 |
 |
 |
| |
Ledipasvir and Sofosbuvir for Untreated HCV Genotype 1 Infection
|
| |
| |
Download the PDF here
Download the PDF here
Download the PDF here
New England Jnl of Medicine
April 12, 2014
Nezam Afdhal, M.D., Stefan Zeuzem, M.D., Paul Kwo, M.D., Mario Chojkier, M.D., Norman Gitlin, M.D., Massimo Puoti, M.D., Manuel Romero-Gomez, M.D., Ph.D., Jean-Pierre Zarski, M.D., Ph.D., Kosh Agarwal, M.D., Peter Buggisch, M.D., Graham R. Foster, Ph.D., Norbert Bräu, M.D., M.B.A., Maria Buti, M.D., Ph.D., Ira M. Jacobson, M.D., G. Mani Subramanian, M.D., Ph.D., Xiao Ding, Ph.D., Hongmei Mo, M.D., Jenny C. Yang, Pharm.D., Phillip S. Pang, M.D., Ph.D., William T. Symonds, Pharm.D., John G. McHutchison, M.D., Andrew J. Muir, M.D., M.H.S., Alessandra Mangia, M.D., and Patrick Marcellin, M.D., Ph.D. for the ION-1 Investigators
Background
In phase 2 studies, treatment with the all-oral combination of the nucleotide polymerase inhibitor sofosbuvir and the NS5A inhibitor ledipasvir resulted in high rates of sustained virologic response among previously untreated patients with hepatitis C virus (HCV) genotype 1 infection.
Methods
We conducted a phase 3, open-label study involving previously untreated patients with chronic HCV genotype 1 infection. Patients were randomly assigned in a 1:1:1:1 ratio to receive ledipasvir and sofosbuvir in a fixed-dose combination tablet once daily for 12 weeks, ledipasvir-sofosbuvir plus ribavirin for 12 weeks, ledipasvir-sofosbuvir for 24 weeks, or ledipasvir-sofosbuvir plus ribavirin for 24 weeks. The primary end point was a sustained virologic response at 12 weeks after the end of therapy.
Results
Of the 865 patients who underwent randomization and were treated, 16% had cirrhosis, 12% were black, and 67% had HCV genotype 1a infection. The rates of sustained virologic response were 99% (95% confidence interval [CI], 96 to 100) in the group that received 12 weeks of ledipasvir-sofosbuvir; 97% (95% CI, 94 to 99) in the group that received 12 weeks of ledipasvir-sofosbuvir plus ribavirin; 98% (95% CI, 95 to 99) in the group that received 24 weeks of ledipasvir-sofosbuvir; and 99% (95% CI, 97 to 100) in the group that received 24 weeks of ledipasvir-sofosbuvir plus ribavirin. No patient in either 12-week group discontinued ledipasvir-sofosbuvir owing to an adverse event. The most common adverse events were fatigue, headache, insomnia, and nausea.
Conclusions
Once-daily ledipasvir-sofosbuvir with or without ribavirin for 12 or 24 weeks was highly effective in previously untreated patients with HCV genotype 1 infection. (Funded by Gilead Sciences; ION-1 ClinicalTrials.gov number NCT01701401.)
Although the treatment of patients infected with the hepatitis C virus (HCV) has evolved greatly in recent years, newly approved regimens for the treatment of patients with HCV genotype 1 infection still include weekly injections of recombinant human interferon alfa and ribavirin administered according to body weight.1-3 The only interferon-free option currently approved by the Food and Drug Administration for the treatment of HCV genotype 1 infection is 24 weeks of sofosbuvir and ribavirin for patients who are ineligible to receive interferon; the reported response rate is 68%.4
Interferon treatment is associated with a number of side effects, including influenza-like symptoms, depression, and cytopenia.5,6 The side effects of ribavirin include hemolytic anemia, fatigue, pruritus, and rash.7 The elimination of interferon and ribavirin from treatment regimens is expected to reduce the incidence and severity of adverse events, to simplify the treatment of patients with HCV infection, and to provide a treatment option for patients who are ineligible for treatment with interferon or ribavirin. Treatment guidelines recently issued by the American Association for the Study of Liver Diseases and the Infectious Diseases Society of America recommend interferon-free combinations of newly approved direct-acting antiviral agents, but the supporting data are from small, phase 2 studies.8
Ledipasvir (Gilead Sciences) is a new HCV NS5A inhibitor with potent antiviral activity against HCV genotypes 1a and 1b.9 Sofosbuvir is a nucleotide polymerase inhibitor approved for the treatment of HCV genotypes 1 through 4 in combination with ribavirin, with or without peginterferon.1 In phase 2 trials, the combination of ledipasvir and sofosbuvir with or without ribavirin resulted in high rates of sustained virologic response among previously untreated patients with HCV genotype 1 infection, including those with compensated cirrhosis.10,11 In the ION-2 phase 3 trial, previously treated patients, including those with cirrhosis, had rates of sustained virologic response of 94 to 99% after 12 or 24 weeks of treatment with ledipasvir-sofosbuvir, with or without ribavirin.12 We conducted the ION-1 phase 3 trial to assess the efficacy and safety of 12 or 24 weeks of a fixed-dose combination of ledipasvir and sofosbuvir, with or without ribavirin (Ribasphere, Kadmon Pharmaceuticals), in previously untreated patients with chronic HCV genotype 1 infection, including those with compensated cirrhosis.
