 |
 |
 |
| |
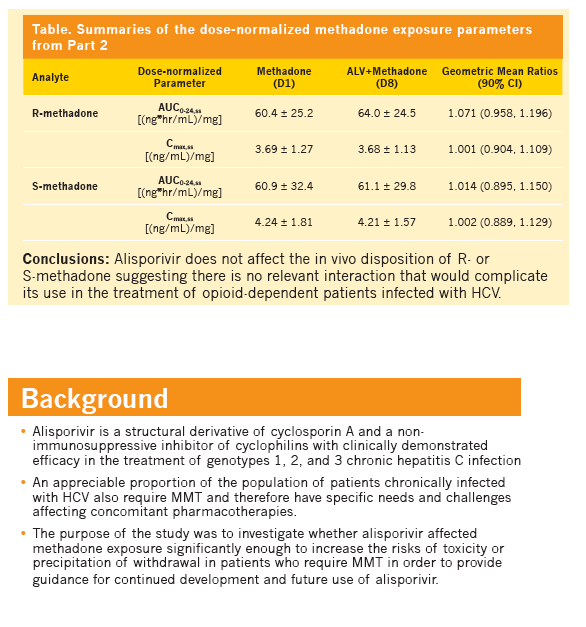
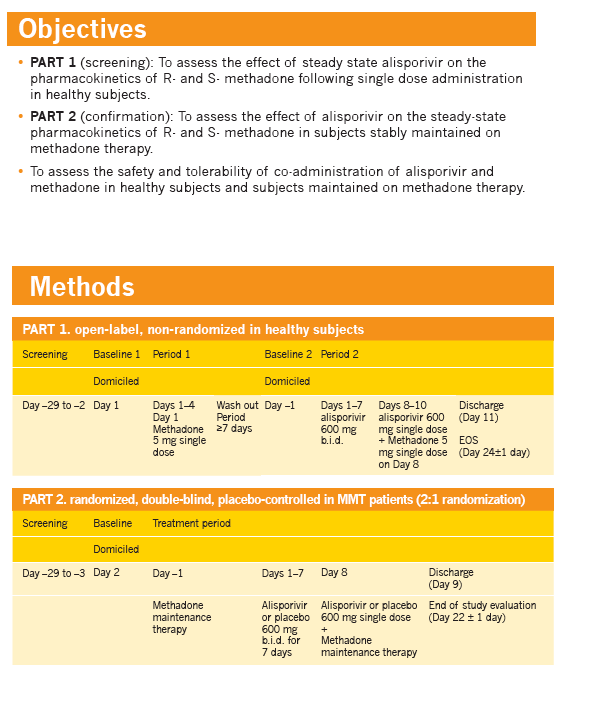
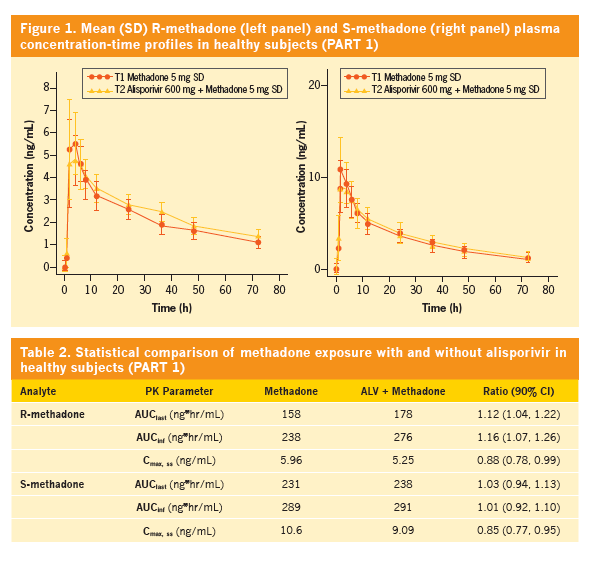
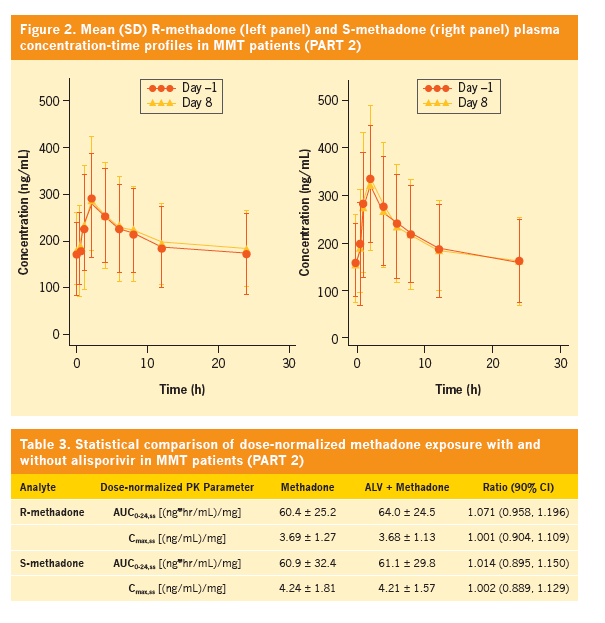
Effect of alisporivir (ALV) on the pharmacokinetics (PK), safety, and tolerability of methadone in healthy subjects and opioid-dependent patients on stable methadone maintenance therapy (MMT)
|
| |
| |
Reported by Jules Levin
EASL 2014 April 9-13 London, UK
STEVEN J. KOVACS*1, JUNE KE1, AVANTIKA BARVE1, LYNN R. WEBSTER2, YI CHENG3, JIE ZHANG1, ROBERT MAIETTA4, GANGADHAR SUNKARA1, DANIEL S. STEIN1
NOVARTIS INSTITUTES FOR BIOMEDICAL RESEARCH (NIBR) 1EAST HANOVER, NJ, USA; 2CRI LIFETREE, SALT LAKE CITY, UT, USA; 3BEIJING NOVARTIS PHARMA CO. LTD, SHANGHAI, CHINA; 4NIBR, CAMBRIDGE, MA, USA
