 |
 |
 |
| |
Treatment of HCV with ABT-450/r-Ombitasvir and Dasabuvir with Ribavirin (in previously untreated patients with HCV genotype 1 infection and no cirrhosis)
|
| |
| |
Download the PDF here
Download the PDF here
New England Jnl of Medicine April 11 2014
EASL - The International Liver Congress 2014 49th Annual Meeting of the European
Association for the Study of the Liver
London, United Kingdom
April 9-13
Jordan J. Feld, M.D., M.P.H., Kris V. Kowdley, M.D., Eoin Coakley, M.D., Samuel Sigal, M.D., David R. Nelson, M.D., Darrell Crawford, M.D., Ola Weiland, M.D., Humberto Aguilar, M.D., Junyuan Xiong, M.S., Tami Pilot-Matias, Ph.D., Barbara DaSilva-Tillmann, M.D., Lois Larsen, Ph.D., Thomas Podsadecki, M.D., and Barry Bernstein, M.D.
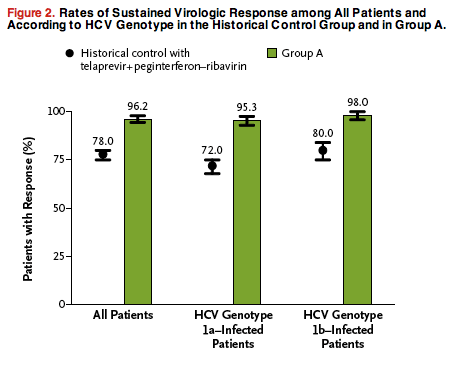
Figure 2. Rates of Sustained Virologic Response among All Patients and
According to HCV Genotype in the Historical Control Group and in Group A.
Background
The interferon-free combination of the protease inhibitor ABT-450 with ritonavir (ABT-450/r) and the NS5A inhibitor ombitasvir (also known as ABT-267) plus the nonnucleoside polymerase inhibitor dasabuvir (also known as ABT-333) and ribavirin has shown efficacy against the hepatitis C virus (HCV) in patients with HCV genotype 1 infection. In this phase 3 trial, we evaluated this regimen in previously untreated patients with HCV genotype 1 infection and no cirrhosis.
Methods
In this multicenter, randomized, double-blind, placebo-controlled trial, we assigned previously untreated patients with HCV genotype 1 infection, in a 3:1 ratio, to an active regimen consisting of a single-tablet coformulation of ABT-450/r-ombitasvir (at a once-daily dose of 150 mg of ABT-450, 100 mg of ritonavir, and 25 mg of ombitasvir), and dasabuvir (250 mg twice daily) with ribavirin (in doses determined according to body weight) (group A) or matching placebos (group B). The patients received the study treatment during a 12-week double-blind period. The primary end point was sustained virologic response at 12 weeks after the end of treatment. The primary analysis compared the response rate in group A with the response rate (78%) in a historical control group of previously untreated patients without cirrhosis who received telaprevir with peginterferon and ribavirin. Adverse events occurring during the double-blind period were compared between group A and group B.
Results
A total of 631 patients received at least one dose of the study drugs. The rate of sustained virologic response in group A was 96.2% (95% confidence interval, 94.5 to 97.9), which was superior to the historical control rate. Virologic failure during treatment and relapse after treatment occurred in 0.2% and 1.5%, respectively, of the patients in group A. The response rates in group A were 95.3% among patients with HCV genotype 1a infection and 98.0% among those with HCV genotype 1b infection. The rate of discontinuation due to adverse events was 0.6% in each study group. Nausea, pruritus, insomnia, diarrhea, and asthenia occurred in significantly more patients in group A than in group B (P<0.05 for all comparisons). Reductions in the hemoglobin level were all of grade 1 or 2; reductions of grade 1 and 2 occurred in 47.5% and 5.8%, respectively, of the patients in group A, whereas grade 1 reductions occurred in 2.5% of the patients in group B.
Conclusions
In previously untreated patients with HCV genotype 1 infection and no cirrhosis, a 12-week multitargeted regimen of ABT-450/r-ombitasvir and dasabuvir with ribavirin was highly effective and was associated with a low rate of treatment discontinuation. (Funded by AbbVie; SAPPHIRE-I ClinicalTrials.gov number, NCT01716585.)
