| |
Mapping Diverse Global HCV Genotype Distribution- Europe/1b in Far East
|
| |
| |
Download the PDF here
Download the PDF here
BMS Submits First All-Oral, Interferon-Free and Ribavirin-Free Treatment Regimen for Regulatory Review in Japan for Patients with Chronic Hepatitis C Infection
http://www.natap.org/2013/AASLD/AASLD_08.htm
Simeprevir has been approved in Japan for the treatment of genotype 1 chronic hepatitis C infection.....http://www.natap.org/2013/HCV/093013_02.htm
Genotype 1b:
Abbvie - AASLD: Interferon- and Ribavirin-free Regimen of ABT-450/r + ABT-267 in HCV Genotype 1b-infected Treatment-naïve Patients and Prior Null Responders - (11/04/13)
BMS - AASLD: All-Oral Combination of Daclatasvir Plus Asunaprevir in Interferon-Ineligible Naïve/Intolerant and Nonresponder Japanese Patients Chronically Infected With HCV Genotype 1b: Results From a Phase 3 Trial - (11/06/13)
As reported by Jurgen Rockstroh, MD, in his NATAP AASLD report [http://www.natap.org/2013/AASLD/AASLD_109.htm], in the NIAID SYNERGY Trial [http://www.natap.org/2013/AASLD/AASLD_30.htm] 60 HCV mono-infected, treatment naïve, GT-1, patients were consecutively enrolled into 3 arms of a phase 2 clinical trial and received: Arm A - sofosbuvir with ledipasvir (400mg/90mg respectively once daily in a fixed dose combination (FDC)) for 12 weeks, Arm B - FDC + GS-9669 (500mg/day), a non nucleoside NS5B inhibitor for 6 weeks, or Arm C - FDC + GS-9451 (80mg/day), an HCV protease inhibitor for 6 weeks (47). 80-95% of study participants were African-American. In this exploratory trial virological responses were very good with 100% SVR12 in the SOF/LDV (12 weeks arm) (n=20), 90% SVR4 in the SOF/LDV/9669 (6 weeks arm) (n=20) and again 100% SVR4 in the SOF/LDV/9451 (6 weeks arm) (n=20). Only one relapse occurred in the SOF/LDV/9669 arm. Tolerability in all arms was good with no treatment related discontinuation or SAEs. Again this 3 DAA combination study underlines that multiple DAA combinations will work and allow to get rid of interferon as well as ribavirin which is accompanied by greatly improved tolerability. Also the combination of 3 DAAs may allow to shorten treatment duration to 6 weeks at least in easier to treat naïve patient populations which may be very important in the more adherence challenged patient groups.
"In North America, genotype 1a predominates, followed by genotypes 1b, 2a, 2b, and 3a. In Europe, genotype 1b is predominant, followed by genotypes 2a, 2b, 2c, and 3a. Genotypes 4 and 5 are found almost exclusively in Africa. Subtype 1a is prevalent in North and South America, Europe, and Australia, and subtype 1b is common in North America and Europe and is found in parts of Asia. Genotype 2 is present in most developed countries but is less common than genotype 1. Genotype 3a appears to be prevalent among injected drug users and may have entered North America and the United Kingdom with the widespread use of heroin in the 1960s [3]. In Egypt, genotype 4a is the most prevalent genotype, and up to 20% of the population is infected with this subtype. Other genotype 4 subtypes are found in the Middle East as well as North and Central Africa. Genotype 6 is common in several distinct locations in South-east Asia, such as Hong Kong and Vietnam. Genotype 5a is responsible for at least 30% of cases in South Africa but is seldom found elsewhere. The distribution of genotypes is also influenced by the mode of transmission and racial group distribution. In Europe and the United States, individuals who acquire HCV from a blood transfusion are likely to be infected with subtype 1b; however, with the introduction of blood donation screening, the number of individuals with this subtype is likely to decrease. Genotypes 1a and 3a are the most prevalent genotypes among intravenous drug users; with the decline in blood transfusions, acquired HCV infection may become the predominant genotype in Europe and the United States." [European Scientific Journal November 2012 I think]
Global distribution of HCV genotypes and clinical
implications
Geographical differences appear to exist in the distribution of HCV genotypes (Table 2) [4,5,33,40,41]. Genotype 1a, the prototype sequence used in the development of early HCV diagnostic assays and frequently found associated with intravenous drug abuse, is most commonly detected in the United States and Europe. Genotype 1b, principally transmitted via blood transfusions and currently the most common genotype, is distributed worldwide with a high prevalence in the United States and Europe as well as Japan, where 1b is responsible for over 70% of cases of HCV infection [4,42]. Genotypes 2a and 2b, representing 10-30% of global HCV types, are common in North America, Europe and Japan, while genotype 2c is found in Northern Italy. Genotype 3 is most prominent in the Indian subcontinent as well as Southeast Asia and Indonesia. Of interest, subtype 3a is particularly prevalent in injecting drug users in Western Europe and the United States, and, in combination with genotype 1a, accounts for 70% of new HCV infection cases due to the efficient route of transmission [4,43]. Genotype 4 appears to be prevalent in North Africa and the Middle East, while genotypes 5 and 6 are most frequently reported in South Africa and Hong Kong, respectively [44]. Although the global distribution of HCV genotypes can be summarized in this fashion, the prevalence of HCV genotypes may vary in urban settings with immigrant populations, where less common genotypes may be locally predominant. Furthermore, the prevalence of HCV genotypes and subtypes may also vary in major metropolitan areas that serve as centers for travel and which tend to have greater numbers of injecting drug users [45]. In addition, a portion of HCV-infected patients have mixed genotypes and subtypes, with 1a and 1b being the most common mixture [33]. These mixed infections may be more common than first thought and may be an important factor associated with the immune response to HCV infection and response to therapy [46].

