 |
 |
 |
| |
NOVEL INTERVENTIONS TO TARGET INCREASED INFLAMMATION IN HIV-INFECTED INDIVIDUALS
|
| |
| |
excerpted from this CROI NATAP Report:
21st Conference on Retroviruses and Opportunistic Infections: Review - Eric S. Daar, M.D. Chief, Division of HIV Medicine Harbor-UCLA Medical Center Professor of Medicine David Geffen School of Medicine at UCLA - (03/21/14)
Several explanations have been proposed to explain persistent inflammation in the setting of virologic suppression in HIV-infected individuals. A leading hypothesis is that during the first weeks of HIV infection there is massive loss of lymphoid tissue in the gastrointestinal tract that allows for bacterial translocation and resultant circulating lipopolysaccharide (LPS) and monocyte activation. This hypothesis can be tested by determining if agents that decrease intestinal bacteria and endotoxemia would decrease levels of systemic inflammation. In addition, other agents known to reduce inflammation could also be studied to assess the impact on markers of inflammation and end organ disease.
Headline:
· Several drugs with potential to influence levels of microbial translocation and gut driven inflammation showed little effect on markers of endotoxemia or inflammation.
· Statin therapy reduces systemic inflammation.
Study findings:
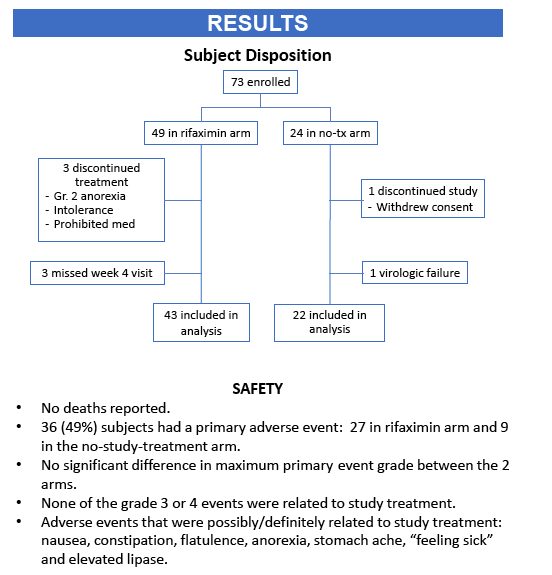