Approximately 184 million people worldwide have chronic hepatitis C virus (HCV) infection, and more than 350,000 people die of HCV-related liver disease each year.1,2 Until recently, the standard of care for chronic HCV genotype 1 infection was a first-generation protease inhibitor, telaprevir or boceprevir, with peginterferon and ribavirin; this therapy resulted in rates of sustained virologic response of 67 to 75% among previously untreated patients.3,4 The new standard of care is peginterferon and ribavirin combined with either the nucleotide nonstructural (NS) 5B polymerase inhibitor sofosbuvir or the protease inhibitor simeprevir.5 Peginterferon-based treatment is associated with clinically significant systemic events, including influenza-like symptoms and depression. Variables including patients' age, race, HCV genotype, IL28B genotype, HCV viral load at baseline, and degree of liver fibrosis have been shown to affect the response to peginterferon-based therapy and other therapies in development.3,4,6-9
ABT-450 is an HCV NS3/4A protease inhibitor with nanomolar potency in vitro. ABT-450 is administered with low-dose ritonavir, an inhibitor of the cytochrome P-450 enzyme CYP3A4. As a pharmacologic enhancer of ABT-450, ritonavir facilitates once-daily dosing, and administration of ABT-450 with ritonavir (ABT-450/r) results in higher drug exposures than administration of ABT-450 alone.10 Ombitasvir (also known as ABT-267) is an HCV NS5A inhibitor with picomolar potency in vitro. Dasabuvir (also known as ABT-333) is a nonnucleoside NS5B polymerase inhibitor with nanomolar potency in vitro.
In a phase 2b trial involving previously untreated patients with HCV genotype 1 infection, 96% of the patients who received ABT-450/r, ombitasvir, dasabuvir, and ribavirin for 12 weeks had a sustained virologic response at 24 weeks after the end of treatment,11 suggesting that a multitargeted approach may maximize the response rate, such that most patients would have a sustained virologic response without the development of drug resistance. Here we report the results of the phase 3, international, multicenter, randomized, double-blind, placebo-controlled SAPPHIRE-I trial evaluating the safety and efficacy of 12 weeks of an all-oral regimen of ABT-450/r-ombitasvir and dasabuvir with ribavirin in previously untreated patients with HCV genotype 1 infection and no cirrhosis.
Methods
Patients
Patients were screened from November 2012 through May 2013 at 79 sites in North America, Europe, and Australia. Eligible patients were adults, 18 to 70 years of age, with chronic HCV genotype 1 infection, no cirrhosis, and a plasma HCV RNA level of more than 10,000 IU per milliliter, who had never received antiviral treatment for HCV infection. Patients were excluded if they had a positive test for hepatitis B surface antigen or anti-human immunodeficiency virus antibody during screening. Details of the eligibility criteria are provided in the Supplementary Appendix, available with the full text of this article at NEJM.org.
Study Design
Patients were randomly assigned, in a 3:1 ratio, to active treatment (group A) or placebo (group B) (Figure 1). Randomization was stratified according to HCV genotype (1a vs. non-1a) and IL28B genotype (CC vs. non-CC). During the double-blind period, patients in group A received 12 weeks of treatment with oral coformulated ABT-450/r-ombitasvir (at a once-daily dose of 150 mg of ABT-450, 100 mg of ritonavir, and 25 mg of ombitasvir) and dasabuvir (at a dose of 250 mg twice daily) with ribavirin, administered twice daily in a dose that was determined according to body weight (1000 mg daily if the body weight was <75 kg and 1200 mg daily if the body weight was ≥75 kg). Patients in group B received matching placebos during the double-blind period. After the double-blind period, patients in group B received the active regimen as open-label therapy for 12 weeks.
The investigators, patients, and sponsor were unaware of the treatment assignments during the double-blind period. To prevent implicit unblinding, laboratory results for HCV RNA, hemoglobin, hematocrit, alanine aminotransferase, aspartate aminotransferase, and bilirubin (indirect and total) levels were concealed from these parties. Details are provided in the Supplementary Appendix. The study is ongoing, and all the patients will be followed for 48 weeks after the end of active treatment. Efficacy results in group A through post-treatment week 12 and safety data for both study groups are presented.
