 |
 |
 |
| |
Ledipasvir and Sofosbuvir Combination Improves Patient-Reported Outcomes During Treatment of Chronic Hepatitis C: The Result from ION-1 Clinical Trial
|
| |
| |
Reported by Jules Levin
EASL 2014 April 9-13 London, UK
EASL: All Oral Fixed-Dose Combination Ledipasvir/Sofosbuvir With Or Without Ribavirin for 12 or 24 Weeks in Treatment-Naive Genotype 1 HCV-Infected Patients: the Phase 3 ION-1 Study - (04/14/14)
EASL: New DAA Phase 3 Studies for genotype 1 at EASL - in both Treatment-naives, treatment-experienced; simultaneous publications - (04/21/14)
Zobair M. Younossi, Maria Stepanova, Patrick Marcellin, Nizam Afdhal, Sharon L. Hunt
1. Center for Liver Diseases, Department of Medicine, Inova Fairfax Medical Campus, Falls Church VA, United States.
2. Viral Hepatitis Research Unit in Hopital Beaujon, Clichy, France.
3. Hepatology, Beth Israel Deaconess Medical Center, Boston, MA, United States.
4. Betty and Guy Beatty Center for Integrated Research, Inova Health System, Falls Church VA, United States.

Program abstract
Background and aims: Interferon-based regimens have substantial negative impact on PROs and Quality-of-life. We aimed to assess PROs in a cohort of treatment-naïve CH-C patients treated with SOF+LDV ± ribavirin (RBV).
Methods: Chronic Liver Disease Questionnaire-HCV (CLDQ-HCV), Short Form-36 (SF-36), Functional Assessment of Chronic Illness Therapy-Fatigue (FACIT-F), and Work Productivity and Activity Index: Specific Health Problem (WPAI:SHP) were administered at baseline, during and post-treatment to CH-C subjects treated with SOF+LDV+RBV or SOF+LDV.
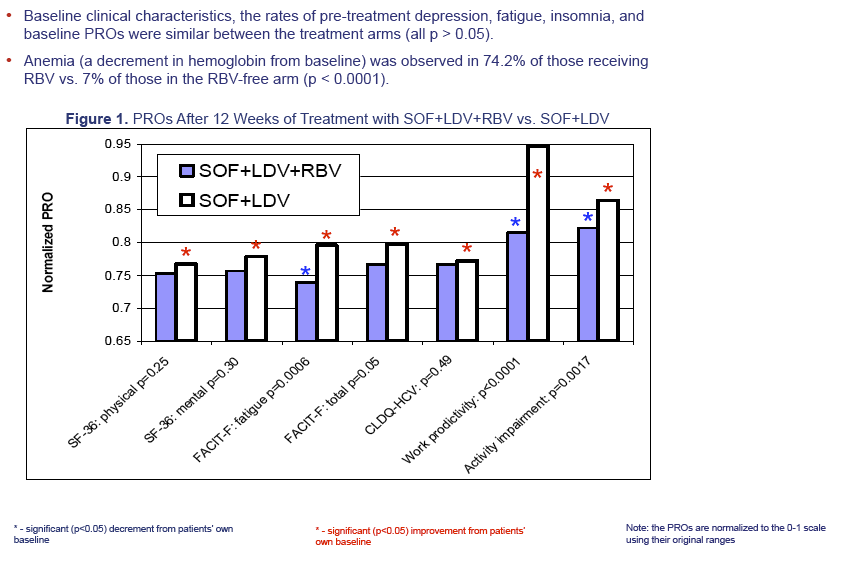
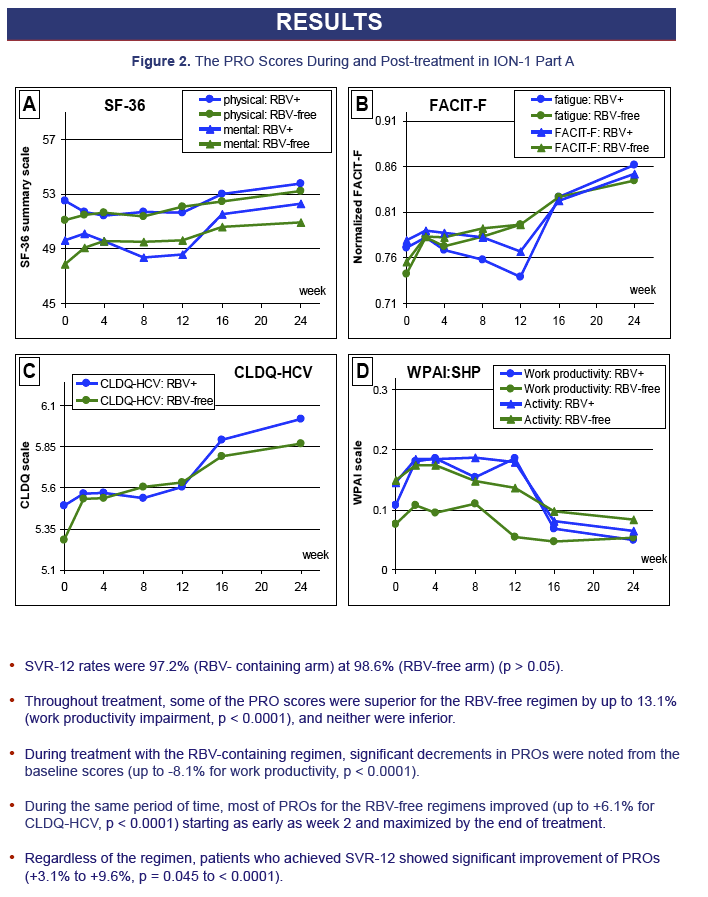