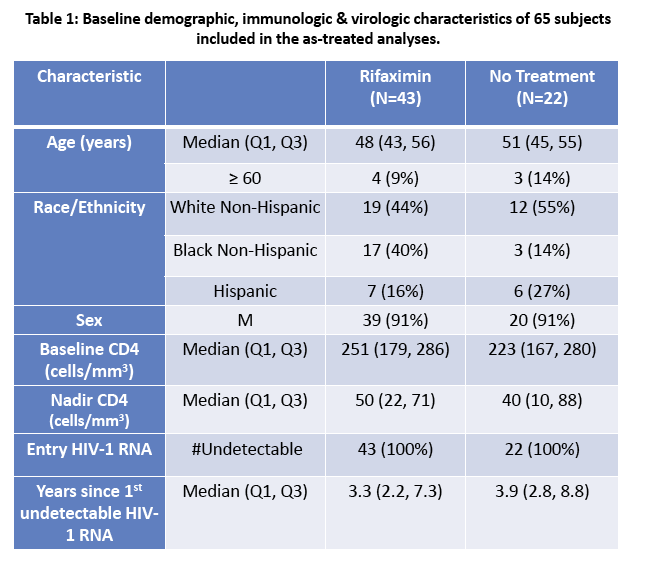
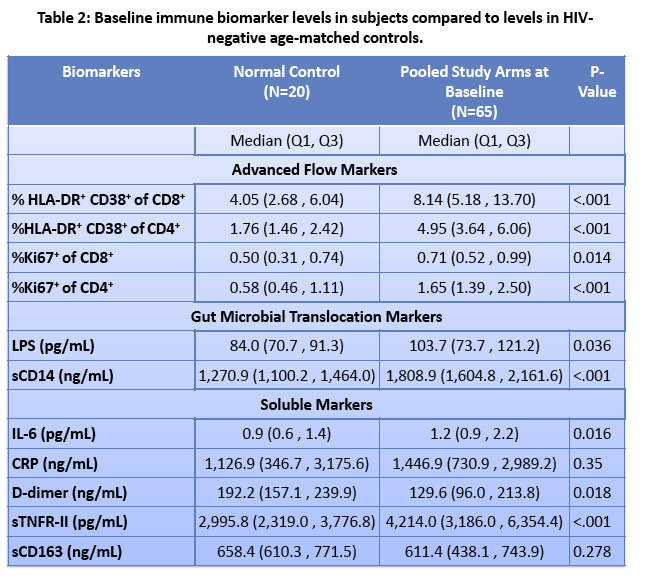
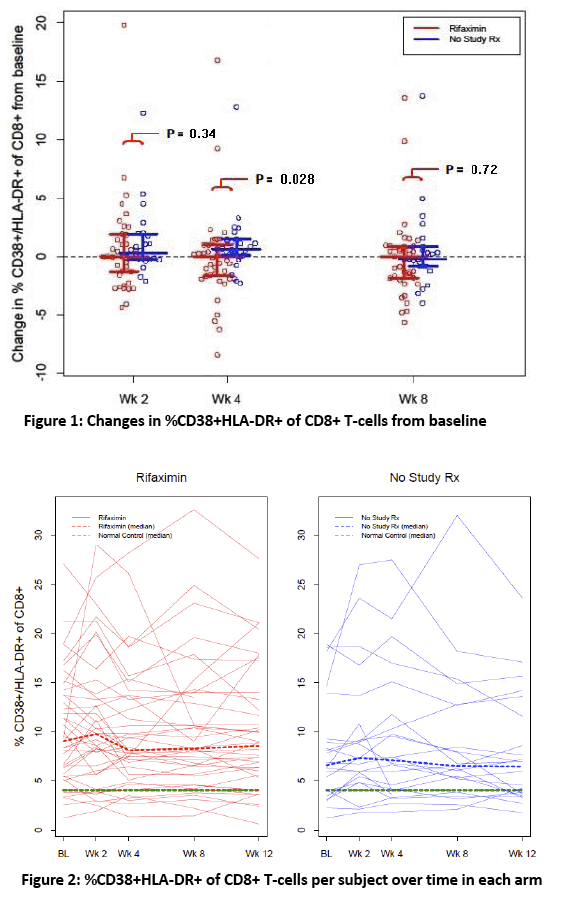
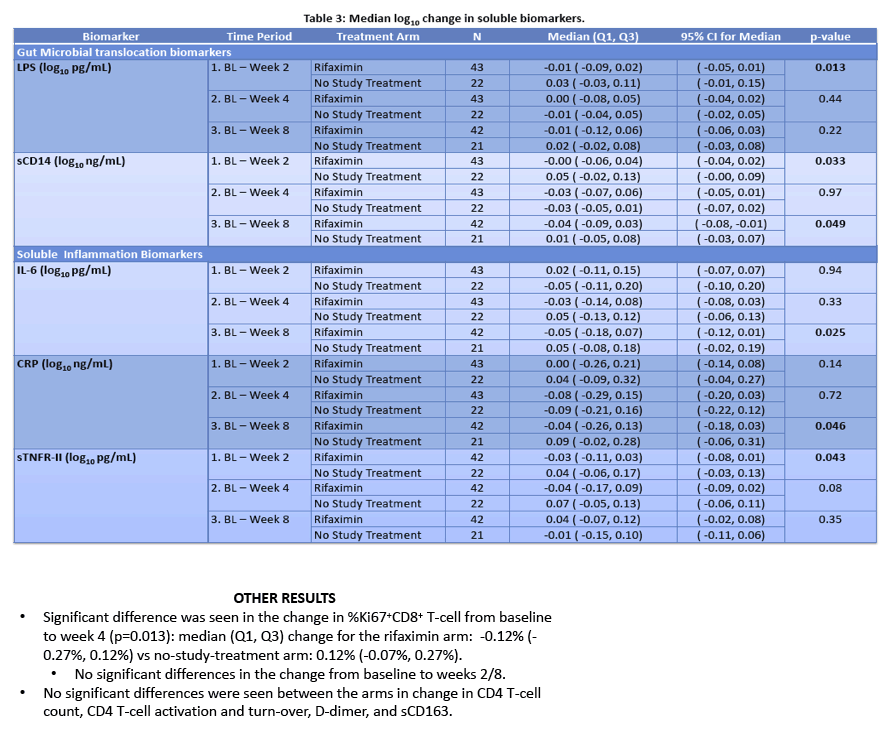
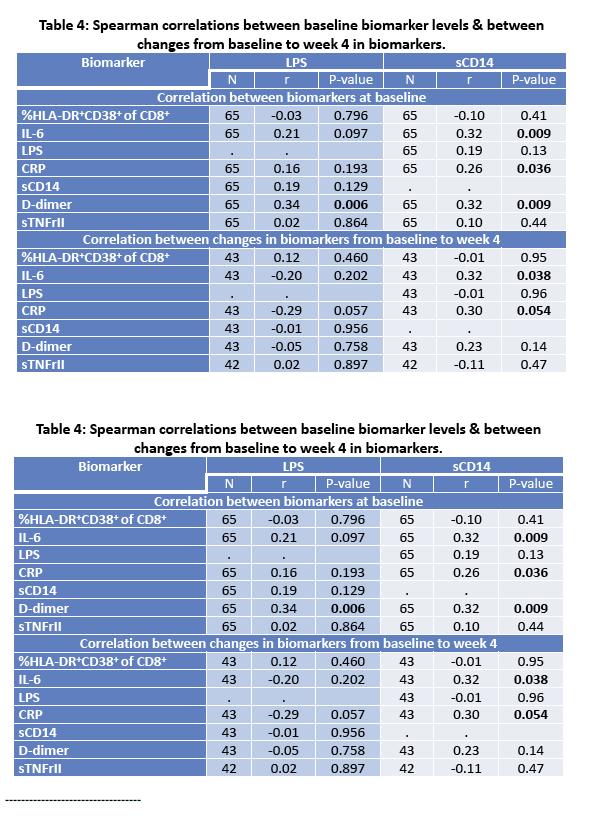
· Rifaximin is a nonabsorbable antibiotic that alters gastrointestinal microbiome and has potential to reduce bacterial translocation, as seen in its ability to reduce LPS levels in patients with cirrhosis. ACTG study A5286 randomized patient with virologic suppression and CD4 cells <350 cells/uL to Rifaximin 550 mg twice daily versus no therapy. After 4 weeks of treatment there was a marginal but significant reduction in CD8 T-cell activation; however, there was no effect on LPS, sCD14 or sCD163 (markers of monocyte activation), IL-6, D-dimer or sTNFr-II levels (27).