Recruiting. This study is currently recruiting participants. Verified April 2014 by Novartis
Pharmacokinetics, Pharmacodynamics and Safety of DEB025/Alisporivir in Combination With Ribavirin Therapy in Chronic Hepatitis C Genotype 2 and 3 Patients Who Have Previously Failed Interferon Therapy or Are Intolerant or Unable to Take Interferon (ALtitude II)
Recruiting Pharmacokinetics, Pharmacodynamics and Safety of DEB025 Plus Ribavirin in Chronic Hepatitis C Genotype 2 and 3 Treatment naïve Patients



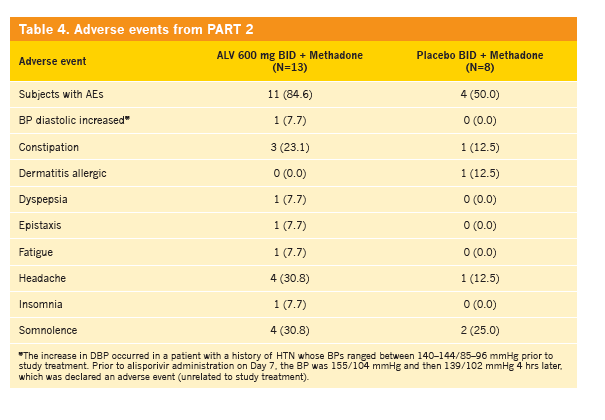
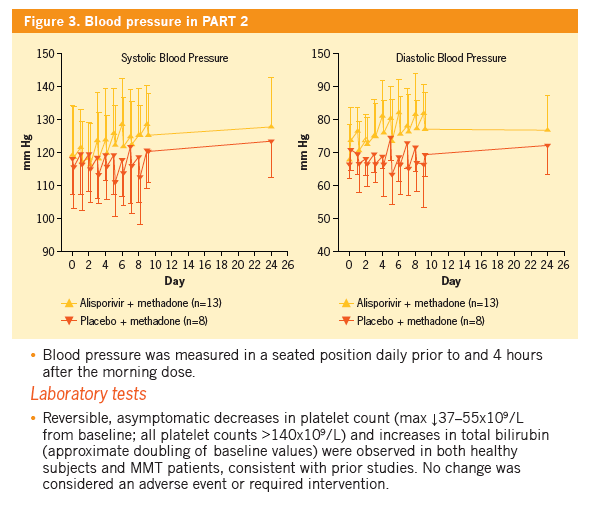
|
| |
|
 |
 |
|
|