 |
 |
 |
| |
Pill Burden and Treatment Duration Reduce
Adherence to IFN-Free Hepatitis C Therapy in an Urban Cohort
|
| |
| |
Reported by Jules Levin
CROI 2014 March 3-6 Boston, MA
Tess Petersen1, Lori A. Gordon4, Kerry Townsend3, Monica Calderon4, Sreetha Sidharthan1, Rachel Silk2, Anu Osinusi3,5, Henry Masur1, Shyam Kottilil3, Anita Kohli1,2
1 Critical Care Medicine Department, National Institutes of Health Clinical Center, National Institutes of Health, Bethesda, Maryland, 2 Clinical Research Directorate/Clinical Monitoring Research Program, Leidos Biomedical Research, Inc. (formerly SAIC-Frederick, Inc.), Frederick National Laboratory for Cancer Research, Frederick, Maryland, 3 Laboratory of Immunoregulation, National Institute of Allergy and Infectious Diseases, National Institutes of Health, Bethesda, Maryland, 4 Clinical Center Pharmacy Department, National Institutes of Health, Bethesda, Maryland, 5 Institute of Human Virology, University of Maryland, Baltimore

Program abstract-
Background: Potent oral directly acting antiviral (DAA) HCV therapy with low pill burdens and short therapeutic courses are promising replacements for
interferon and ribavirin based regimens. The impact of these improvements on patient adherence and virologic outcomes has not been described.
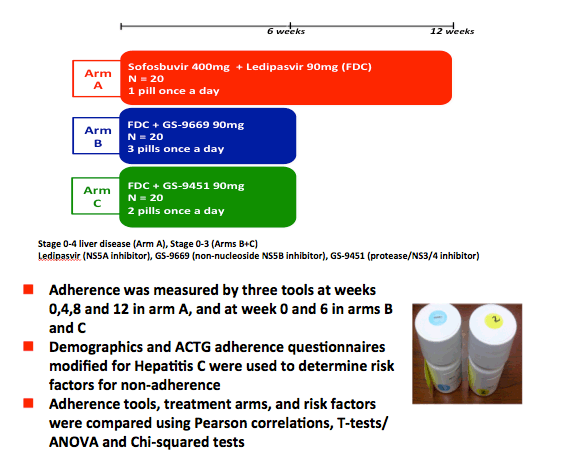
Methodology: Sixty HCV infected, treatment na•ve, genotype 1 patients were enrolled into 3 arms of a phase 2 clinical trial and received: sofosbuvir +
ledipasvir (400/90mg one pill once daily in a fixed dose combination (FDC)) for 12 weeks, FDC + GS-9451 (80mg/day), two pills, once daily for 6 weeks,
or FDC + GS-9669 (500mg/day), three pills, once daily for 6 weeks. Serial measurements of virologic correlates (HCV RNA; deep sequencing of viral
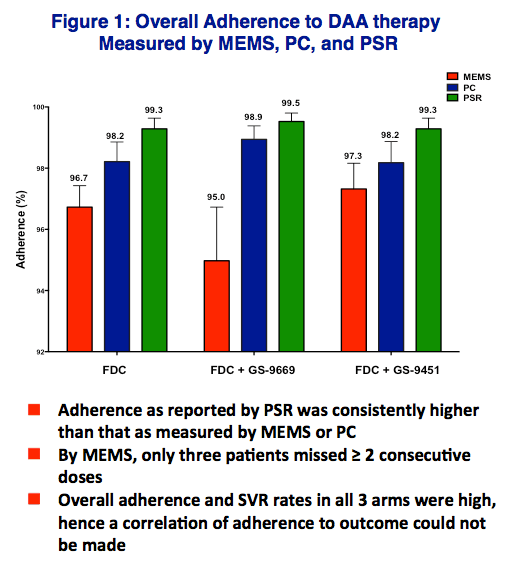
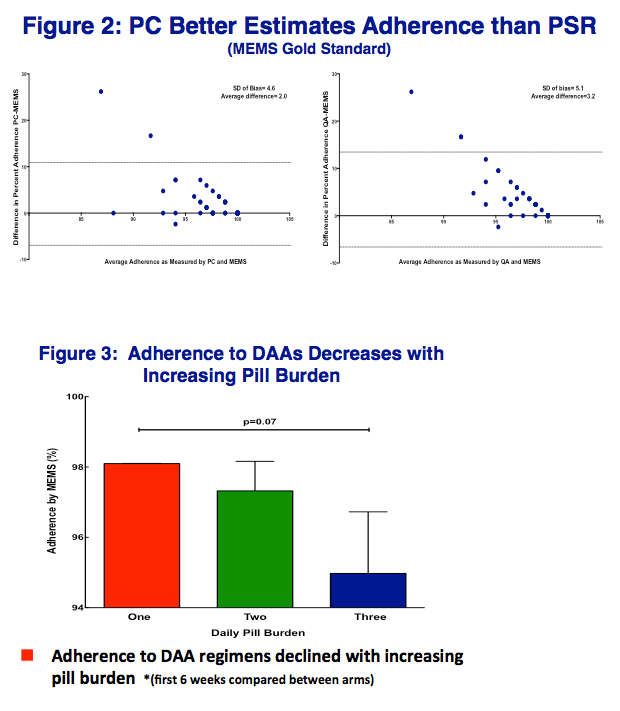
mutations) were performed. Adherence was measured using three tools: MEMS caps (MEMS), pill counts (PC) and patient report (PR). Demographics and
modified ACTG questionnaires were used to determine risk factors for non-adherence. Adherence tools, treatment arms, and risk factors were compared
using Pearson correlations, T-tests/ANOVA and Chi-squared tests respectively.