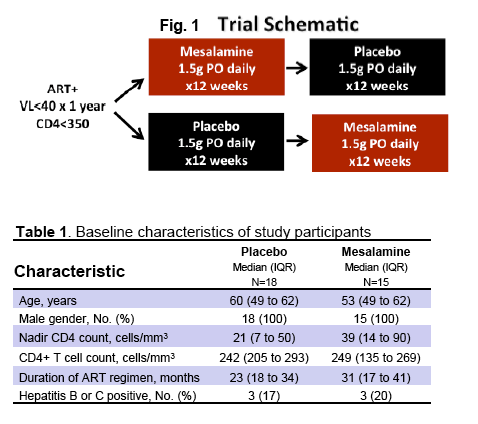
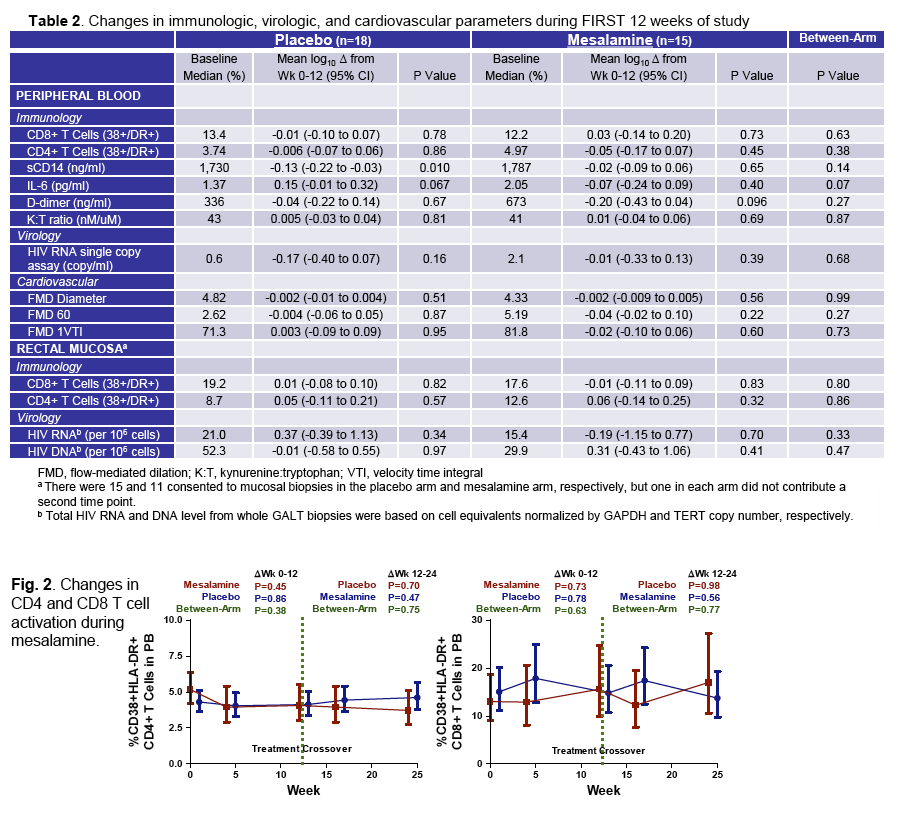
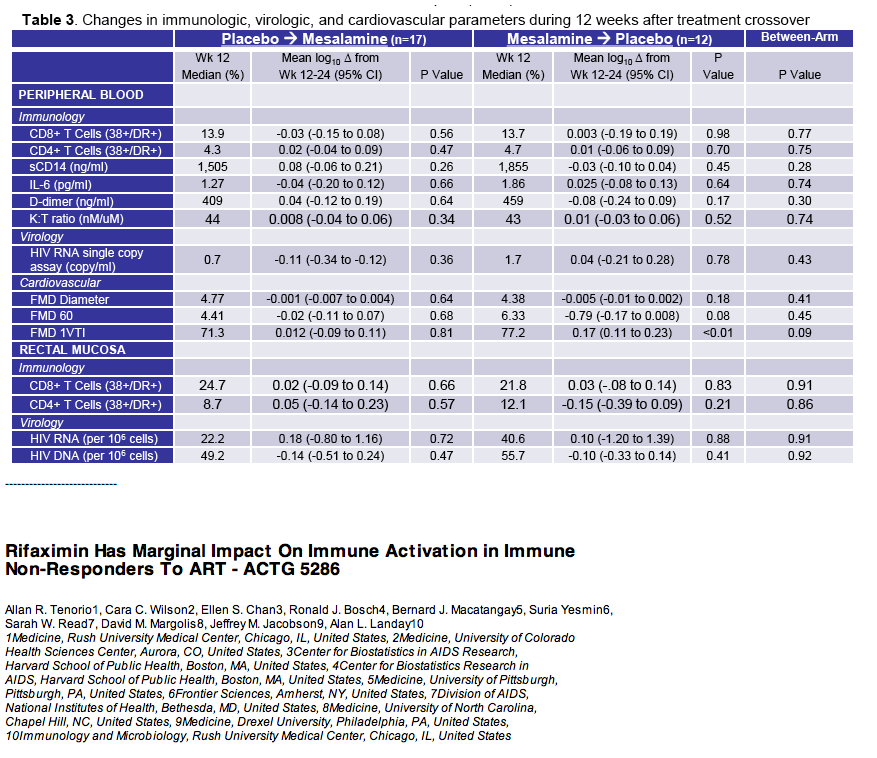
· Mesalamine is a mucosally active anti-inflammatory agent which was used in patients virologically suppressed with CD4 cells <350 cells/uL in a crossover design where 15 received active drug at 1.5 grams daily for 12 weeks and 18 placebo. After 12 weeks patients were crossed-over to the other study arm. Ultimately there was no change in CD4 or CD8 T-cell activation, sCD14, IL-6, D-dimer or effect on flow mediated dilatation, an assessment of endothelial function (28).
