 |
 |
 |
| |
Three ART Regimens Don't Differ in 144-Week "Target Not Detected" Response
|
| |
| |
Download the PDF here
CROI 2014, March 3-6, 2014, Boston
Mark Mascolini
Regimens containing elvitegravir, efavirenz, or atazanavir/ritonavir did not differ in 144-week viral suppression to a "target-not-detected" (TND) response on the Amplicor assay, according to results of two trials that enrolled antiretroviral-naive people [1]. Regardless of antiretroviral therapy (ART) assignment, higher proportions of people with a pretreatment viral load below 100,000 copies or with at least 95% adherence achieved the TND goal.
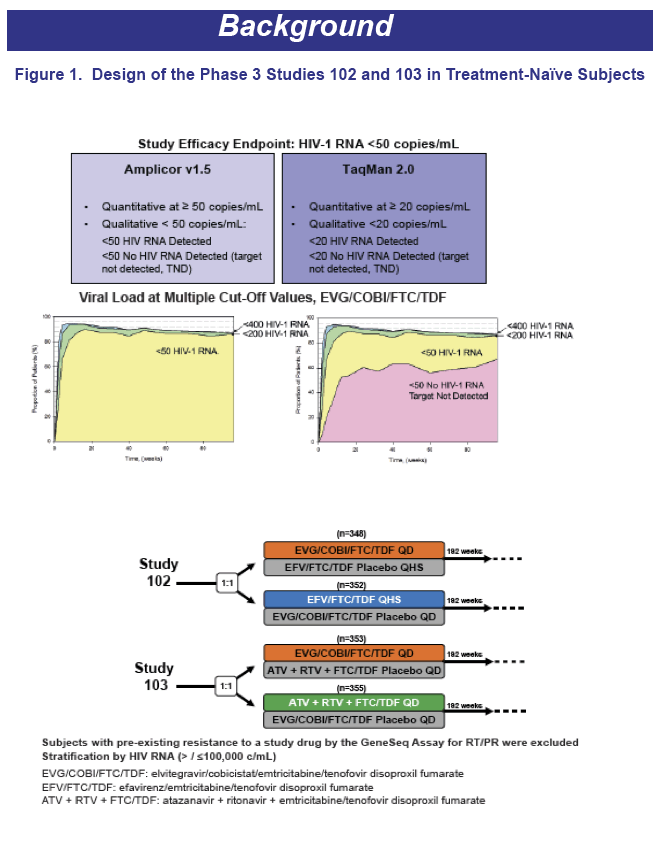
When viral load stands below 50 copies on the Amplicor assay, the result can be (1) HIV RNA detected but not quantified or (2) TND. Gilead Sciences researchers who conducted this analysis observed that some but not all studies found a higher risk of subsequent rebound with a measurable sub-50 viral load than with TND [2,3]. To learn more about how contemporary first-line regimens and baseline traits affect TND response, they analyzed results of two phase 3 double-blind trials that pitted single-tablet elvitegravir, cobicistat, tenofovir (TDF), and emtricitabine (FTC) against efavirenz plus TDF/FTC (study 102) or atazanavir/ritonavir plus TDF/FTC (study 103).
With the standard 50-copy cutoff, 144-week virologic results did not differ significantly between regimens in either of the two trials. When the investigators looked at 144-week TND results, they found that just under two thirds of participants in all treatment arms attained that goal. Reflecting the faster viral suppression achieved with integrase inhibitors than other antiretroviral classes, higher proportions in the elvitegravir arms of both trials reached TND in the first months of treatment, but that difference disappeared by week 24 in study 102 and by week 84 in study 103.
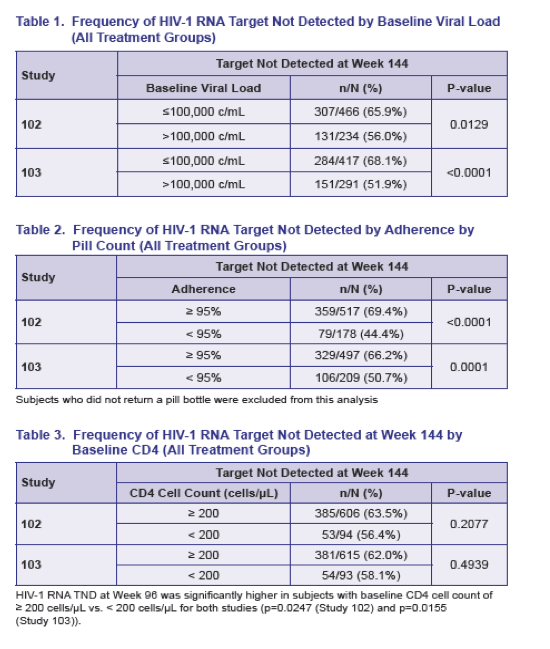
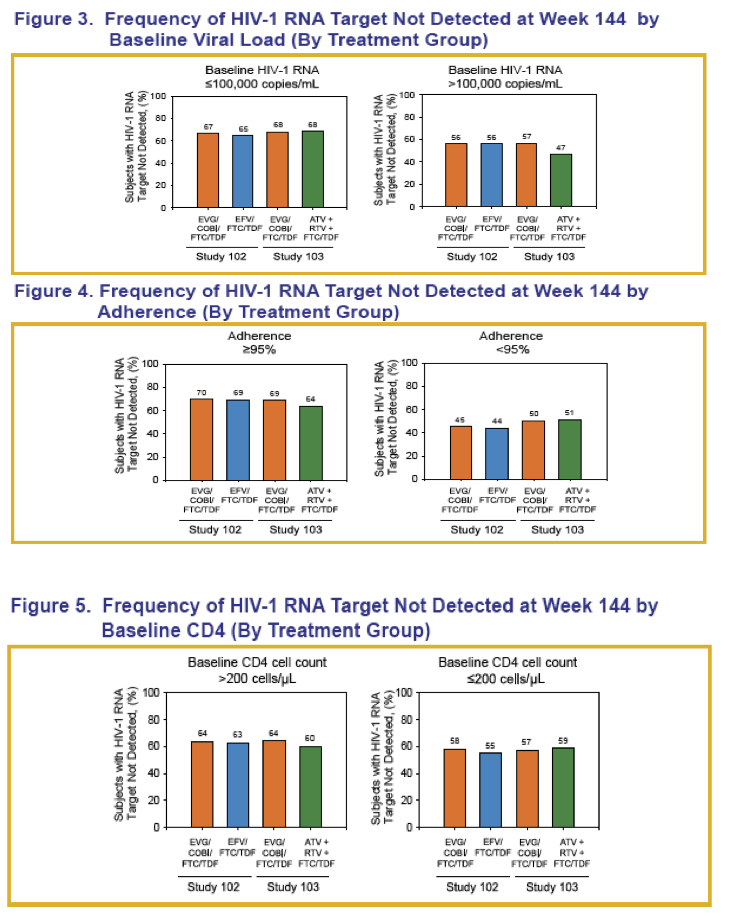
When the Gilead team combined treatment arms in each trial, they found that pretreatment viral load and adherence did affect proportions of participants who achieved TND at week 144. In study 102 they determined that 65.9% with a baseline load at or below 100,000 copies versus 56.0% with a higher baseline load reached TND (P = 0.0129). In study 103 proportions achieving TND were 68.1% in the lower baseline load group and 51.9% in the higher group (P < 0.0001).
