| |
Daclatasvir EMA Summary of Product Characteristics/Label
|
| |
| |
Download the PDF here
European Medicines Agency. Daklinza (daclatasvir) summary of product characteristics.
2014; http://www.ema.europa.eu/docs/en_GB/document_library/EPAR_-
_Product_Information/human/003768/WC500172848.pdf.
http://www.ema.europa.eu/docs/en_GB/document_library/EPAR_-_Product_Information/human/003768/WC500172848.pdf
http://www.ema.europa.eu/docs/en_GB/document_library/EPAR_-_Product_Information/human/003768/WC500172848.pdf
Package leaflet: Information for the patient
Daklinza 60 mg film-coated tablets
daclatasvir
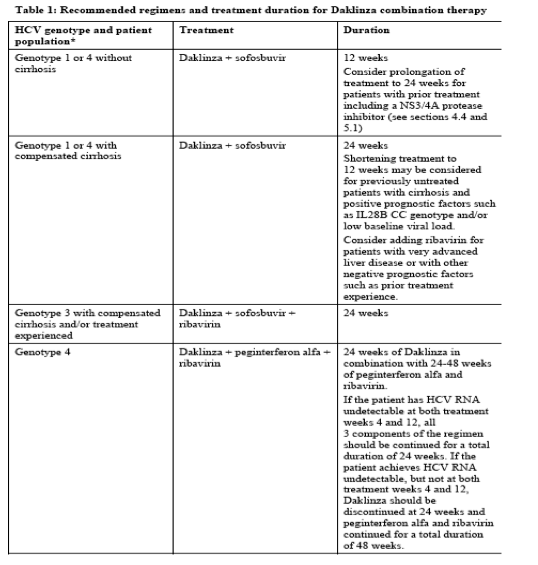
Daklinza is indicated in combination with other medicinal products for the treatment of chronic
hepatitis C virus (HCV) infection in adults (see sections 4.2, 4.4 and 5.1).
Antiviral activity in cell culture
Daclatasvir is an inhibitor of HCV genotypes 1a and 1b replication in cell-based replicon assays with effective concentration (50% reduction, EC50) values of 0.003-0.050 and 0.001-0.009 nM, respectively, depending on the assay method. The daclatasvir EC50 values in the replicon system were 0.003-1.25 nM for genotypes 3a, 4a, 5a and 6a, and 0.034-19 nM for genotype 2a as well as 0.020 nM for infectious genotype 2a (JFH-1) virus.
Daclatasvir showed additive to synergistic interactions with interferon alfa, HCV nonstructural protein 3 (NS3) protease inhibitors, HCV nonstructural protein 5B (NS5B) non-nucleoside inhibitors, and HCV NS5B nucleoside analogues in combination studies using the cell-based HCV replicon system. No antagonism of antiviral activity was observed.
No clinically relevant antiviral activity was observed against a variety of RNA and DNA viruses, including HIV, confirming that daclatasvir, which inhibits a HCV-specific target, is highly selective for HCV.
|
|
| |
| |
|
|
|