 |
 |
 |
| |
An Interferon- and Ribavirin-Free 12-Week Regimen of Once-Daily VX-135 and Daclatasvir in Treatment-Naïve Patients With Genotype 1 HCV Infection
|
| |
| |
Reported by Jules Levin
EASL 2014 April 9-13 London, UK
E. Gane1, C. Stedman2, V. Garg3, S. George3, T. Kieffer3, J. Krop3, L. Lan3, R. Rubin3, and A. Lawal3
1. Auckland Clinical Studies, Grafton, New Zealand; 2. Christchurch Clinical Studies, Christchurch, New Zealand; 3. Vertex Pharmaceuticals Incorporated, Boston, MA, USA
Vertex Announces Sustained Viral Response Rate (SVR4) Data from All-Oral Study of VX-135 in Combination with Daclatasvir in Hepatitis C - (01/31/14)
VX-135, A Once-daily Nucleotide HCV Polymerase Inhibitor, Was Well Tolerated And Demonstrated Potent Antiviral Activity When Given With Ribavirin In Treatment-naïve Patients With Genotype 1
HCV......http://www.natap.org/2013/AASLD/AASLD_14.htm



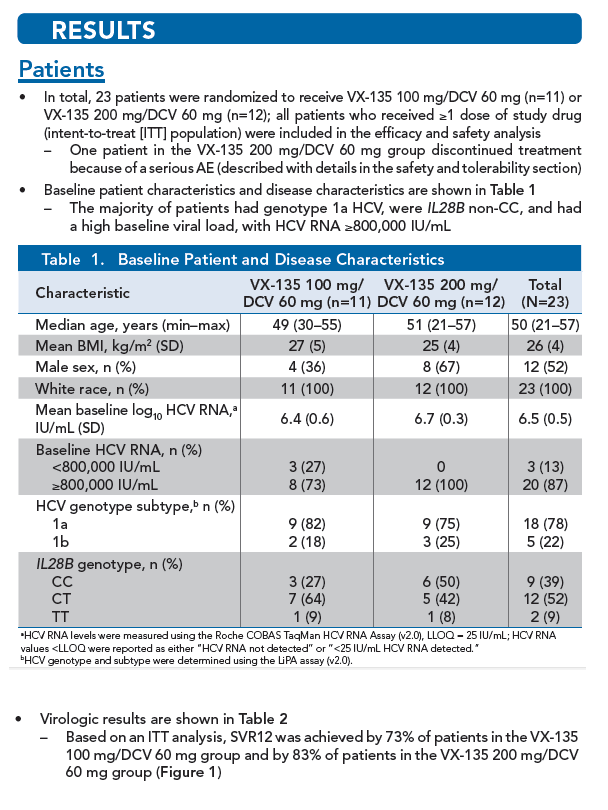

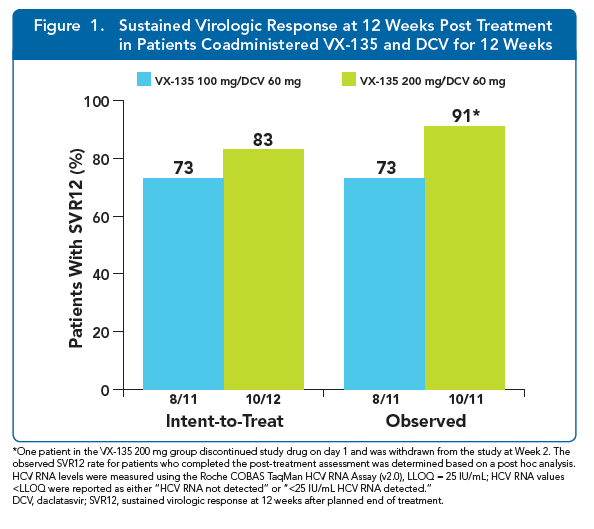

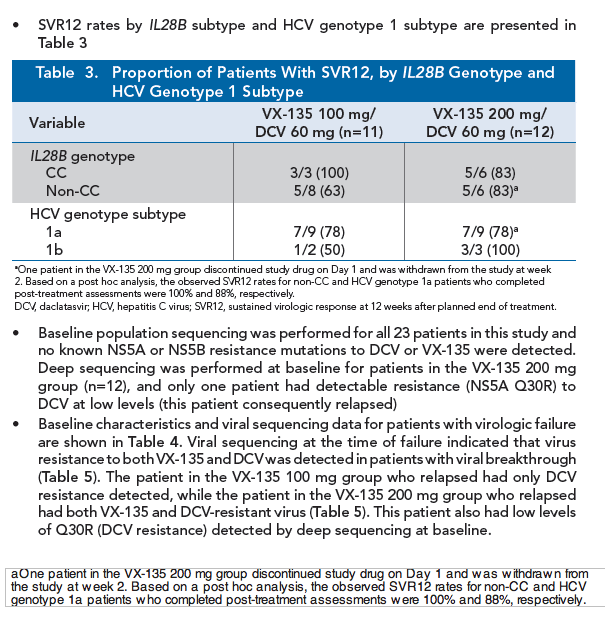
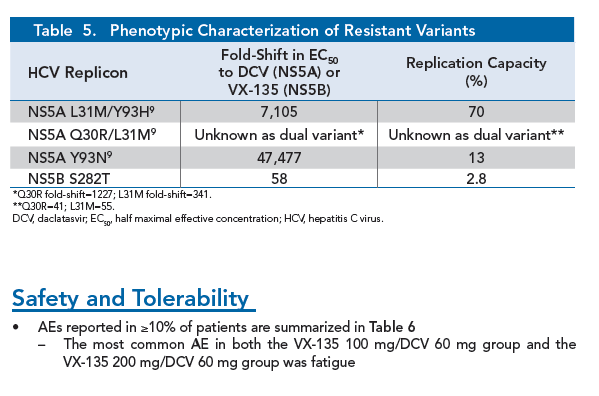
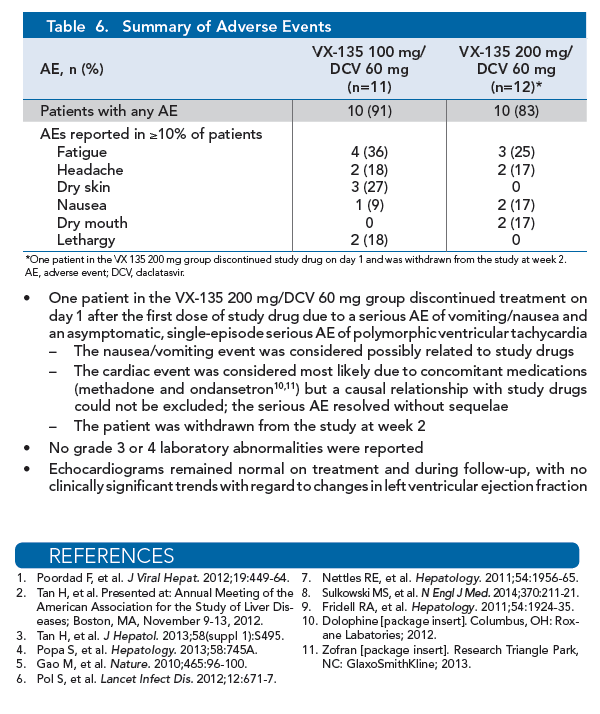
|
| |
|
 |
 |
|
|