| |
HCV Sex Transmission, Early Reports from 1995/2002 Among Men, Women at Johns Hopkins STD Clinics & other Studies, HIV+ MSM from the Netherlands
|
| |
| |
Download the PDF here
Download the PDF here
Download the PDF here
HCV Sexual Transmission - 2 studies below from 1995 at Johns Hopkins STD Clinics finding HCV sex transmission among non-IDU men & women, mostly African-Americans, & in Netherlands among HIV+ MSM; and links below to studies from 2002/2003/2006 reporting the same, particularly sex transmission to non-IDU heterosexual HIV+ women
In the publication below following the Hopkins study is a study published in 2007 from the Netherlands finding: "this article is the first to suggest an increase in HCV incidence over time among HIV-positive but not HIV-negative MSM on the basis of 20 years of prospective longitudinal cohort data. Additional phylogenetic analysis of HIV/HCV-coinfected cohort and hospital cases strongly supports the presence of an MSM-specific transmission network, probably sexual, in Amsterdam.......We retrospectively screened 1836 men who have sex with men (MSM) participating in the Amsterdam Cohort Studies (1984-2003) for hepatitis C virus (HCV) antibodies. HCV incidence was 0.18/100 person-years (PY) in human immunodeficiency virus (HIV)-positive MSM (8/4408 PY [95% confidence interval {CI}, 0.08-0.36]) but was 0/100 PY in MSM without HIV (0/7807 PY [95% CI, 0.00-0.05]). After 2000, HCV incidence among HIV-positive men increased 10-fold to 0.87/100 PY (5/572 PY [95% CI, 0.28-2.03]). Additional hospital cases ( n=34) showed that MSM in Amsterdam who acquired HCV infection after 2000 reported high rates of ulcerative sexually transmitted infections (59%) and rough sexual techniques (56%), denied injection drug use, and were infected mainly with the difficult-to-treat HCV genotypes 1 (56%) and 4 (36%). Phylogenetic analysis showed 3 monophyletic clusters of MSM-specific HCV strains. The emergence of an MSM-specific transmission network suggests that HIV-positive MSM with high-risk sexual behaviors are at risk for sexually acquired HCV. Targeted prevention and routine HCV screening among HIV-positive MSM is needed to deter the spread of HCV.
Early Reports [2006] of Sexual HCV Transmission in HIV+ MSM, HIV+ Non-IDU Women, and HCV Sexual Transmission at Johns Hopkins STD Clinics in Baltimore Reported in 1995 Publication where they reported: "Anti-HCV was detected in 7% and 4% of non-injection drug-using males and females. respectively......Females with anti-HCV-positive sex partners were 3.7 times more likely to have anti-HCV than females with anti-HCV-negative sex partners.......This investigation provides some evidence that sexual transmission of HCV occurs among patients attending the Baltimore STD clinics.......Anti-HCV was more frequent in male and female patients who had greater numbers of sex partners (even after adjusting for age and other associated factors), and anti-HCV was associated with other sexually transmitted diseases, such as HIY and Trichomonas infections......Ninety-five percent of patients in our investigation were African-American.......even after excluding injection drug users, the rate of anti-HCV was 13.3% among persons attending the Johns Hopkins Emergency Department and 9.6% among patients visiting STD clinics......In this investigation, anti-HCV was found in 18% of males with past homosexual exposure; these males were 3.6 times more likely to have anti-HCV than male STD patients with only heterosexual partners........Studies that have found little or no transmission of HCV to sex partners differ from ours in that they evaluated longterm, monogamous sex relationships [15-19]. Though it is difficult to make direct comparisons, it is reasonable to assume that the number of sex partners, the prevalence of other sexually transmitted diseases, and perhaps even the frequency and types of sexual activity would be different in STD patients than in hemophiliacs and transfusion recipients and their spouses. If the sexual transmission of HCV, like HIV and hepatitis B, is enhanced in the presence of genital ulceration or other STDs, then the more frequent occurrence of these conditions in STD patients may explain the greater estimate of sexual transmission derived from studies in this setting [32-35]. In addition, since sexual transmission of HCV in any setting appears to be infrequent, a large number of partnerships must be studied to demonstrate features indicative of transmission."
"Males represented 53% of the study population and. compared with females. had a higher prevalence of anti-HCV (P = .05). anti-HIV (P = .002). and syphilis (P = .006). Males were also more likely than females (P <.001) to have >45 lifetime sex partners (highest quartile) and to have a history of crack cocaine use. Among males. anti-HCV was associated with older age (P < .001). a history of Trichomonas (P = .002) or syphilis (P = .046) infections. homosexual exposures (P = .003). and anal-receptive sex (P= .03; table 1). Anti-HCY was also more prevalent in males reporting >45 lifetime sex partners (the highest quartile; P <.001). in those who smoked cigarettes (P < .001) but not marijuana (P = .978). and in males acknowledging alcohol use more than once a week (P =.006). In addition. serologic evidence of syphilis (P = .016) and HIV infections (P <.00 I) was associated with anti-HCV......
