 |
 |
 |
| |
Adherence to Prescribed Doses of ABT-450/r/Ombitasvir, Dasabuvir, and Ribavirin in the Phase 3 PEARL-II, PEARL-III, and PEARL-IV Trials
|
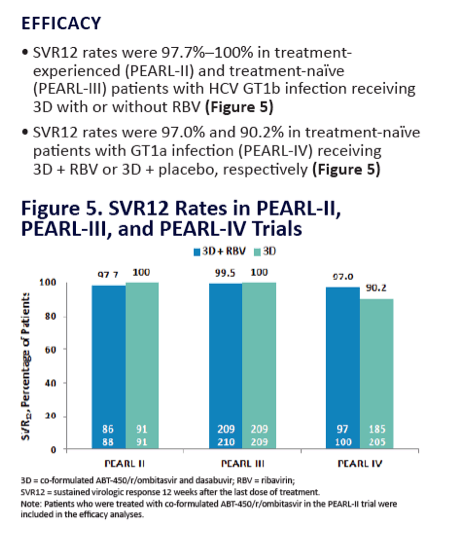
| |
| |
Reported by Jules Levin
AASLD 2014 Nov 7-11 Boston
David Bernstein1, Rui T Marinho2, Daniel E Cohen3, Fritz Bredeek4, Ferenc Schneider5, Gunnar Norkrans6, Manuela Curescu7, Michael Bennett 8, Marina Maevskaya9, Jeff rey Fessel10, Wangang Xie3, Yan Luo3, Jeff rey V Enejosa3
1Hofstra North Shore-LIJ School of Medicine, Manhasset, New York, United States; 2Centro Hospitalar Lisboa Norte, Medical School of Lisbon, Lisbon, Portugal; 3AbbVie Inc., North Chicago, Illinois, United States; 4Metropolis Medical Group, San Francisco, California, United States; 5Markusovszky Hospital, Szombathely, Hungary;
6Sahlgrenska University Hospital, Goteborg, Sweden; 7Life Search SRL, Timisoara, Romania; 8Medical Associates Research Group, San Diego, California, United States; 9First Moscow State Medical Universita naIM Sechenov, Moscow, Russia; 10Kaiser Permanente, San Francisco, California, United States








|
| |
|
 |
 |
|
|