 |
 |
 |
| |
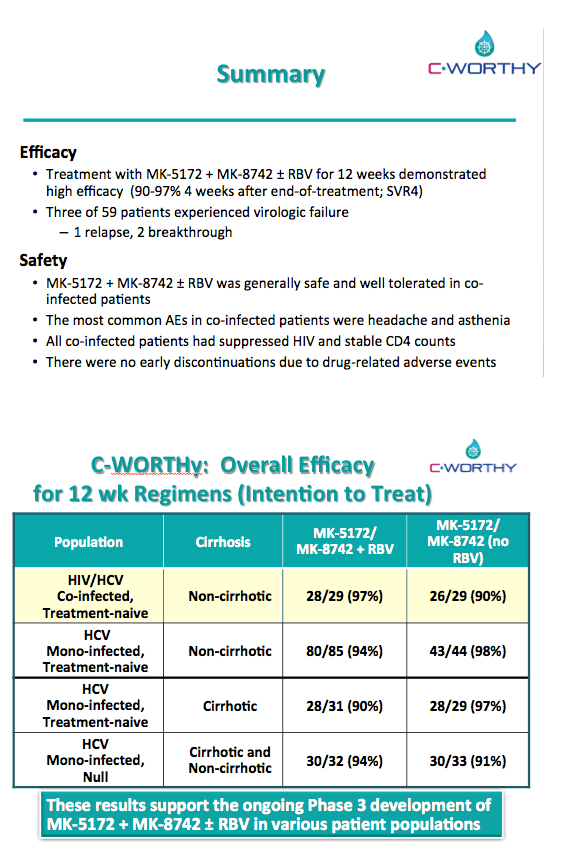
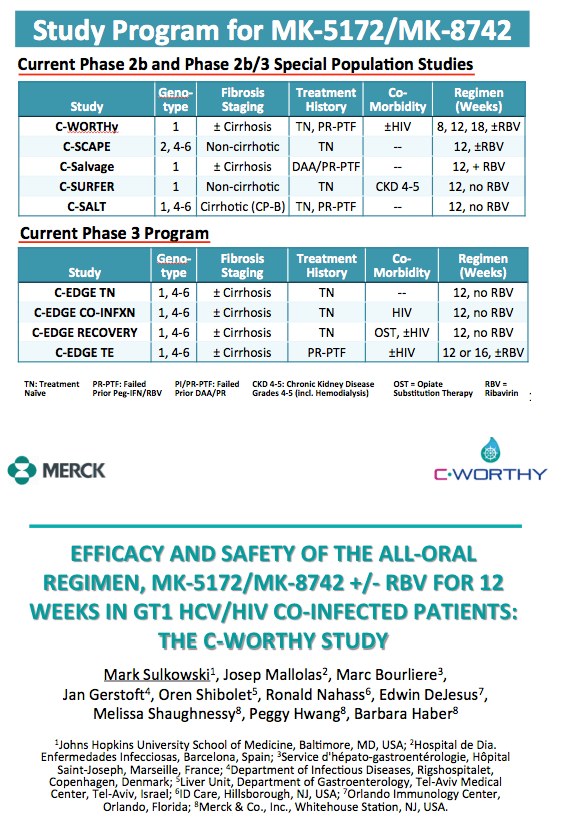
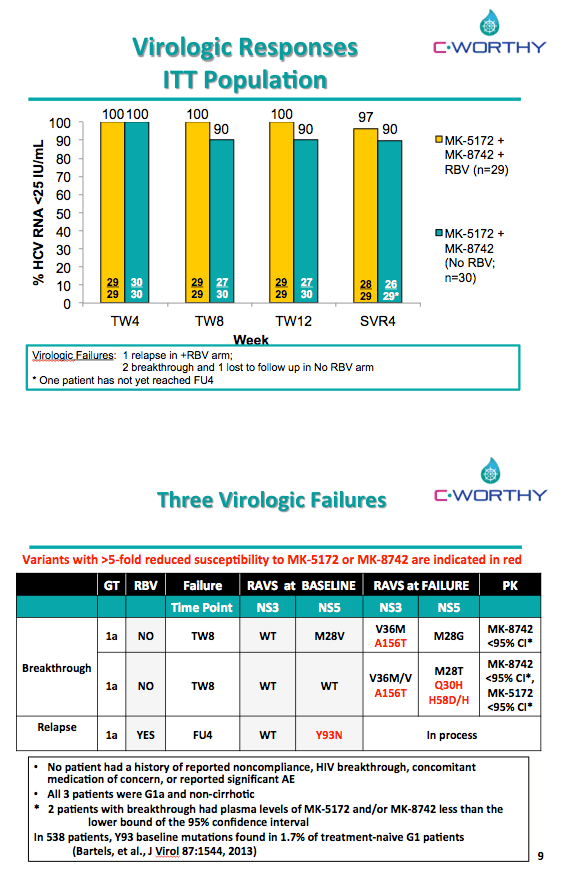
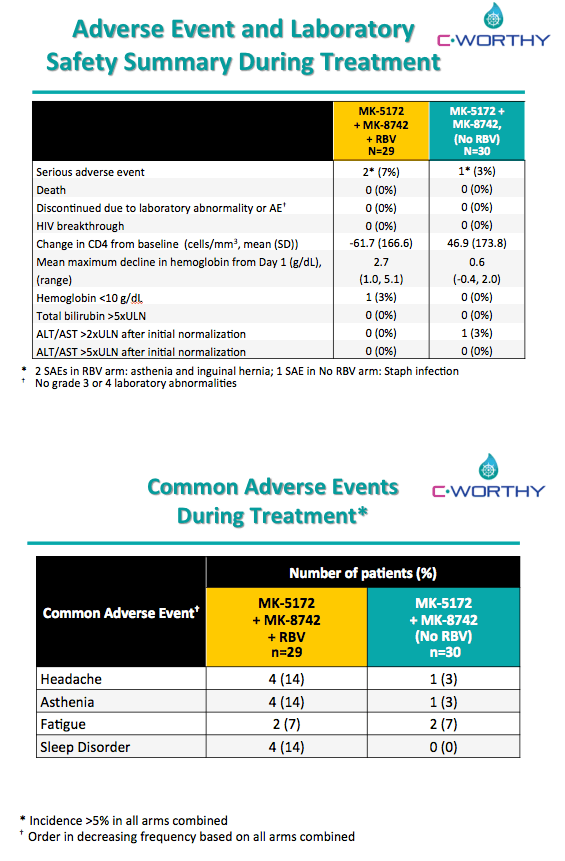
EFFICACY AND SAFETY OF THE ALL-ORAL REGIMEN, MK-5172/MK-8742 +/- RBV FOR 12 WEEKS IN GT1 HCV/HIV CO-INFECTED PATIENTS: THE C-WORTHY STUDY
|
| |
| |
Reported by Jules Levin
EASL 2014 April 9-13 London, UK
Mark Sulkowski1, Josep Mallolas2, Marc Bourliere3,
Jan Gerstoft4, Oren Shibolet5, Ronald Nahass6, Edwin DeJesus7,
Melissa Shaughnessy8, Peggy Hwang8, Barbara Haber8
1Johns Hopkins University School of Medicine, Baltimore, MD, USA; 2Hospital de Dia. Enfermedades Infecciosas, Barcelona, Spain; 3Service
d'hépato-gastroentérologie, Hôpital Saint-Joseph, Marseille, France; 4Department of Infectious Diseases, Rigshospitalet, Copenhagen, Denmark; 5Liver Unit, Department of Gastroenterology, Tel-Aviv Medical Center, Tel-Aviv, Israel; 6ID Care, Hillsborough, NJ, USA; 7Orlando Immunology Center, Orlando, Florida; 8Merck & Co., Inc., Whitehouse Station, NJ, USA.



|
| |
|
 |
 |
|
|