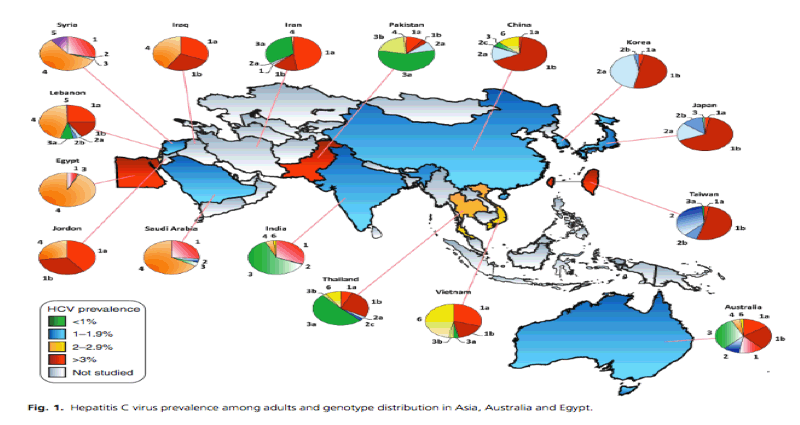
Hepatitis C virus epidemiology in Asia, Australia and Egypt/Genotype Distribution/ HCV Prevalence
http://www.natap.org/2011/HCV/080611_02.htm
China
Genotype 1b was the most prevalent at 66.2%, and genotype 2a showed a prevalence of 13.7%. Statistically significant geographical differences were observed, and genotype 6 was only observed in the South (56). A more recent study from Hong Kong sampled 1055 IDUs and non-IDUs in 1998-2004. The non-IDU population showed a genotype 1b prevalence of 63.6%. Genotypes 2a and 3 had prevalence rates of 3.1 and 3.9%, respectively, and genotype 6a was found in 23.6% of participants. The IDU population showed statistically different genotype distributions, where genotype 6a was seen in 58.5% and 1b in 33.0% (55).
Taiwan
A study of 418 chronic HCV patients at a tertiary referral hospital and another on 1164 patients from three hyperendemic areas found very similar genotype distribution with 1b and 2a being dominant: 1 [48% with 1a (2.6%), 1b (45.5%)], 2 [39.5% with 2a/c (30.9%), and 2b (6.9%)], 3a (1%), 4 (0.2%), 6 (0.5%)
Thailand
An analysis of 45 samples collected in 2004 from four separate regions of Thailand showed genotype 3 as dominant: 1 [33.3% with 1a (6.7%) and 1b (26.7%)], 2c (4.4%), 3 [53.3% with 3a (51.1%) and 3b (2.2%)] and 6 (8.9%) (305). Additional studies sampled blood donors and estimated genotype 3 at 44% of the infected population (318, 319). In contrast, a sample of 46 chronic liver disease patients found genotype 1 as most prevalent. Genotype 1 and its subtypes comprised 48% (320).
Vietnam
The most common HCV genotypes in Vietnam were 1 and 6. There were a number of genotype studies (322, 322, 323, 326, 327). A study in 70 RNA-positive blood donors in Hanoi reported the following genotypes: 1 [47.1% with 1a (30.0%) and 1b (17.1%)], 3 [5.8% with 3a (2.9%) and 3b (2.9%)] and 6 [47.1% with 6a (37.1%), 6e (8.6%) and 6i (1.4%)] (328). Genotype 6 was reported to occur in South China as well as Vietnam, Laos, Thailand and Myanmar (328). In 79 HCV RNA-positive donors from Ho Chi Minh City and four HCV RNA-positive donors from Hanoi, genotype 1 was the predominant genotype (54.0%), composed of genotype 1a (27.0%), 1b (23.0%) and mixed genotype 1 (4%) (323). However, 41% of the genotyped samples were not classifiable into genotypes 1, 2 or 3, and further analysis indicated the majority of the unknown samples were genotype 6a (19.3%) (327). This suggested a geographical distribution of HCV genotypes in Vietnam. Smaller studies reported genotype 1 as the predominant genotype, ranging from 42.8 to 75.0% (322, 326), with the majority typed as genotype 1a (23.8-50.0%), followed by 1b (23.8-25.0%).