In study 102 at week 144, among people with at least 95% adherence by pill count, 69.4% reached TND, compared with 44.4% of people with less adherence (P < 0.0001). Respective proportions with TND in study 103 were 66.2% and 50.7% (P = 0.0001). Pretreatment CD4 count above or below 200 did not affect TND outcomes in either trial.
Among people with a pretreatment load above 100,000 copies, TND response at week 144 differed somewhat between the elvitegravir and atazanavir arms in study 103 (57% versus 47%). No other between-arm comparison showed as much of a TND response difference by baseline viral load, baseline CD4 count, or adherence.
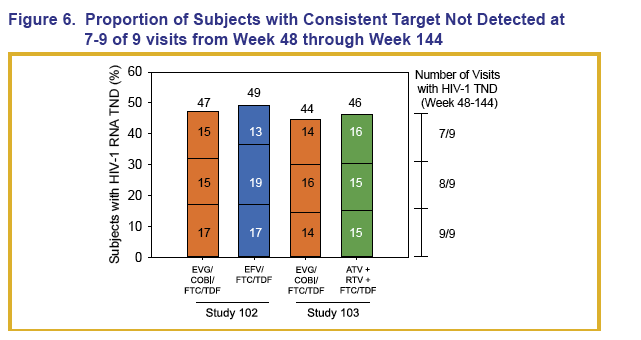
About 15% to 17% of participants in each treatment arm had consistent TND results at 7 of 9, 8 of 9, or 9 of 9 visits between weeks 48 and 144.
The investigators suggested that the worse TND response in people with a pretreatment viral load above 100,000 copies supports guidelines recommending early ART to reach the lowest viral load possible.
References
1. White KL, Wei X, Zhong L, et al. Efficacy of first-line ARV regimens: an exploratory "target not detected" analysis. CROI 2014. Conference on Retroviruses and Opportunistic Infections. March 3-6, 2014. Boston. Abstract 546.
2. Henrich TJ, Wood BR, Kuritzkes DR. Increased risk of virologic rebound in patients on antiviral therapy with a detectable HIV load <48 copies/mL. PLoS One. 2012;7:e50065.
3. Doyle T, Smith C, Vitiello P, et al. Plasma HIV-1 RNA detection below 50 copies/mL and risk of virologic rebound in patients receiving highly active antiretroviral therapy. Clin Infect Dis. 2012;54:724-732.




Increased Risk of Virologic Rebound in Patients on Antiviral Therapy with a Detectable HIV Load <48 Copies/mL......http://www.plosone.org/article/info:doi/10.1371/journal.pone.0050065
(pdf attached to this report) - Plasma HIV-1 RNA Detection Below 50 Copies/mL and Risk of Virologic Rebound in Patients Receiving Highly Active Antiretroviral Therapy, CID 2012 Doyle et al

|
| |
|
 |
 |
|
|