 |
 |
 |
| |
Safety and Efficacy of IFN-free, Sofosbuvir/RBV Therapy in HIV/HCV Liver Transplanted Patients
|
| |
| |
Sofosbuvir and Daclatasvir Combination Therapy in a Liver Transplant Recipient With Severe Recurrent Cholestatic Hepatitis C - (11/25/13)
AASLD: Pretransplant Sofosbuvir and Ribavirin to Prevent Recurrence of HCV Infection After Liver Transplantation - (11/06/13)
AASLD: Sofosbuvir and Ribavirin for the Treatment of Established Recurrent Hepatitis C Infection After Liver Transplantation: Preliminary Results of a Prospective, Multicenter Study - (11/05/13)
AASLD: Initial Evaluation of the Sofosbuvir Compassionate Use Program for Patients With Severe Recurrent HCV Following Liver Transplantation - (11/06/13)
AASLD: (telaprevir/boceprevir) Sustained Virological Response After Protease Inhibitor-based Therapy For Hepatitis C Recurrence After Liver Transplantation: A Multicentric European Experience - (11/18/13)
Reported by Jules Levin
CROI 2014 March 3-6 Boston, MA
Ana Moreno1*, Maria J Perez-Elias1, Rafael Barcena2, CarmenQuereda1, Jose L Casado1, Fernando Dronda1, Santos del Campo2,Maria L. Mateos3, Alberto Diaz1and Santiago Moreno1
Services of Infectious Diseases1, Liver Transplant Unit2, and Microbiology (Viral Hepatitis)3. Hospital Ram—n y Cajal.Madrid. Spain.

Program abstract-
Background: Long-term outcome in HCV/HIV-coinfected subjects undergoing liver transplantation (LT) relies on the management of HCV recurrence.
Currently available peg-IFN-based therapies are associated to high toxicity and low antiviral efficacy, leading to worse outcomes in comparison to HCV
monoinfected subjects.
Methodology: To describe for the first time the on-treatment safety and efficacy of the combination of 400mg/day of sofosbuvir (SFB) -a new direct
antiviral agent against HCV targeting the HCV-NS5B polymerase- plus ribavirin (RBV) in 7 HIV/HCV-coinfected patients with HCV recurrence after LT.
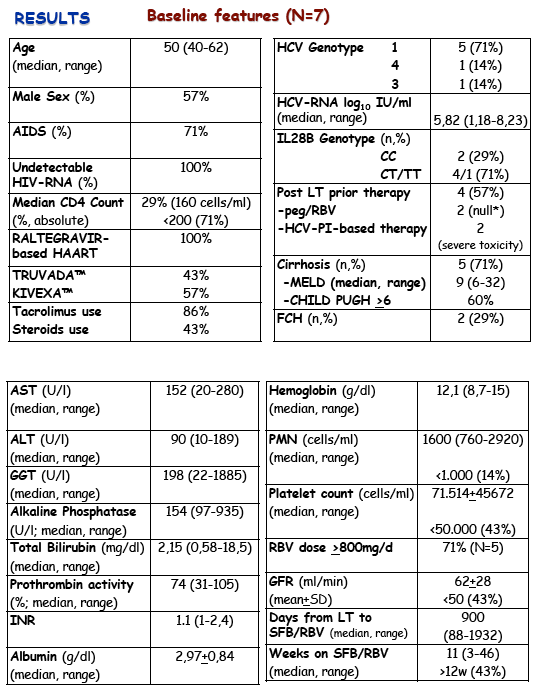
Results: Mean age 49±7, male 57%, 71% prior AIDS, 100% undetectable HIV-RNA, median CD4 counts 160 cells/ml (47-451). 71% HCV-G1 (n=5),