Methods
Patients
We enrolled patients at 99 sites in the United States and Europe from October 17, 2012, to May 17, 2013. Eligible patients were 18 years of age or older, had chronic HCV genotype 1 infection, and had not received treatment for HCV infection previously. There were no upper limits for age or body-mass index. The protocol (available with the full text of this article at NEJM.org) specified that patients with cirrhosis could account for approximately 20% of the study population. The presence of cirrhosis was defined as a liver-biopsy specimen showing evidence of cirrhosis (Metavir stage F4 [on a scale of F0 to F4, with higher stages indicating a greater degree of fibrosis] or Ishak score of 5 or 6 [on a scale of 0 to 6, with higher scores indicating a greater degree of fibrosis]), a FibroScan score of more than 12.5 kPa (on a scale of 1.5 to 75.0 kPa, with higher scores indicating a greater degree of fibrosis), or a FibroTest score of more than 0.75 (on a scale of 0 to 1, with higher scores indicating more severe fibrosis) and an aspartate aminotransferase:platelet ratio index of more than 2 (with higher scores indicating a greater likelihood of extensive fibrosis). The full set of eligibility criteria, including details of the assessment of cirrhosis, are provided in the study protocol.
Study Design
In this multicenter, randomized, open-label trial, all the patients received a fixed-dose combination tablet containing 90 mg of ledipasvir and 400 mg of sofosbuvir, administered orally once daily. Ribavirin was administered orally twice daily, with the dose determined according to body weight (1000 mg daily in patients with a body weight <75 kg, and 1200 mg daily in patients with a body weight ≥75 kg).
Patients were randomly assigned in a 1:1:1:1 ratio to one of four treatment groups: ledipasvir-sofosbuvir for 12 weeks, ledipasvir-sofosbuvir plus ribavirin for 12 weeks, ledipasvir-sofosbuvir for 24 weeks, or ledipasvir-sofosbuvir plus ribavirin for 24 weeks. Enrollment occurred in two phases. First, 200 patients underwent randomization and were treated. After patients in the 12-week groups reached post-treatment week 4, the data and safety monitoring committee reviewed safety data collected during treatment for all four groups and response data at post-treatment week 4 in the 12-week groups. A planned interim analysis indicated that the rate of sustained virologic response at 4 weeks after the end of treatment was more than 60%, and therefore the prespecified criteria for futility were not met (see the Supplementary Appendix, available at NEJM.org), and an additional 665 patients were randomly assigned to the four treatment groups. Randomization was stratified according to HCV genotype 1 subtype (1a or 1b) and the presence or absence of cirrhosis.
Study Oversight
This study was approved by the institutional review board or independent ethics committee at each participating site and was conducted in compliance with the principles of the Declaration of Helsinki, Good Clinical Practice guidelines, and local regulatory requirements. The study was designed and conducted according to the protocol by the sponsor (Gilead Sciences) in collaboration with the principal investigators. The sponsor collected the data, monitored the study conduct, and performed the statistical analyses. An independent data and safety monitoring committee reviewed the progress of the study.
The investigators, participating institutions, and sponsor agreed to maintain confidentiality of the data. All the authors had access to the data and assume responsibility for the integrity and completeness of the reported data. All the authors affirm that the study was conducted with fidelity to the protocol. The first draft of the manuscript was prepared by a professional writer who is an employee of the sponsor and by the first author, with input from all coauthors.
Study Assessments
Screening assessments included measurement of the serum HCV RNA level and IL28B genotyping, in addition to standard laboratory and clinical tests. The serum HCV RNA level was measured with the use of the COBAS TaqMan HCV Test, version 2.0, for use with the High Pure System (Roche Molecular Systems), which has a lower limit of quantification of 25 IU per milliliter. HCV genotype and subtype were determined with the use of the Versant HCV Genotype INNO-LiPA 2.0 assay (Siemens Healthcare Diagnostics). IL28B genotype was determined by means of polymerase-chain-reaction amplification and sequencing of the rs12979860 single-nucleotide polymorphism.
