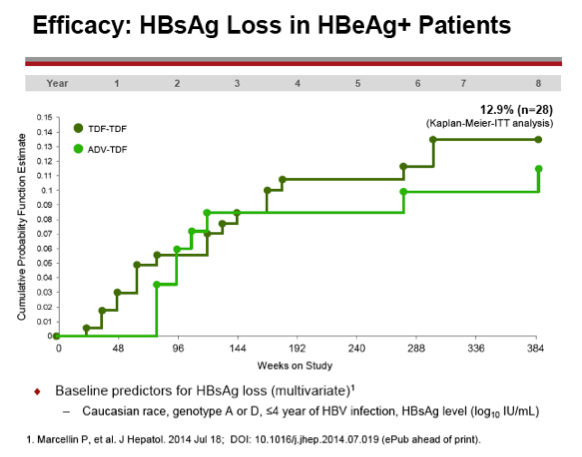
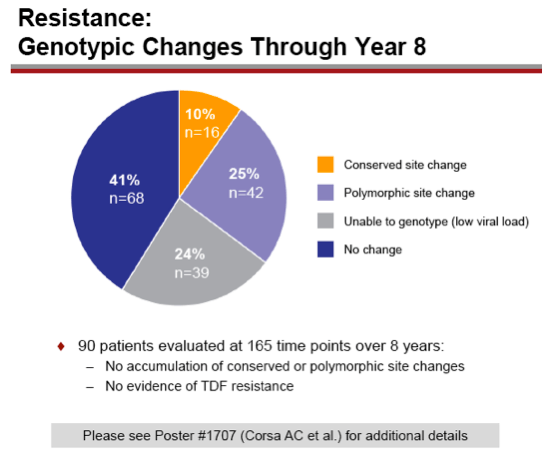
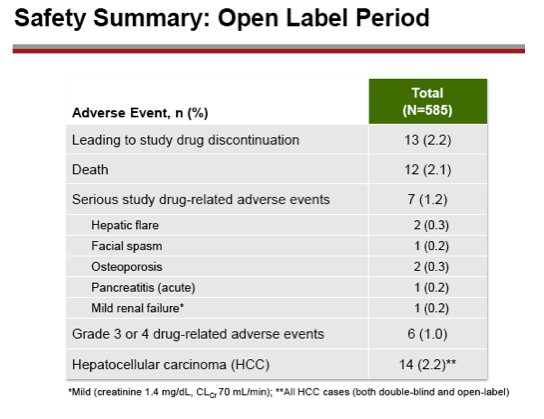
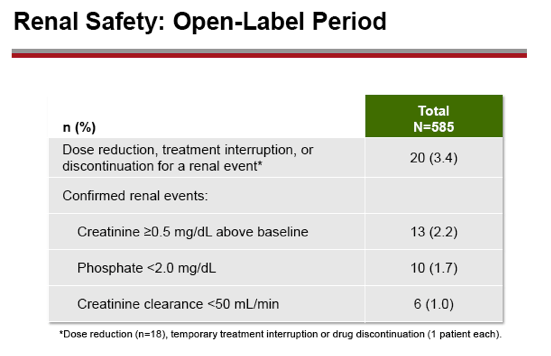
Results: A total of 641 patients were randomized and treated; 585 (91%) entered the TDF extension phase at Year 1, and 412 (64%) remained on study at Year 8. Efficacy results are summarized in the table. Durable viral suppression was maintained, and 7 additional patients (5 HBeAg+ and 2 HBeAg- ) experienced loss of HBsAg (5 patients with seroconversion to anti-HBs) between Years 5-8. No resistance to TDF was detected through Year 8. Through Year 8, a confirmed renal event (either ≥0.5 mg/dL increase in serum creatinine, or serum phosphorus <2 mg/dL, or creatinine clearance <50 mL/min) was observed in 2.2% of patients, and BMD (T scores) of hip and spine were stable between Years 4-8.
Conclusions: Long term results from these two studies show TDF to be safe and effective with no evidence of resistance through 8 years.



|
| |
|
 |
 |
|
|