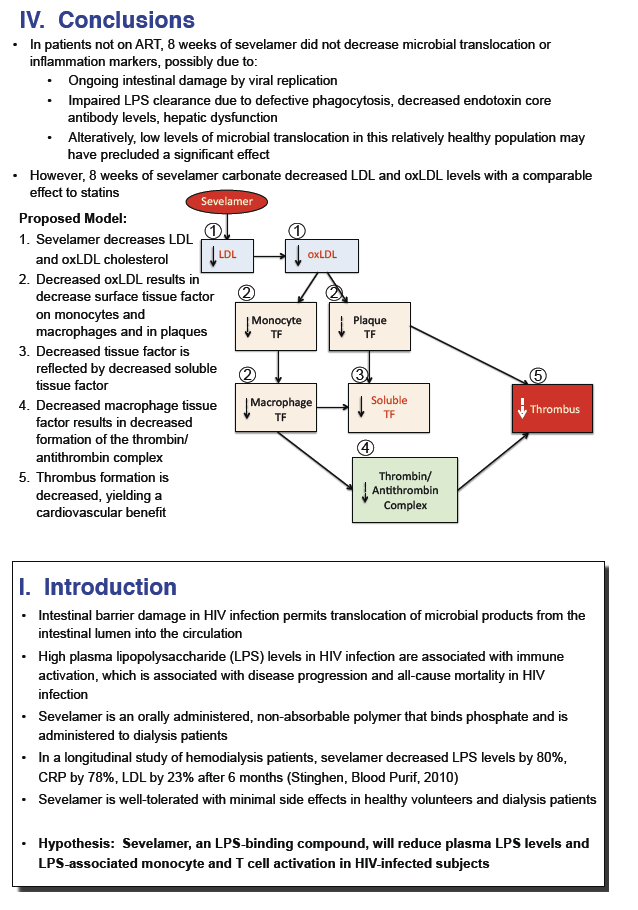
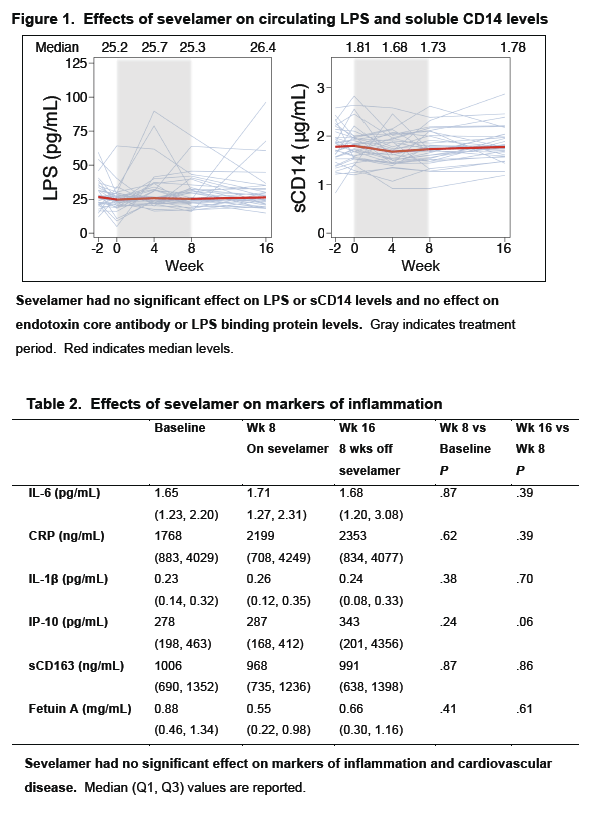
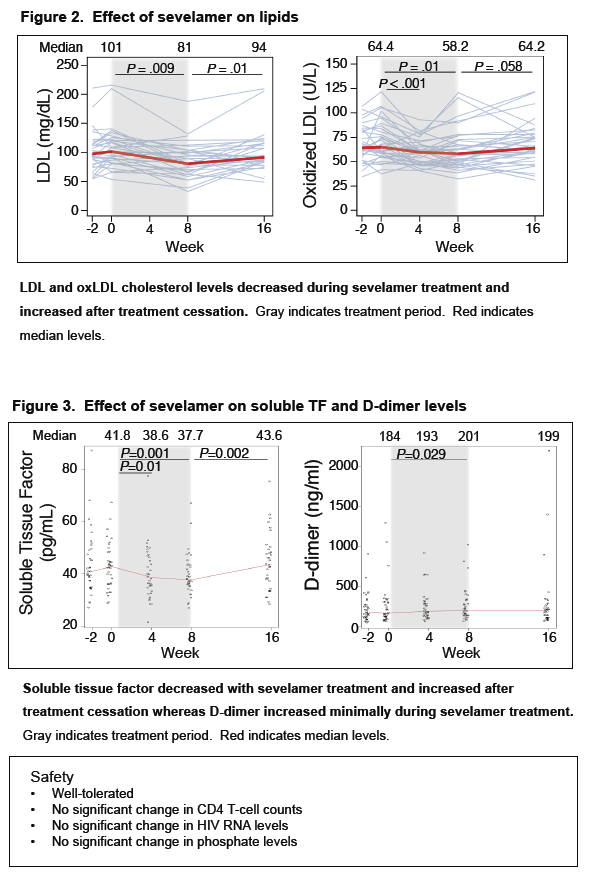
· Sevelamir is a phosphate binder that has been shown to decrease LPS and LDL cholesterol in patients on hemodialysis. A study evaluated whether this agent would reduce LPS levels and T cell activation in untreated HIV-infected patients. It enrolled 40 patients and administered the drug at a dose of 1600 mg three-times daily. After 8 weeks of therapy there was no significant decline in LPS, sCD14, IL-6, CRP or D-dimer, while there was an expected decrease in LDL, oxidated LDL and in Soluble Tissue Factor (29).