Assessments during treatment included standard laboratory testing, measurement of the serum HCV RNA level, assessment of vital signs, electrocardiography, and symptom-directed physical examinations. All adverse events were recorded and graded according to a standardized scale (see the study protocol).
Population sequencing or deep sequencing of the NS5A and NS5B regions of the HCV RNA was performed for all the patients at baseline. For all the patients who had virologic failure, deep sequencing of the NS5A and NS5B regions was conducted at baseline and at the time of failure. The sequences obtained at the time of virologic failure were compared with sequences from baseline samples and references in order to detect resistance-associated variants that emerged during treatment. We report resistance-associated variants that were present in more than 1% of the sequence reads.
End Points
The primary efficacy end point was a sustained virologic response at 12 weeks after the end of treatment. It was assessed in all the patients who underwent randomization and received treatment.
Statistical Analysis
In the primary efficacy analysis, the rate of sustained virologic response in each of the treatment groups was compared with an adjusted historical rate of 60%, with the use of a two-sided exact one-sample binomial test. This 60% rate was based on calculated rates of sustained virologic response of 65% in phase 3 trials of telaprevir13 and boceprevir,14 allowing for a rate that was 5 percentage points lower, in exchange for an expected improved safety profile and shorter duration of treatment. The weighted average rates of response in the telaprevir and boceprevir trials were estimated to be approximately 70% among patients without cirrhosis and 44% among those with cirrhosis (see the Supplementary Appendix for details on the calculation of the control rate). We determined that a sample of 200 patients in each treatment group would provide the study with more than 91% power to detect an improvement of at least 13 percentage points in the rate of sustained virologic response from the adjusted historical null rate of 60%, with the use of a two-sided, exact one-sample binomial test at a significance level of 0.0125, based on a Bonferroni correction. The two-sided 95% exact confidence interval calculated by the Clopper-Pearson method is provided for the rate of sustained virologic response at 12 weeks after the end of treatment in each of the four treatment groups.
Results
Baseline Characteristics
Of the 1015 patients who were initially screened, 870 underwent randomization, and 865 began treatment (Table S1 and Figure S1 in the Supplementary Appendix). A total of 5 patients underwent randomization but were not treated owing to withdrawal of consent (4 patients) or erroneous randomization (1 patient). The demographic and baseline clinical characteristics of the patients were generally balanced among the four treatment groups, except that the two treatment groups that received ribavirin had higher proportions of patients with the CC allele of IL28B than did the groups that received ledipasvir-sofosbuvir alone (Table 1).
Overall, 67% of the patients had HCV genotype 1a infection, 12% were black, 70% had the non-CC IL28B genotype, 16% had cirrhosis, 3% had a platelet count of less than 90,000 per cubic millimeter, and 4% had an albumin level of less than 3.5 g per deciliter. A total of seven patients had both a platelet count of less than 90,000 per cubic millimeter and an albumin level of less than 3.5 g per deciliter. A total of 19% of the patients had a history of depression, 14% had insomnia, 12% had anxiety, and 9% had a history of diabetes mellitus or were taking medication for diabetes. Approximately 41% of the patients were enrolled in Europe.
Efficacy
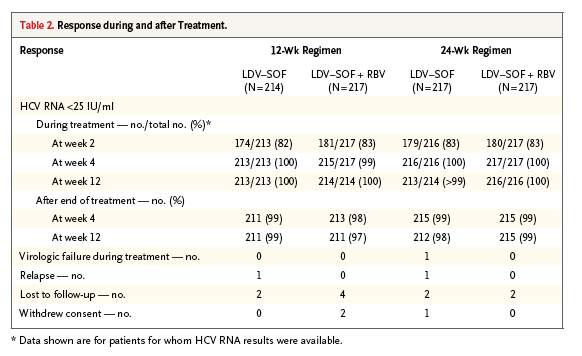
The rates of sustained virologic response in all four treatment groups were superior to the historical rate of 60% (P<0.001 for all comparisons). The rates of sustained virologic response 12 weeks after the end of treatment were as follows: among 214 patients who received 12 weeks of ledipasvir-sofosbuvir, 211 had a sustained virologic response (99%; 95% confidence interval [CI], 96 to 100); among 217 who received 12 weeks of ledipasvir-sofosbuvir plus ribavirin, 211 had a sustained virologic response (97%; 95% CI, 94 to 99); among 217 who received 24 weeks of ledipasvir-sofosbuvir, 212 had a sustained virologic response (98%; 95% CI, 95 to 99); and among 217 who received 24 weeks of ledipasvir-sofosbuvir plus ribavirin, 215 had a sustained virologic response (99%; 95% CI, 97 to 100) (Table 2).