India
The most prevalent genotype was 3, with estimates ranging between 61.8 and 80.2% (101, 102, 104-106, 126-135). A study of 2118 patients across the country found genotypes 1 (31.2% with 1a/b=92.4%, 1c=7.6%), 2 (0.5%), 3 (61.8% with 3a/b=94.9%, 3g/k=5.1%), 4a/d (4.5%) and 6 (1.9%) (126).
Japan
There were a number of publications from 1993 to 2000, and almost all indicated genotype 1b as dominant (158, 159, 171, 187-189). In a 2000 study with 166 samples from an endemic area, the following genotypes were identified: 1b (63%), 2a (25%) and mixed/other (12%) (158)
Korea
The Park et al. study (201) reported the following genotypes: 1 [50.3% with 1a (3.0%) and 1b (47.3%)], 2 [45.0% with 2a (42.6%) and 2b (2.4%)] and mixed/other (4.7%).
Pakistan
Genotype distribution information was derived from three studies, which agreed that genotype 3 is the most prevalent genotype (208, 225, 245). The largest study included 3351 individuals from across the county and found the following genotypes: 1 [11.5% with 1a (8.3%) and 1b (3.0%)], 2 [8.4% with 2a (7.5%) and 2b (0.8%)], 3 [67.5% with 3a (49.1%) and 3b (17.7%)] (208).
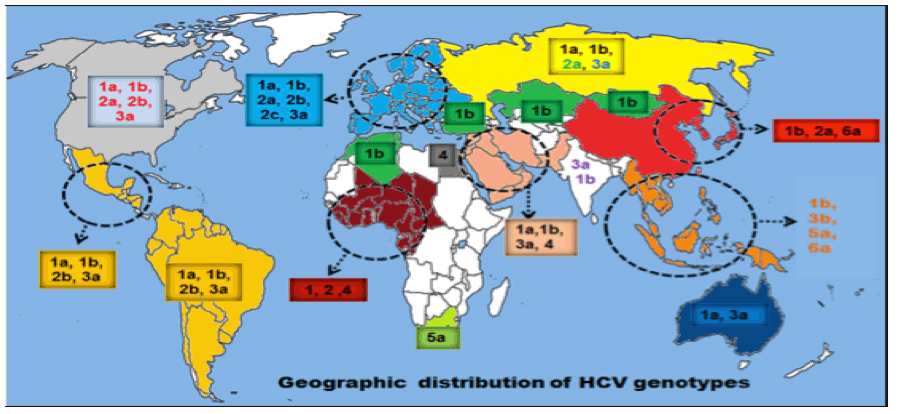
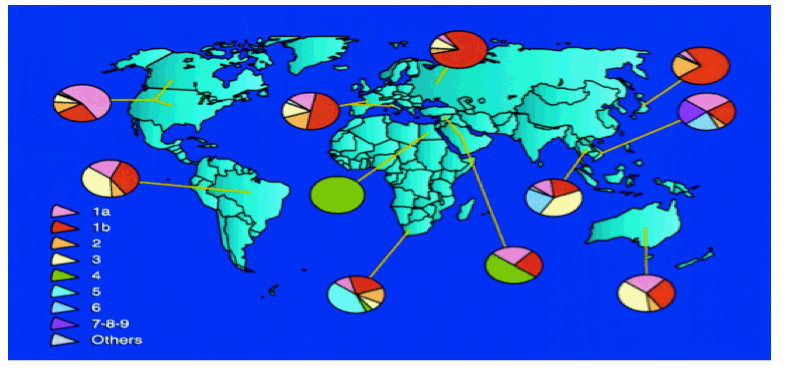
"HCV subtypes 1a and 1b are the most common genotypes in the United States (Fig. 3) (145). These subtypes also are predominant in Europe (35, 82, 91). In Japan, subtype 1b is responsible for up to 73% of cases of HCV infection (124). Although HCV subtypes 2a and 2b are relatively common in North America, Europe, and Japan, subtype 2c is found commonly in northern Italy. HCV genotype 3a is particularly prevalent in intravenous drug abusers in Europe and the United States (101). HCV genotype 4 appears to be prevalent in North Africa and the Middle East (1, 18), and genotypes 5 and 6 seem to be confined to South Africa and Hong Kong, respectively (17, 116). HCV genotypes 7, 8, and 9 have been identified only in Vietnamese patients (129), and genotypes 10 and 11 were identified in patients from Indonesia (127). There has been disagreement about the number of genotypes into which HCV isolates should be classified. Investigators have proposed that genotypes 7 through 11 should be regarded as variants of the same group and classified as a single genotype, type 6 (30, 83, 118, 128).".....http://cmr.asm.org/content/13/2/223.full
|
|
| |
| |
|
|
|