Study Oversight
All the patients provided written informed consent. The study was conducted in accordance with the International Conference on Harmonisation guidelines, applicable regulations, and the principles of the Declaration of Helsinki. The study protocol was approved by the independent ethics committee or institutional review board at each study site. The study was designed jointly by the study investigators and the sponsor (AbbVie). The investigators gathered the data, and the sponsor conducted the data analyses. All the authors had full access to the data and signed confidentiality agreements with the sponsor regarding the data. The first draft of the manuscript was written by a medical writer who is an employee of the sponsor, with input from all the authors. All the authors reviewed and provided feedback on all subsequent versions of the manuscript and made the decision to submit the manuscript for publication. All the authors vouch for the completeness and accuracy of the data and analyses presented and affirm that the study was conducted and reported with fidelity to the protocol (available at NEJM.org).
Efficacy Assessments
A central laboratory determined the HCV RNA level with the use of the COBAS TaqMan real-time reverse-transcriptase-polymerase-chain-reaction assay, version 2.0 (Roche). The lower limits of detection and quantification were 15 IU per milliliter and 25 IU per milliliter, respectively. Details regarding the collection of plasma samples, the criteria for virologic failure, and resistance testing are provided in the Supplementary Appendix.
Safety Assessments
Adverse events were assessed at each study visit. The investigator at each site classified events as mild, moderate, or severe. Data on all adverse events were collected from the start of study-drug administration until 30 days after receipt of the last dose. Data on serious adverse events were collected throughout the entire study period. Here we report data on adverse events and serious adverse events occurring during the double-blind period and the 30-day period after the last dose of active study drugs was administered. Clinical laboratory testing was performed at visits during the double-blind treatment period and at weeks 4 and 48 after the end of the treatment period.
Efficacy End Points
The primary efficacy end point was sustained virologic response (HCV RNA level <25 IU per milliliter) at 12 weeks after the end of treatment. Secondary efficacy end points included normalization of the alanine aminotransferase level, sustained virologic response at post-treatment week 12 according to HCV genotype (1a or 1b), virologic failure during treatment, and post-treatment relapse. The primary analysis was performed after all the patients in group A reached post-treatment week 12 and all those in group B reached week 12 of open-label treatment (Figure 1). Primary analysis data are reported.
Normalization of the alanine aminotransferase level was defined as a final level that was no more than the upper limit of the normal range during the double-blind treatment period in patients whose baseline level had been higher than the upper limit of the normal range. Patients were considered to have virologic failure during treatment if they had a confirmed HCV RNA level of 25 IU per milliliter or more after the HCV RNA level had been lower than 25 IU per milliliter during treatment, if they had a confirmed increase in the HCV RNA level of more than 1 log10 IU per milliliter above the nadir observed during treatment, or if all the HCV RNA values during the treatment with the study drug administered for at least 6 weeks were 25 IU per milliliter or more. Virologic relapse was defined as a confirmed HCV RNA level of 25 IU per milliliter or more between the final treatment visit and 12 weeks after receipt of the last dose of study drug among patients who completed treatment (duration of treatment, ≥77 days), who had an HCV RNA level that was lower than 25 IU per milliliter at the final treatment visit during the double-blind period, and who had HCV RNA data available after treatment.
Statistical Analysis
Analyses were performed in the modified intention-to-treat population, which included all the patients who underwent randomization and received at least one dose of the study drug during the double-blind period. The primary efficacy analyses assessed noninferiority and superiority with respect to the rate of sustained virologic response at post-treatment week 12 associated with the active regimen (ABT-450/r-ombitasvir and dasabuvir with ribavirin) by comparing it with a calculated historical control rate of 78% (95% confidence interval [CI], 75 to 80). This control rate was based on response rates among previously untreated patients without cirrhosis who received telaprevir and peginterferon-ribavirin.12
To establish that the rate of sustained virologic response at post-treatment week 12 associated with the active regimen was noninferior to the historical control rate, the lower boundary of the 95% confidence interval for the rate of sustained virologic response at post-treatment week 12 in group A had to exceed the upper boundary of the 95% confidence interval for the control rate minus 10.5 percentage points (70%). To establish that the rate of sustained virologic response at post-treatment week 12 associated with the active regimen was superior to the historical control rate, the lower boundary of the 95% confidence interval for the rate in group A had to exceed the upper boundary of the confidence interval for the historical rate (80%).
The two-sided 95% confidence intervals were calculated with the use of the normal approximation to the binomial distribution. We calculated that a sample of 600 patients (450 patients in group A) would provide the study with more than 90% power to show noninferiority and superiority of the active regimen, assuming a rate of sustained virologic response at post-treatment week 12 of 92%.