......Female participants. Females comprised 47% of the study population. As with males. anti-HCY in females was associated with increasing age (P < .001). greater numbers of lifetime heterosexual partners (P < .001). cigarette smoking (P = .003). and more than weekly use of alcohol (P < .001; table 2). Associations of anti-HCY with a history of pregnancy (P = .03) and with laboratory evidence of vaginal trichomonas infection (P <. 001) were also noted. Factors not significantly associated with anti-HCV in males or females included laboratory and clinical evidence of genital herpes. Chlamydia. and gonococcal infections. age of first intercourse. and history of crack cocaine use. Histories of exchanging sex for money or drugs were also associated with anti-HCV among females before we removed patients with a history of injection drug use from the analysis."
http://www.natap.org/2006/CROI/CROI_49.htm
http://www.natap.org/2003/april/042103_7.htm........."..HCV seroprevalence was 89.7% among injection drug users, compared with 10.5% among noninjection drug users.. Women in HERS who did not inject drugs may have acquired HCV sexually.. The 10.5% HCV seroprevalence among noninjection drug users is much higher than that in the general population, which might be explained by higher numbers of sex partners and a higher prevalence of HCV infection in sex partners among women included in HERS"
Women & HCV.....http://www.natap.org/2002/june/060302_1.htm
In the Hopkins Study: Studies that have found little or no transmission of HCV to sex partners differ from ours in that they evaluated longterm, monogamous sex relationships.......Ninety-five percent of patients in our investigation were African-American.......even after excluding injection drug users, the rate of anti-HCV was 13.3% among persons attending the Johns Hopkins Emergency Department and 9.6% among patients visiting STD clinics........This investigation provides some evidence that sexual transmission of HCV occurs among patients attending the Baltimore STD clinics. Females with anti-HCV-positive sex partners were 3.7 times more likely to have anti-HCY than females with anti-HCY-negative sex partners.......frequent in male and female patients who had greater numbers of sex partners (even after adjusting for age and other associated factors), and anti-HCY was associated with other sexually transmitted diseases, such as HIY and Trichomonas infections.......Males whose sex partners were anti-HCV positive were not more likely to have anti-HCV than those whose sex partners were anti-HCV negative. This observation may mean that female-to-male transmission of HCV is less efficient than from male to female, as previously shown for gonococcal, Chlamydia. and HIV infections.......In this investigation, anti-HCV was found in 18% of males with past homosexual exposure; these males were 3.6 times more likely to have anti-HCV than male STD patients with only heterosexual partners. Although this association became nonsignificant when HIV was included in the multivariate model, because of the interrelatedness of male homosexual exposure and HIV, this most likely represents an overadjustment [49]. In a separate logistic regression model inclusive of all factors except HIV, homosexual exposures remained associated with anti-HCV.
---------------------------------
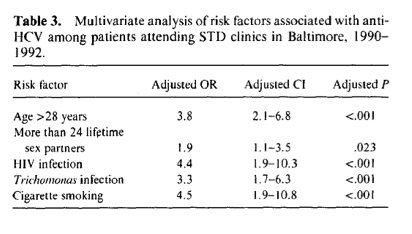
Sexual Transmission of Hepatitis C Virus among Patients Attending Sexually Transmitted Diseases Clinics in Baltimore-An Analysis of 309 Sex Partnerships......among non-IDUs......Anti-HCV was detected in 7% and 4% of non-injection......156 (86% of whom were anti-HCV positive) acknowledged a history of injection drug use and were excluded from further analyses of anti-HCV Anti-HCY was more correlates. An additional 135 persons were not assessed because of missing information on injection drug use (n = 9). HCY status (n = 72). or sex practices (n = 54). drug-using males and females. respectively.......females whose male sex partners were anti-HCY positive were 3.7 times more likely to have anti-HCY (10.2%) than those whose sexual partners were anti-HCY negative (3.0%; P= .039).
1.
The Journal of Infectious Diseases 1995
David L. Thomas, Jonathan M. Zenilman, Harvey J. Alter, James W. Shih, Noya Galai, Anthony V. Carella, and Thomas C. Quinn
Division of Infections Diseases. Johns Hopkins University School of Medicine. Department of Epidemiology. Johns Hopkins University School of Public Health. and Baltimore City Health Department. Baltimore; Department of Transfusion Medicine and Laboratory of lnununoregulation. National Institute ofAllergy and Infectious Diseases. National Institutes ofHealth. Bethesda. Marvland
"The basis for the higher prevalence of anti-HCV found in inner-city populations in the United States is unclear. In Baltimore, anti-HCY has been reported among 18% of patients attending the Johns Hopkins Emergency Department [27], 15%of patients attending the STD clinics [9], 9%of females attending the Johns Hopkins prenatal clinics [28], and 88% of participants in a study of injection drug use [6]. Injection drug use is a proven route of HCY transmission and contributes to the increased prevalences of anti-HCV. However, even after excluding injection drug users, the rate of anti-HCV was 13.3% among persons attending the Johns Hopkins Emergency Department and 9.6%among patients visiting STD clinics.....