Results: 1431 treatment-naive CH-C subjects were enrolled into 12-Week treatment groups: age 52.0±11.1, 59.2% males, 87.0% whites, 15.7% cirrhosis, BMI 26.6±4.8. Baseline demographic and co-morbidities (depression, fatigue, insomnia) were similar between the groups. SVR-12 was 97.2% and 97.7% for SOF+LDV+RBV and SOF+LDV (P=0.78). During treatment, 74.2% (RBV-containing) vs. 7.0% (RBV-free) of patients experienced anemia (hemoglobin drop>2g/dL; p< 0.0001). Throughout treatment, some of the PRO scores, especially those related to fatigue, activity and work productivity, were superior for RBV-free regimen (by up to 13.1%; p-values ≤0.5 to < 0.0001). During treatment with RBV-containing regimen, significant decrements in PROs were noted from baseline scores (up to -8.1%, p< 0.0001); while most of PROs for the RBV-free regimens improved (up to +6.1%, p< 0.0001). This improvement was noted as early as treatment week 2, and maximized by end of treatment. Regardless of the regimen, patients who achieved SVR-12 showed significant improvement of PROs (+3.1% to +9.6%, p=0.045 to < 0.001).
Conclusions: SOF+LDV (RBV-free regimen) for treatment-naïve genotype 1 CH-C patients is associated with high efficacy and significantly better PRO scores both during treatment and after achieving SVR-12.

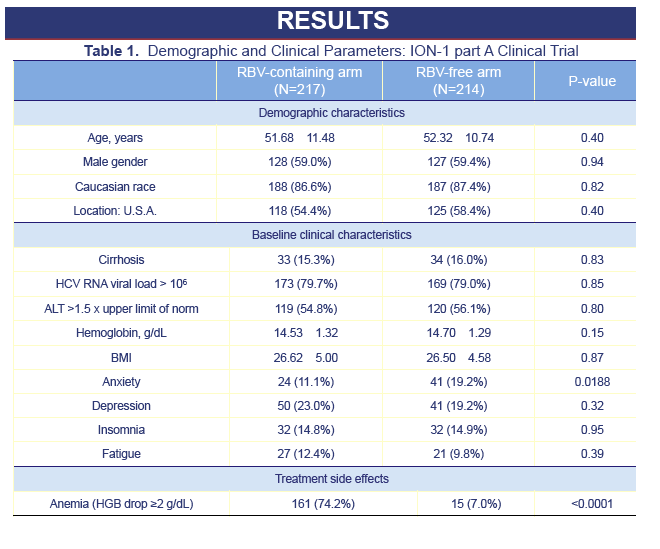
from Jules: during 12 weeks on treatment with SOF/Lepidasvir all measures of PRO, patient reported outcomes improved, and as you can see 12 weeks after treatment ended improvements continued. I expect over time after an SVR further improvements in all categories. For patients in the Rbv arm you can see slight dips in measures while on treatment but after treatment ended every measure improved. The SOF+LDV regimen does not have interferon or ribavirin & actually causes improvement of PROs DURING treatment, you can see improvements begin as early as week 2 and continue improvement through treatment & further continued after treatment during 12-week follow-up. This is the first time in HCV history that someone has shown a good treatment without side effect can improve patients PROs while on treatment. Obviously after treatment most patients with SOF+LDV will achieve SVR and will have improvement of PROs.
|
| |
|
 |
 |
|
|