Secondary analyses assessed whether the rates of sustained virologic response at post-treatment week 12 in HCV genotype 1a-infected and HCV genotype 1b-infected subgroups of group A were superior to calculated rates for these subgroups in the historical control group (72% [95% CI, 68 to 75] in patients with HCV genotype 1a infection and 80% [95% CI, 75 to 84] in those with HCV genotype 1b infection).12 If the lower boundary of the 95% confidence interval for the rate of sustained virologic response at post-treatment week 12 exceeded 75% among patients with HCV genotype 1a infection or 84% among those with HCV genotype 1b infection, the rate with the active regimen was considered to be superior to the historical control rate in that subgroup. Details of the noninferiority and superiority analyses and sample-size determination are provided in the Supplementary Appendix. A fixed-sequence testing procedure was used to maintain the type I error rate at a level of 0.05 as the primary and secondary efficacy end points were analyzed in a specific order (see the Supplementary Appendix).
SAS software, version 9.3, for the UNIX operating system (SAS Institute) was used for all analyses. All statistical tests and 95% confidence intervals were two-sided, with a significance level of 0.05. Differences in baseline characteristics between the treatment groups were evaluated with the use of the chi-square test for categorical data and one-way analysis of variance for continuous data. Comparisons of rates of normalization of the alanine aminotransferase level, adverse events, and laboratory abnormalities were performed with the use of Fisher's exact test.
The relationship between prespecified baseline characteristics (e.g., fibrosis score) and the rate of sustained virologic response at post-treatment week 12 was analyzed by means of stepwise logistic regression to determine independent predictors of sustained virologic response at post-treatment week 12. The fibrosis score (on a scale from F0, indicating no fibrosis, to F4, indicating cirrhosis) was determined by means of liver biopsy (Metavir, Batts-Ludwig, Knodell, International Association for the Study of the Liver, Scheuer, Laennec, or Ishak scoring system), FibroTest, or FibroScan (Echosens). Additional details of the stepwise logistic-regression analysis and fibrosis scoring are provided in Table S2 in the Supplementary Appendix.
Results
Patients
A total of 855 patients were screened, 636 underwent randomization, and 631 (473 patients in group A and 158 in group B) received at least one dose of study drug (Figure S1 in the Supplementary Appendix). The final date for the collection of data to be used in the analysis of the rate of sustained virologic response at post-treatment week 12 was November 4, 2013. The characteristics of the patients at baseline are shown in Table 1.
Efficacy
By week 4 of the double-blind treatment period, the HCV RNA level was below 25 IU per milliliter in 99.4% of the patients who were receiving the active regimen (461 of the 464 patients in group A with data available) (Figure S2 in the Supplementary Appendix). In the modified intention-to-treat analysis, the rate of sustained virologic response at post-treatment week 12 was 96.2% (95% CI, 94.5 to 97.9) in group A (455 of 473 patients) (Figure 2). This rate was noninferior and superior to the historical control rate with telaprevir plus peginterferon-ribavirin.
The rate of sustained virologic response at post-treatment week 12 was 95.3% (95% CI, 93.0 to 97.6) among patients with HCV genotype 1a infection (307 of 322 patients) and 98.0% (95% CI, 95.8 to 100) among those with HCV genotype 1b infection (148 of 151). These rates were superior to the historical control rates in the respective subgroups (Figure 2).
Figure 3 shows the rates of sustained virologic response at post-treatment week 12 in additional patient subgroups. The rates were similarly high in all subgroups, including those defined by IL28B genotype (96.5% with CC and 96.0% with non-CC), race (96.4% among black patients and 96.2% among nonblack patients), fibrosis score at baseline (97.0% with a score of F0 or F1, 94.3% with a score of F2, and 92.5% with a score of F3), and baseline HCV RNA level (98.1% with a level <800,000 IU per milliliter and 95.7% with a level ≥800,000 IU per milliliter). The rate of sustained virologic response at post-treatment week 12 was 93.5% among patients who had a modification of the ribavirin dose (29 of 31 patients) and 96.4% among those who did not have a dose modification (426 of 442).
Stepwise logistic-regression analysis showed that characteristics including age, sex, race, ethnic group, IL28B genotype, fibrosis score, and baseline viral load did not have a significant effect on the sustained virologic response at post-treatment week 12. Only high baseline body-mass index (the weight in kilograms divided by the square of the height in meters) was associated with a reduced rate, although the effect was modest (odds ratio, 0.89; P=0.02); even among patients with a body-mass index of 30 or more, the rate of sustained virologic response at post-treatment week 12 was high (91.5%) (Table S4 in the Supplementary Appendix).
