 |
 |
 |
| |
A Phase I/IIa Study Assessing 7-Day Dosing of MK-3682 (formerly IDX21437)in Subjects Infected With Hepatitis C Virus (HCV)
|
| |
| |
Reported by Jules Levin
AASLD Boston Nov 7-11 2014
EJ Gane1, E Sicard2, S Popa3, X-J Zhou4, M-F Temam5, J Chen4, D Frank4, EF Donovan4, K Pietropaolo4, D Mayers4
1Auckland Clinical Studies, Auckland, NZ; 2Algorithme Pharma, Montreal, QC, Canada; 3Arensia, Chisinau, Republic of Moldova;4Idenix Pharmaceuticals, Inc., a wholly-owned subsidiary of Merck & Co., Inc., Cambridge, MA, United States; 5Idenix SARL, an MSD company, Montpellier, France

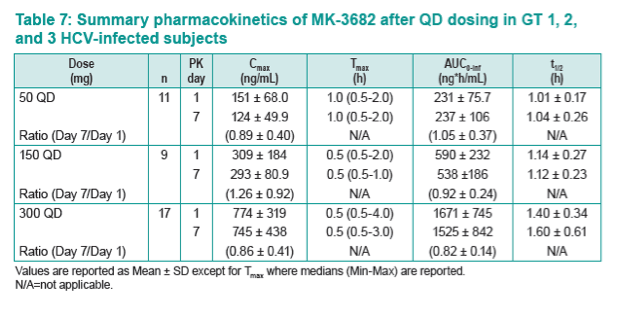
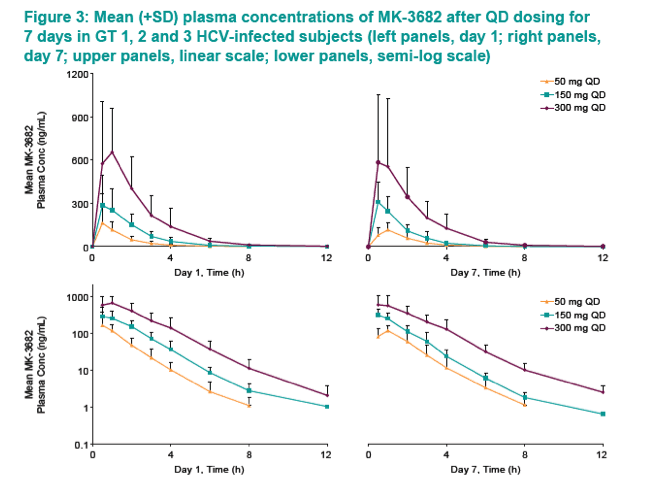
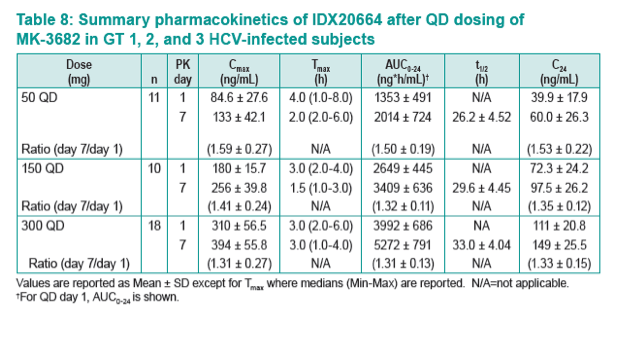
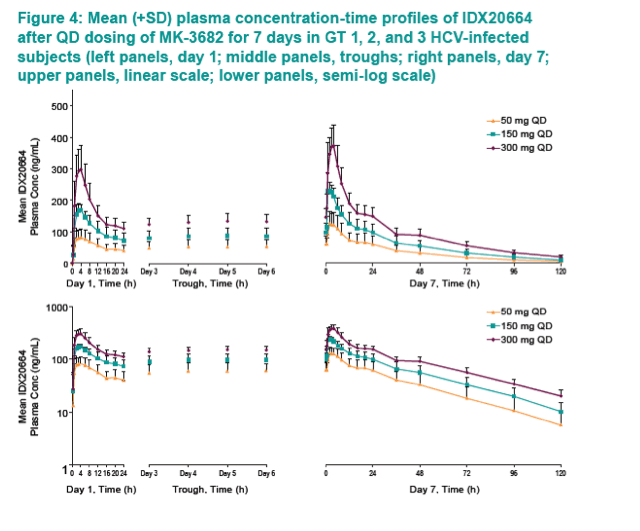
|
| |
|
 |
 |
|
|