 |
 |
 |
| |
HIGH RATE OF SUSTAINED VIROLOGIC RESPONSE IN PATIENTS WITH HCV GENOTYPE-1A INFECTION: A PHASE 2 TRIAL OF FALDAPREVIR, DELEOBUVIR AND PPI-668, WITH AND WITHOUT RIBAVIRIN
|
| |
| |
Reported by Jules Levin
EASL 2014 April 9-13 London, UK
J. Lalezari1, L. Holland1, E. Glutzer1, P. Vig2, M. Elgadi3, J.O. Stern3, R. Colonno2, S. Halfon2, E. Ruby2, N. Huang2, E. Nash2, and N. Brown2
1Quest Clinical Research (San Francisco, CA, U.S.A.); 2Presidio Pharmaceuticals (San Francisco, CA, U.S.A.); 3Boehringer Ingelheim Pharmaceuticals (Ridgefield, CT, U.S.A.)



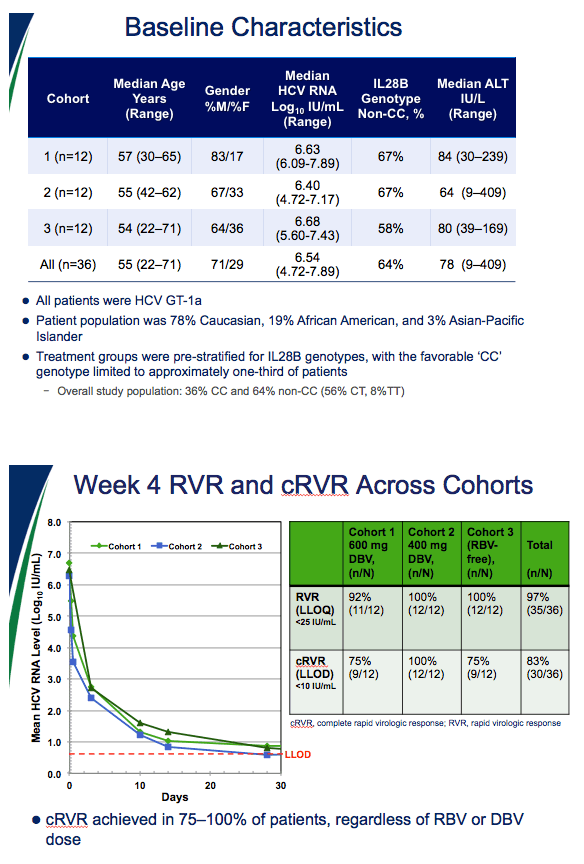

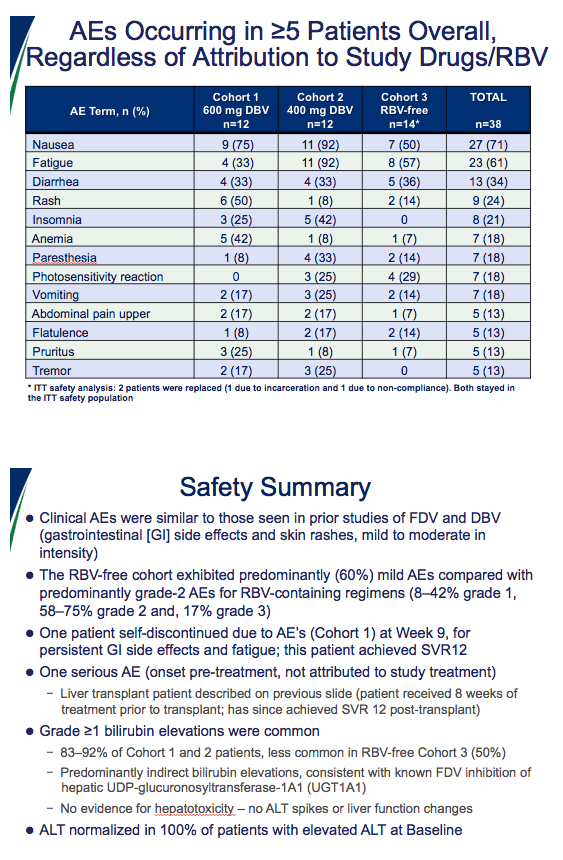

|
| |
|
 |
 |
|
|