Patients in group A took an average of 98.9% or more of their prescribed doses of each pill. The rate of normalization of the alanine aminotransferase level was 97.0% in group A (352 of 363 patients), as compared with 14.9% in group B (17 of 114) (P<0.001).
Virologic Failure during Treatment and Relapse
Among the 473 patients in group A, 1 patient (0.2%) had virologic failure during the double-blind treatment period. In this HCV genotype 1a-infected patient who had adhered to the treatment regimen, the HCV RNA level became quantifiable at treatment week 12. A total of 7 of 463 patients (1.5%) had a relapse by post-treatment week 12, of whom 5 had a relapse at or before the visit at post-treatment week 4.
Each of the eight patients who had virologic failure during treatment or relapse had at least one amino acid variant that was known to confer resistance to one of the three direct-acting antiviral agents included in the regimen. The most frequently detected variants in the seven patients with HCV genotype 1a infection at the time of virologic failure during treatment or relapse were D168V (in six patients) in the protein NS3, M28T (in two) and Q30R (in three) in the protein NS5A, and S556G (in three) in the protein NS5B. The single patient with HCV genotype 1b infection who had relapse had variants Y56H and D168V in the protein NS3, L31M and Y93H in the protein NS5A, and S556G in the protein NS5B at the time of relapse.
Safety
During the double-blind period, 87.5% of the patients who were receiving the active regimen (group A), as compared with 73.4% of those receiving placebo (group B), had an adverse event (P<0.001) (Table 2). Events were mild in most patients. The most common adverse events were fatigue (in 34.7% of the patients in group A and 28.5% of those in group B) and headache (in 33.0% of the patients in group A and 26.6% of those in group B); the frequency of these events did not differ significantly between the study groups (P>0.05 for both comparisons). Among adverse events that occurred in more than 10% of patients in either group, five occurred in significantly more patients in group A than in group B: nausea (23.7% vs. 13.3%), pruritus (16.9% vs. 3.8%), insomnia (14.0% vs. 7.6%), diarrhea (13.7% vs. 7.0%), and asthenia (12.1% vs. 3.8%) (P<0.05 for each comparison).
Discontinuation due to an adverse event occurred in 0.6% of the patients in each group (3 patients in group A and 1 in group B). Of the 3 patients in group A who discontinued owing to an adverse event, 2 did not have a sustained virologic response at post-treatment week 12, and 1, a patient who discontinued the study drug during week 11, had a sustained virologic response at post-treatment week 12. Serious adverse events occurred in 10 patients in group A (2.1%) (Table 2, and Table S6 in the Supplementary Appendix). No patient in group B had a serious adverse event during the double-blind period.
Clinically significant chemical and hematologic abnormalities of grade 3 and 4 that occurred during the double-blind period are shown in Table 2. The most frequent chemical abnormality of grade 3 or 4 in group A was a grade 3 elevated total bilirubin level (in 13 of 469 patients [2.8%]); no patient had an elevation of grade 4. Elevations in the bilirubin level involved predominantly indirect bilirubin. In 9 of these 13 patients, the elevation was observed at a single visit. A total of 2 of these 13 patients had clinically recognized jaundice. All elevated bilirubin levels improved or resolved without discontinuation of the study drug.
In group A, 4 of 469 patients (0.9%) had an elevated alanine aminotransferase level of grade 3 or 4. In these 4 patients, the alanine aminotransferase level declined with ongoing treatment and was either within the normal range or at grade 1 at the first post-treatment visit. One of these 4 patients had an associated grade 2 elevation in the total bilirubin level (predominantly indirect bilirubin), which declined to within the normal range at the first post-treatment visit. All these patients had a sustained virologic response at post-treatment week 12.
Grade 1 reductions in the hemoglobin level (ranging from below the lower limit of the normal range to 10.0 g per deciliter) occurred in 47.5% of patients in group A and 2.5% of those in group B. Grade 2 reductions (<10.0 to 8.0 g per deciliter) occurred in 5.8% of patients in group A and in no patients in group B. There were no hemoglobin reductions of grade 3 or 4 (<8.0 g per deciliter) in either study group (Table S7 in the Supplementary Appendix). The ribavirin dose was modified owing to adverse events in 5.5% of the patients in group A. No patient discontinued the study treatment owing to anemia. One patient received erythropoietin. No patient received a transfusion.