HCV-G3 (n=1), HCV-G4 (n=1). Most subjects (71%) were IL28B non-CC. Only P2 was completely na•ve to anti-HCV therapy (14%). Previously to SFB/
RBV 57% (n=4) had already received post-LT antiviral therapy, with standard peg-IFN/RBV (n=2, null response in both), or triple therapy with boceprevir
(n=1) or telaprevir (n=1), in both with premature discontinuation due to life threatening toxicity despite negative HCV-RNA. All subjects showed histological
severity: cirrhosis 71% (n=5, 60% CHILD-PUGH >6 and prior decompensation in 40%), 29% (n=2) fibrosing cholestatic hepatitis (FCH), both female
with HCV-G1. Overall 86% were on tacrolimus-based immunosuppresive therapy, 43% were currently on steroids. All patients were on raltegravir-based
HAART: 4 with KIVEXA™ (fixed dose 3TC/Abacavir), 3 with TRUVADA™ (fixed dose FTC/Tenofovir), P7 also received maraviroc. Median time from LT to
therapy was 900 days (88-1932). To date, the median time on SFB/RBV is 7 weeks (2-40) and three (43%) have completed at least 12 weeks. Median
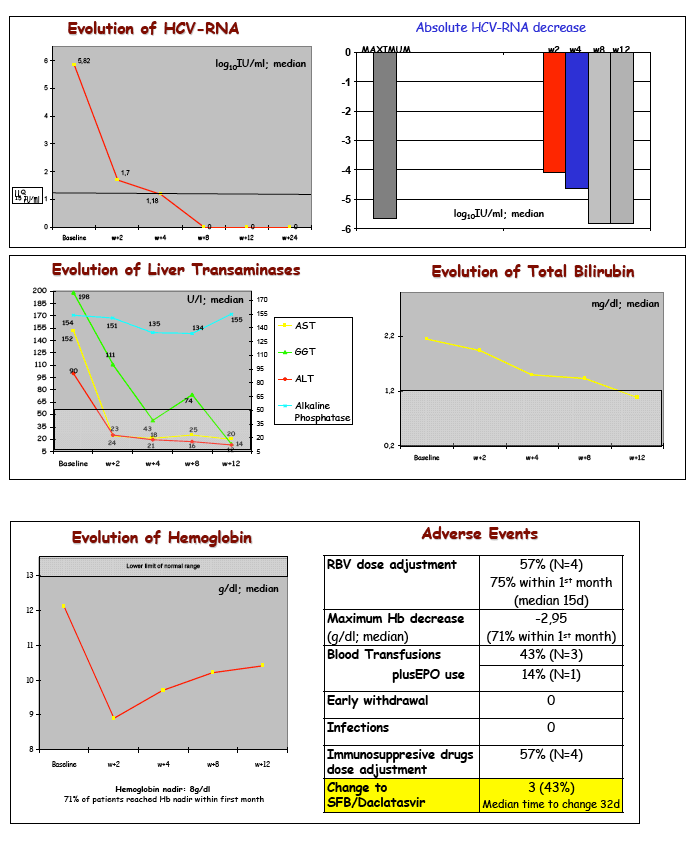
baseline HCV-RNA was 5.73log10 IU/ml. Median HCV-RNA decreases during therapy: w+2, -3,94log10IU/ml, w+4, -4,15log10IU/ml; 100% of patients
with at least 4 weeks of therapy (n=6) reached RVR (<15 IU/ml at w4). During therapy there were no cases of HCV-RNA or HIV-RNA rebound. Median
initial dose of RBV was 800mg/d, with dose adjustments needed in 4 subjects (57%), all of them within the first 8 weeks, in three with blood transfusions.
EPO was transiently used only in P3 (FCH). Liver transaminases reached normal vales within the fist two weeks in 100%. There were not premature
discontinuations, infections, or SFB-related adverse events.
Conclusions: An IFN-free, SFB/RBV regimen showed an excellent safety and efficacy profile in HIV/HCV-coinfected patients with severe liver damage
related to HCV recurrence after LT. 100% reached RVR, with rapid normalization of liver function tests. Viral response was the rule regardless baseline
HCV-RNA, IL28B or HCV genotype, or previous pattern of response to peg-IFN based therapy.

|
| |
|
 |
 |
|
|