 |
 |
 |
| |
AASLD/IDSA Guidelines Improves Restrictions Language
|
| |
| |
Jules Levin, NATAP
"Our recent addition to the Guidance prepared by a committee of leading liver experts from AASLD and The Infectious Diseases Society of America (IDSA) proposed that the sickest patients be treated first, but all patients who receive advice from their doctor to take newest medications should not be denied.......limitations of workforce and societal resources may limit the feasibility of treating all patients within a short period of time........ The decision across the board should be in the hands of the clinician and the patient to make the decision. Unfortunately payers across America are denying treatment when a doctor has prescribed it for their patient. We adamantly disagree with this decision. Our Guidance is not intended to be used by payers to deny access to treatment"
click on Practice Guidelines at http://www.aasld.org/Pages/Default.aspx
--------------
WHEN AND IN WHOM TO INITIATE HCV THERAPY.....Fri Oct 24
http://www.hcvguidelines.org/full-report/when-and-whom-initiate-hcv-therapy
Successful hepatitis C treatment results in sustained virologic response (SVR), which is tantamount to virologic cure, and as such, is expected to benefit nearly all chronically infected persons. Evidence clearly supports treatment in all HCV-infected persons, except those with limited life expectancy (less than 12 months) due to non-liver-related comorbid conditions (See Unique Patient Populations). Urgent initiation of treatment is recommended for some patients, such as those with advanced fibrosis or compensated cirrhosis (see Table 1).
Recommendations for when and in whom to initiate treatment
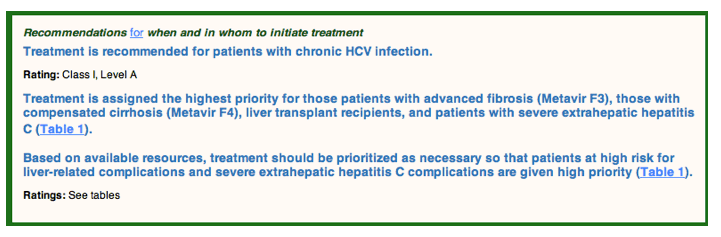
The most immediate and high-impact benefits of SVR will be realized by populations that are at the highest risk for liver-r
---------------------
HCV Treatment Recommendations Now Includes Information on Prioritizing Patients Under Limited Resources
http://www.aasld.org/practiceguidelines/Pages/hcvpatientslimitedresources.aspx
The American Association for the Study of Liver Disease (AASLD) and the Infectious Diseases Society of America (IDSA), in collaboration with the International Antiviral Society-USA (IAS-USA), today released the latest section of their website, www.hcvguidelines.org, which assists clinicians treating patients with hepatitis C virus (HCV). The new section is titled, "When and in Whom to Initiate HCV Therapy."
It is estimated that between 3 and 4 million Americans are infected with HCV and have chronic liver disease as a result. Recent direct-acting antivirals are extremely effective in treating the virus and hold great potential for cure. However, limitations of workforce and societal resources may limit the feasibility of treating all patients within a short period of time.
With the addition of the new section, www.hcvguidelines.org now offers clinicians information on how to prioritize treatment for those patients who will derive the most benefit or will have the greatest impact on limiting further HCV transmission. Highest priority should be given to patients with advanced fibrosis with compensated cirrhosis and liver transplant recipients and high priority given to patients at high risk for liver-related complications and severe extra-hepatic HCV complications. The guidance provides further detailed information on additional conditions that warrant prioritization of treatment.
The website, developed by a panel of 27 liver disease and infectious diseases specialists and a patient advocate, provides recommendations based on the latest evidence and on consensus of the panelists. It will be updated regularly to keep pace with improved diagnostic tools and new drug options as they become available.
"The benefits of curing HCV are clear from the standpoint of individual patients as well as that of the health and welfare of our society. This new guidance will help clinicians determine the best course of therapy for each patient given their unique condition," said Barbara Murray, MD, president of IDSA.
The three organizations will continue to work together to provide future sections of the guidance website as well as timely updates. The next two sections are underway and cover monitoring patients following treatment and management of acute HCV infection.
"New therapies recently approved by the FDA and those that are in the pipeline and will reach market soon will completely change the landscape for patients with hepatitis C," said Steven Echard, AASLD chief executive officer. "We have filled a void in providing reputable and timely information to healthcare providers and their patients by addressing whom to treat and when, as well as identifying patients that are in immediate need of treatment and those who can safely wait for the next generation of drugs."
-----------------------
AASLD Position on Treating Patients with Chronic Hepatitis C Virus
http://www.aasld.org/aboutus/publicpolicy/Pages/aasldhcvposition.aspx
The American Association for the Study of Liver Diseases (AASLD) is the leading organization of scientists and health care professionals committed to preventing and curing liver disease, and to promoting liver health and quality patient care. AASLD has always put the patient first by providing clinicians with the latest rigorous data on the best treatments for patients with liver diseases, including hepatitis C.