Discussion
This large, international, phase 3 trial showed the efficacy of an interferon-free, all-oral antiviral therapy for previously untreated patients with HCV genotype 1 infection and no cirrhosis. In the modified intention-to-treat analysis, the rate of sustained virologic response at post-treatment week 12 among patients who received 12 weeks of ABT-450/r-ombitasvir and dasabuvir with ribavirin was 96.2%, a rate that was noninferior and superior to the historical control rate with telaprevir and peginterferon-ribavirin. This multitargeted regimen resulted in a low rate of virologic failure (virologic failure occurred during treatment in 0.2% of the patients, and relapse after the end of treatment in 1.5%), which limited the number of patients in whom drug resistance developed.
The rate of sustained virologic response at post-treatment week 12 observed in our study compares favorably with the rates of 67 to 89% reported in phase 3 trials involving previously untreated patients with HCV genotype 1 infection who received peginterferon and ribavirin with a direct-acting antiviral agent, such as telaprevir, boceprevir, simeprevir, or sofosbuvir.3,4,6,7 Findings from small, phase 2 studies suggest that peginterferon-free regimens of direct-acting antiviral agents with or without ribavirin may be associated with high rates of sustained virologic response at post-treatment week 12.9,11,13-17 The rate observed here is consistent with results of a phase 2b trial in which treatment for 12 weeks with ABT-450/r, ombitasvir, dasabuvir, and ribavirin was associated with a sustained virologic response at post-treatment week 24 in 96% of previously untreated patients with HCV genotype 1 infection.11
The efficacy of different treatment regimens with antiviral agents may vary according to HCV genotype (1a or 1b).6,7,9,18 In this trial, the rates of sustained virologic response at post-treatment week 12 were similar among patients with HCV genotype 1a infection and those with HCV genotype 1b infection (95.3% and 98.0%, respectively). Previous reports have indicated reduced response rates with peginterferon-containing therapies among patients with certain characteristics, including IL28B non-CC genotype, black race, and high viral load.3,4 In this trial, the rates were high across these subgroups.
The double-blind, placebo-controlled study design allowed the comparison of adverse events between patients receiving the active regimen and those receiving placebo. Fatigue and headache were the most common adverse events, but their frequency did not differ significantly between the study groups. The most common adverse events that occurred significantly more frequently among patients receiving the active regimen were nausea, pruritus, insomnia, diarrhea, and asthenia. The rate of discontinuation due to adverse events was 0.6% in each study group. The rate of serious adverse events was low, with such events occurring in 2.1% of the patients who received the active regimen (10 patients).
The adverse-event profile for the active regimen compares favorably with that for a protease inhibitor plus peginterferon-ribavirin. Rates of 9 to 12% for serious adverse events and rates of 10 to 16% for discontinuation due to adverse events have been reported among previously untreated patients receiving telaprevir or boceprevir with peginterferon-ribavirin.3,4 Clinically significant anemia leading to erythropoietin use, reduction of the ribavirin dose, and occasionally blood transfusions have also been reported.19 In our study, among patients receiving the active regimen during the double-blind period, reductions in the hemoglobin level of grade 1 were common (in 47.5% of patients), hemoglobin reductions of grade 2 were uncommon (in 5.8%), and there were no hemoglobin reductions of grade 3 or 4.
Modifications of the ribavirin dose due to adverse events were relatively uncommon (in 26 patients [5.5%]), and no patient discontinued the study drug owing to anemia. An elevated total bilirubin level was the most frequent laboratory abnormality of grade 3 or 4. The elevations were typically transient and were infrequently associated with jaundice. The elevations in the bilirubin level involved predominantly indirect bilirubin and were consistent with the known inhibitory effect of ABT-450 on the bilirubin transporters OATP1B1 and OATP1B320,21 and the known role of ribavirin in hemolysis.
We excluded from our trial patients with cirrhosis and patients who were using medications contraindicated with ritonavir and ribavirin. Although this study did not include patients who had received prior treatment, Zeuzem et al. now report in the Journal that a study evaluating the same active regimen in patients who had received prior treatment with peginterferon-ribavirin has shown high rates of sustained virologic response at post-treatment week 12.22
In conclusion, a multitargeted approach combining the direct-acting antiviral agents ABT-450/r-ombitasvir and dasabuvir with ribavirin was associated with a high rate of sustained virologic response at post-treatment week 12, with a low rate of treatment discontinuation, among previously untreated patients with HCV genotype 1 infection and no cirrhosis.
|
| |
|
 |
 |
|
|