· Rosuvastatin is a statin that was studied in the context of the SATURN Study to assess its impact on both BMD, as described above, and systemic inflammation. Virologically suppressed patients with LDL <130 mg/dL and increased levels of CD8 T-cell activation and/or hsCRP were randomized to rosuvastatin 10 mg per day or placebo for 48 weeks. Those receiving active drug demonstrated significant declines in a variety of markers of inflammation/activation, including CD4 and CD8 T-cell activation and monocyte activation (sCD14)(30).
Rosuvastatin Reduces Immune Activation and Inflammation in Treated HIV Infection .......http://www.natap.org/2014/CROI/croi_160.htm
CROI: Rosuvastatin Improves Hip Bone Mineral Density but Worsens Insulin Resistance - (03/07/14)
Interpretation:
· Multiple agents acting at the level of the intestinal lumen showed little effect on systemic inflammation or microbial translocation. This could be explained by these agents having insufficient potency or that gastrointestinal pathology may not be the lone mechanism behind persistent inflammation, activation and coagulation in virologically suppressed HIV-infected individuals.
· Statins do appear to reduce systemic inflammation, the clinically relevance of which needs to be explored in larger clinical trials.
----------------
presented at CROI 2014
Reported by Jules Levin
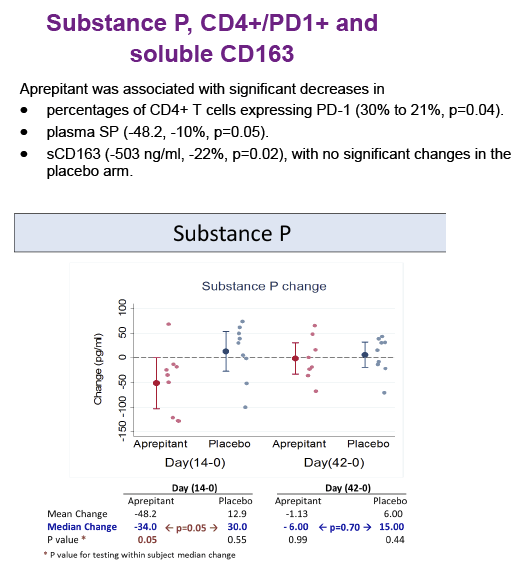
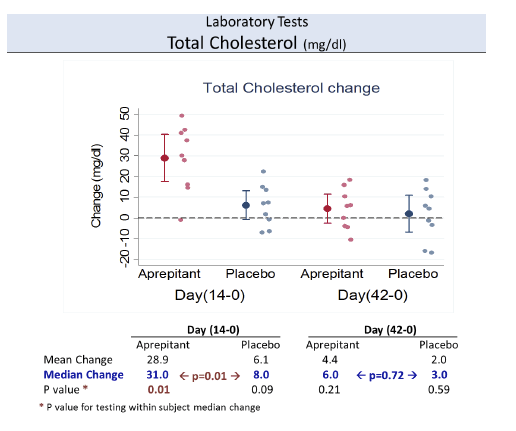
Substance P (SP) is an undecapeptide produced in neurons and immune cells and involved in nociception and inflammation. The effects of
SP are mediated by the neurokinin 1 receptor (NK1R). Increased levels of SP are present in HIV-infected individuals and NK1R antagonists have anti-HIV effects in monocyte-derived macrophages in vitro, by decreasing the expression of CCR5. Aprepitant is an NK1R antagonist approved by FDA as an antiemetic.