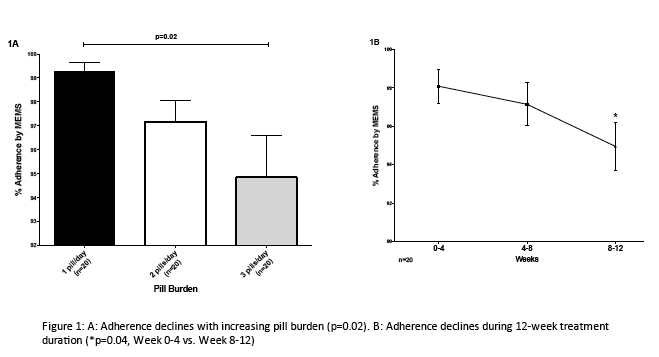
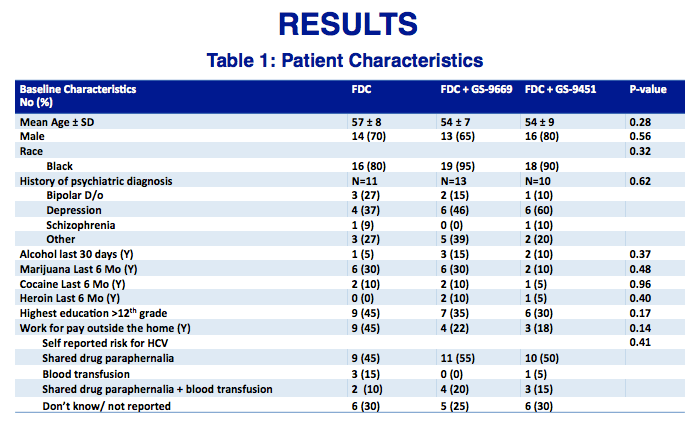
Results: Patients enrolled were African American (88%), male (72%) with ≤ 12th grade education (61%). Psychiatric disease (43%) and recent history of
drug/ETOH abuse (37.5%) were common.
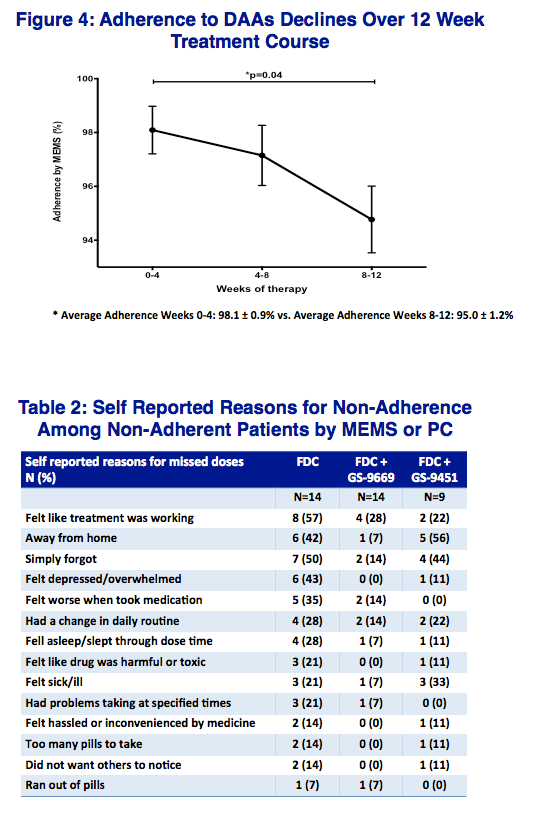
Adherence to DAA regimens declined with increasing pill burden (99.3 ± 1.6% vs. 97.2 ± 3.6% vs. 94.8 ± 7.4%, one, two & three pills/day respectively,
p=0.02 (Fig 1A)). Adherence to therapy declined significantly during the 12-week treatment course (98.1 ± 0.9% vs. 95.0 ± 1.2% weeks 0-4 vs. weeks
8-12, p=0.04 (Fig1B).) Missed doses by MEMS correlated with PC (R2=0.24, p<0.0002), but neither with PR (p=0.14 & p=0.84 respectively). Three
patients missed ≥2 consecutive doses. Adherence was similar in patients with and without early viral suppression (< LLOQ at week 4) and time to viral
suppression similar between adherent and non-adherent (defined as 0.05). Risk factors for non-adherence were feeling inconvenienced (p=0.03) and
children living at home (p=0.01). Common reasons for non-adherence were feeling that drugs were working (39%), forgetting (35%) and absence from
home (32%).
Conclusions: Adherence to short courses of DAA therapy with 1-3 pills once a day was excellent in an urban population with multiple risk factors for nonadherence.
Increased pill burden and duration of treatment decreased adherence. Continued education regarding adherence despite on-therapy viral load
decline may improve outcomes.

|
| |
|
 |
 |
|
|