Our recent addition to the Guidance prepared by a committee of leading liver experts from AASLD and The Infectious Diseases Society of America (IDSA) proposed that the sickest patients be treated first, but all patients who receive advice from their doctor to take newest medications should not be denied. The decision across the board should be in the hands of the clinician and the patient to make the decision. Unfortunately payers across America are denying treatment when a doctor has prescribed it for their patient. We adamantly disagree with this decision.
Our Guidance is not intended to be used by payers to deny access to treatment. In no way does this position contradict the evidence evaluated to produce the Guidance and the recommendation made in the Guidance to treat the sickest first, but recognizes need to treat all.
Some confusion exists about our process of advising physicians. The Guidance committee relied on the best available published evidence and expert opinion. AASLD has produced numerous Practice Guidelines, which require a tremendous amount of time and level of evidence. The Guidance was released based on a thorough review of current scientific evidence along with expert opinion. To wait for further evidence to appear in the literature as opposed to assessing what currently exists and relying on expert opinion -- which is what the committee did -- would be irresponsible to patients and clinicians.
AASLD remains committed to its partnership with IDSA and to continuing to fund and write the practice Guidance. As the only organization solely dedicated to the prevention and cure of liver disease, we applaud the seminal research of our members who made recent treatment options possible. We are concerned about cost, but we also recognize the value of this drug to patients with hepatitis C. It's a one-time treatment, with minimal side effects and a great cure rate.
Treatments currently available and those we are confident will soon receive FDA-approval should be heralded as remarkable advances in curing a disease that was once fatal.
Background on the Development of the Guidance
On September 24, 2014, AASLD and IDSA published online one of the final two sections of their practice Guidance for hepatitis C -- Monitoring Patients Who Are Starting Hepatitis C Treatment, Are in Treatment, or Have Completed Therapy. The other section, Management of HCV Infection, has been approved and will be released in the next few weeks. The Guidance is now complete; however, it will be updated regularly. The online, easily updateable format was created to allow for us to change the Guidance as new drugs are approved by the Food and Drug Administration.
AASLD and IDSA recognized there was a need for clinical guidance. Our Federal partners such as the National Institutes of Health, Centers for Disease Control and Prevention, and the US Department of Health and Human Services were also aware of this need and supported the creation of a practice Guidance to help clinicians who treat patients with hepatitis C. AASLD and IDSA stepped forward to fund and create this Guidance independent of any industry financial support.
--------------------------
The American Association for the Study of Liver Disease (AASLD) and the Infectious Diseases Society of America (IDSA), in collaboration with the International Antiviral Society-USA (IAS-USA), today released the latest section of their website, www.hcvguidelines.org, which assists clinicians treating patients with hepatitis C virus (HCV). The new section is titled, "When and in Whom to Initiate HCV Therapy."
It is estimated that between 3 and 4 million Americans are infected with HCV and have chronic liver disease as a result. Recent direct-acting antivirals are extremely effective in treating the virus and hold great potential for cure.
However, limitations of workforce and societal resources may limit the feasibility of treating all patients within a short period of time.
With the addition of the new section, www.hcvguidelines.org now offers clinicians information on how to prioritize treatment for those patients who will derive the most benefit or will have the greatest impact on limiting further HCV transmission. Highest priority should be given to patients with advanced fibrosis with compensated cirrhosis and liver transplant recipients and high priority given to patients at high risk for liver-related complications and severe extra-hepatic HCV complications. The guidance provides further detailed information on additional conditions that warrant prioritization of treatment.
The website, developed by a panel of 27 liver disease and infectious diseases specialists and a patient advocate, provides recommendations based on the latest evidence and on consensus of the panelists. It will be updated regularly to keep pace with improved diagnostic tools and new drug options as they become available.
"The benefits of curing HCV are clear from the standpoint of individual patients as well as that of the health and welfare of our society. This new guidance will help clinicians determine the best course of therapy for each patient given their unique condition," said Barbara Murray, MD, president of IDSA.
The three organizations will continue to work together to provide future sections of the guidance website as well as timely updates. The next two sections are underway and cover monitoring patients following treatment and management of acute HCV infection.
"New therapies recently approved by the FDA and those that are in the pipeline and will reach market soon will completely change the landscape for patients with hepatitis C," said Steven Echard, AASLD chief executive officer. "We have filled a void in providing reputable and timely information to healthcare providers and their patients by addressing whom to treat and when, as well as identifying patients that are in immediate need of treatment and those who can safely wait for the next generation of drugs."
|
| |
|
 |
 |
|
|