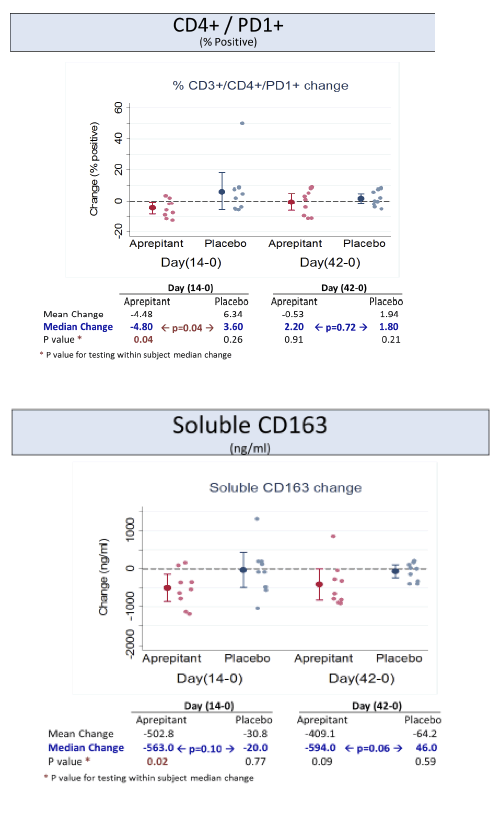
Increased plasma levels of sCD163 are associated with HIV disease progression.
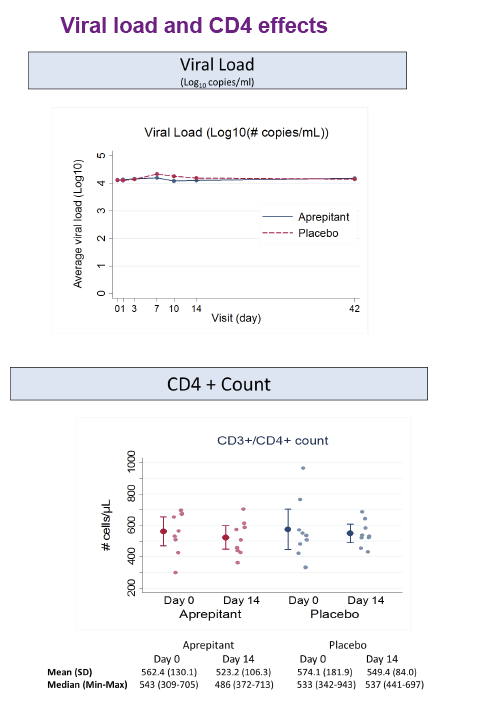
Aprepitant was safe and well tolerated. At the dose used in this proof of concept phase IB study, aprepitant did not show significant antiviral
activity, but its use was associated with decreased levels of CD4+ PD-1 cells, SP and sCD163 suggesting that blockade of the NK1R pathway may have a role in modulating monocyte activation in HIV infection. Prospective studies in virologically suppressed individuals are warranted.
-----------------------
Sevelamer Does Not Decrease Plasma LPS or sCD14 but Does Decrease Soluble Tissue Factor and LDL
Reported by Jules Levin
Netanya Sandler1,2, Xinyan Zhang3, Ronald Bosch3, Nicholas Funderburg4, Janet Robinson4, Daniel Douek1, Cara Wilson5, Steven Deeks6, Michael Lederman4, Rajesh Gandhi7, for the A5296 Study Team
1VRC/NIAID/NIH, Bethesda, MD, USA, 2University of Texas Medical Branch, Galveston, TX, USA, 3Harvard School of Public Health, Statistical & Data Analysis Center, Boston, MA, USA, 4Case Western Reserve University, Cleveland, OH, USA, 5University of Colorado, Aurora, CO, USA, 6University of California, San Francisco, CA, USA, and 7Massachusetts General Hospital, Boston, MA, USA Supported by NIH Grants U01-AI68636, AI68634








|
| |
|
 |
 |
|
|