Of the 865 patients who underwent randomization and were treated, only 3 had virologic failure. A total of 1 patient who received 24 weeks of ledipasvir-sofosbuvir had virologic breakthrough during treatment. This patient, a 63-year-old black man with HCV genotype 1b infection, had plasma concentrations of ledipasvir and GS-331007 (the main circulating metabolite of sofosbuvir) that were below the level of quantification at both weeks 8 and 10, which strongly suggests nonadherence to the study regimen. A total of 2 patients had virologic relapse after finishing treatment: a 56-year-old white man with cirrhosis and HCV genotype 1a infection who received 12 weeks of ledipasvir-sofosbuvir had a relapse by post-treatment week 4, and a 65-year-old black man with cirrhosis and HCV genotype 1b infection who received 24 weeks of ledipasvir-sofosbuvir had a relapse between post-treatment weeks 4 and 12.
Given the very high levels of sustained virologic response observed overall, high response rates were observed in all patient subgroups, including patients with characteristics historically associated with a poor response to treatment, who had response rates similar to those among patients without these characteristics (Figure 1). According to the intention-to-treat analysis, the rates of sustained virologic response in the four treatment groups ranged from 94 to 100% among patients with cirrhosis, 97 to 99% among those with HCV genotype 1a infection, 97 to 99% among those with a non-CC IL28B allele, and 91 to 100% among black patients (Figure S2 in the Supplementary Appendix). Of the seven patients with both an albumin level of less than 3.5 g per deciliter and a platelet count of less than 90,000 per cubic millimeter at baseline, six had a sustained virologic response; the other patient withdrew consent after having received only a single dose of ledipasvir-sofosbuvir.
Virologic Resistance Testing
At baseline, variants associated with resistance to NS5A inhibitors were detected in 92 of 861 patients (11%) and were confirmed by means of deep sequencing in 140 of 861 patients (16%), 135 of whom (96%) had a sustained virologic response. Of the 3 patients who had virologic failure, the 2 with relapses had NS5A-resistant variants at baseline, whereas the patient with virologic breakthrough did not. All three samples obtained at the time of virologic failure had NS5A-resistant variants; the patient with HCV genotype 1a infection had the L31M variant, and both patients with HCV genotype 1b infection had the Y93H variant. Neither of the two samples from patients with relapse showed evidence of mutations conferring resistance to sofosbuvir (Table S4 in the Supplementary Appendix).
Safety
Of the 865 patients who underwent randomization and were treated, 10 discontinued ledipasvir-sofosbuvir prematurely owing to adverse events: 4 patients (2%) in the group that received ledipasvir-sofosbuvir for 24 weeks, and 6 (3%) in the group that received ledipasvir-sofosbuvir plus ribavirin for 24 weeks (Table 3). All 10 of these patients had a sustained virologic response; the shortest duration of therapy among these patients was 8 weeks. No patient who received ledipasvir-sofosbuvir for 12 weeks discontinued treatment early (Table S6 in the Supplementary Appendix).
Of the 33 patients who had a serious adverse event during treatment, 25 were in the 24-week groups (18 patients who received ledipasvir-sofosbuvir and 7 who received ledipasvir-sofosbuvir plus ribavirin) and 8 were in the 12-week groups (1 who received ledipasvir-sofosbuvir and 7 who received ledipasvir-sofosbuvir plus ribavirin). Six serious adverse events occurred in more than 1 patient: cellulitis, chest pain, gastroenteritis, hand fracture, noncardiac chest pain, and pneumonia each occurred in 2 patients (Table S5 in the Supplementary Appendix).
The majority of patients in all the treatment groups (79 to 92%) had at least one adverse event. Of the patients who had adverse events, 93% had only events that were mild to moderate in severity (Table 3). The most common adverse events were fatigue, headache, insomnia, and nausea. Patients in the groups that received ledipasvir-sofosbuvir plus ribavirin for 12 or 24 weeks had higher rates of events known to be associated with ribavirin therapy - fatigue, insomnia, asthenia, rash, cough, pruritus, and anemia - than did those in the corresponding groups that received ledipasvir-sofosbuvir without ribavirin (Table 3).
Patients who received 12 or 24 weeks of ledipasvir-sofosbuvir alone had mean changes (from baseline to the end of treatment) in the hemoglobin level of -0.4 g per deciliter and -0.2 g per deciliter, respectively; in the patients who received 12 or 24 weeks of ledipasvir-sofosbuvir plus ribavirin, the mean changes were -2.2 g per deciliter and -1.9 g per deciliter, respectively. Severe hyperbilirubinemia (bilirubin level of >2.5 times the upper limit of the normal range) developed in nine patients (4%) in the 12-week group that received ribavirin and in seven (3%) in the 24-week group that received ribavirin, as compared with hyperbilirubinemia in one patient (<1%), who had Gilbert's syndrome, in the group that received 24 weeks of ledipasvir-sofosbuvir alone.
Discussion
In this multicenter, randomized, phase 3 study, the rates of sustained virologic response in all four treatment groups were 97% or higher, regardless of whether the regimen was given for 12 or 24 weeks and whether it included ribavirin. The rates of treatment discontinuation were higher in the groups treated for 24 weeks than in the groups treated for 12 weeks, and the rates of side effects were higher in the groups that received ribavirin than in the corresponding groups that did not. This trial showed that a 12-week regimen of ledipasvir-sofosbuvir, without ribavirin, constitutes an effective treatment for patients who have HCV genotype 1 infection, with the lowest rate of adverse events of the four regimens evaluated.
The response to interferon-based regimens for HCV infection varies considerably according to certain characteristics of patients (e.g., baseline viral load, race, HCV genotype, IL28B genotype, and extent of fibrosis) and the presence or absence of an early response during treatment.15 In this trial, the rates of response were generally uniform, regardless of the characteristics of the patients at baseline. Given the high rates of response with 12 weeks of therapy, the possibility of further reducing the duration of therapy was explored in the ION-3 study (also now reported in the Journal).16 ION-3 showed that the addition of ribavirin in an 8-week regimen or the extension of treatment to 12 weeks did not result in better response rates than 8 weeks of ledipasvir-sofosbuvir alone.
The rates of response to interferon-based therapy, including protease-inhibitor-containing regimens, have been low in patients with cirrhosis.13-15,17-19 For the protease-inhibitor-containing regimens, 48 weeks of treatment is recommended for all such patients; response-guided shortening of the duration of therapy has been associated with an unacceptable rate of relapse.13 The low rates of response among patients with cirrhosis reflect both an increased risk of interferon-related side effects20 and an unidentified effect of cirrhosis on responsiveness to treatment. The presence of a low platelet count and an albumin level of less than 3.5 g per deciliter have been identified as risk factors for adverse events, including infection, hepatic decompensation, and death. In this study, the presence of cirrhosis had no marked effect on rates of response or the safety profile, even among patients with thrombocytopenia and a low albumin level. This study, however, was not designed or powered to formally compare the rates of response among patients with cirrhosis and those without cirrhosis.
Virologic failure was extremely rare in this study population, occurring in only 0.3% of patients (3 of 865). In these patients, we did not discern clinical or virologic predictors of virologic failure except suspected nonadherence to therapy, which was documented by means of serum assays for drug levels, in one patient with virologic breakthrough during treatment. The two patients with relapse had no evidence of mutations conferring resistance to sofosbuvir, although both patients had mutations associated with resistance to NS5A inhibitors both at baseline and at the time of relapse. A sofosbuvir-based regimen with a protease inhibitor would be a potential option for retreatment in these patients.
There was no control group in this study, and we were not able to assess which adverse events resulted from ledipasvir-sofosbuvir. The most common adverse events included fatigue, headache, insomnia, and nausea. The type, frequency, and severity of the adverse events observed in the groups that received ledipasvir-sofosbuvir without ribavirin were generally similar to those seen in the placebo group in a previous trial of sofosbuvir and ribavirin in patients with HCV infection.21 However, the groups receiving ledipasvir-sofosbuvir plus ribavirin in our study had a higher incidence of adverse events and laboratory abnormalities that are known to be associated with ribavirin therapy - fatigue, insomnia, cough, pruritus, and anemia.6 Thus, our findings suggest that the addition of ribavirin to a ledipasvir-sofosbuvir regimen increases toxicity without providing additional efficacy.
In conclusion, our study showed that 12 weeks of the single-tablet regimen of ledipasvir-sofosbuvir was a highly effective treatment for a broad range of patients with HCV genotype 1 infection who had not been treated previously. No additional benefit appeared to be associated with the addition of ribavirin or with extension of the duration of treatment to 24 weeks.
|
| |
|
 |
 |
|
|