.........Nonetheless, because of the high prevalence of anti-HCV among non-injection drug-using STD patients, the association of anti-HCV with having a greater number of sex partners and, for females, with having an anti-HCV-positive male partner, and the similarities in HCV RNA sequences between HCY-positive females and their sex partners, we do not believe that unacknowledged injection drug use fully explains the high prevalence of anti-HCV observed in the STD population. Rather, in this setting, sexual transmission of HCV appears to contribute to the high rates of HCV. Prospective studies of sex partners attending STD clinics and of other high-risk urban populations are needed to further clarify the high rates of HCV found among inner city residents in the United States"
Abstract
The prevalence of antibodies to hepatitis C virus (anti-HCV), the behavioral and laboratory derived risk factors for anti-HCV, and the quantity and homology of HCV RNA were assessed among 1039 non-injection drug-using sexually transmitted disease (STD) patients representing 309 sex partnerships. Thirty-seven (7%) of 555 males and 19 (4%) of 484 females had anti-HCV. In logistic regression analyses, factors associated with anti-HCV included age (P < .001), greater numbers of lifetime sex partners (PP = .023), human immunodeficiency virus infection (P < .001), Trichomonas infection (P < .001), cigarette smoking (P < .001), and male homosexual exposure (P = .012). Among couples, females whose sex partners were anti-HCV positive were 3.7 times more likely to have anti-HCV than females whose sex partners were anti-HCY negative (P = .039). The proportion of RNA homology between anti-HCY positive females and their male partners (94%) was higher than among randomly selected patients (82%). Sexual transmission of HCY may contribute to the high prevalence of anti-HCY reported in urban settings.
-------------------------------------------------
In 2006, this published study in Journal of Infectious Diseases the authors were perhaps the 1st to report increased HCV incidence among HIV+ MSM, and discuss reasons why this might be occurring.
Increase in HCV Incidence among Men Who Have Sex with Men in Amsterdam Most Likely Caused by Sexual Transmission
J Infect Dis. (2007)
Thijs J. W. van de Laar,1 Akke K. van der Bij,1 Maria Prins,1,3 Sylvia M. Bruisten,1,3 Kees Brinkman,7 Thomas A. Ruys,6 Jan T. M. van der Meer,3 Henry J. C. de Vries,2,4 Jan-Willem Mulder,8 Michiel van Agtmael,9 Suzanne Jurriaans,5 Katja C. Wolthers,5 and Roel A. Coutinho1,3,10
1Department of Infectious Diseases and 2Sexually Transmitted Infections Outpatient Clinic, Health Service Amsterdam, Departments of 3Internal Medicine (Section of Infectious Diseases, Tropical Medicine and AIDS), 4Dermatology, and 5Medical Microbiology, and 6National AIDS Trial and Evaluation Center, International Antiviral Therapy and Evaluation Center, Academic Medical Centre/University of Amsterdam; 7Department of Internal Medicine, Onze Lieve Vrouwe Gasthuis; 8Department of Internal Medicine, Slotervaart Hospital; and 9Department of Internal Medicine, Section of Infectious Diseases, Vrije Universiteit Medical Center, Amsterdam; and 10Centre for Infectious Disease Control, National Institute for Public Health and the Environment, Bilthoven, The Netherlands
Discussion
A rapidly increasing number of case reports from Europe [11, 12] the United States [13], and Canada [14] have suggested recent outbreaks of sexually transmitted HCV among HIV-1-infected MSM [15-17]. To our knowledge, this article is the first to suggest an increase in HCV incidence over time among HIV-positive but not HIV-negative MSM on the basis of 20 years of prospective longitudinal cohort data. Additional phylogenetic analysis of HIV/HCV-coinfected cohort and hospital cases strongly supports the presence of an MSM-specific transmission network, probably sexual, in Amsterdam.
As in London, Paris, and Rotterdam [11, 17, 27], phylogenetic analysis of incident HCV infections among MSM in Amsterdam showed at least 3 distinct clusters of independent cocirculating HCV lineages; 83% of Amsterdam MSM infected after 2000 harbored a strain present in at least 1 other MSM in the study. This degree of clustering is typical of a common source of infection and confirms sustained HCV transmission among HIV-positive MSM over the past few years. The low evolutionary distances among recent HCV isolates, the shift in genotypes over time (1b and 3a before 2000 vs. 1a and 4d after 2000), and the lack of clustering among isolates predating 2000 all imply a recent spread of HCV that was absent before 2000. The fact that multiple introductions of different HCV genotypes took place suggests behavioral change in MSM rather than evolution of the virus into a variant that is more easily transmitted.
No sexual transmission of HCV has been reported in recent HCV screenings among MSM in Seattle (n=833) [28] and Montreal (n=1085) [4]; 35 (85%) of 41 infections could be traced to IDU. However, persons screened were HIV-negative MSM, a subpopulation almost unaffected by the current HCV outbreaks [29]. Moreover, HCV screening stopped before the onset of the HCV outbreaks reported in Europe. By contrast, Amsterdam MSM infected after 2000 reported no IDU, and, like Parisian MSM [11], they carried MSM-specific HCV strains that differed significantly from those circulating among IDUs. Thus, IDU as an underlying mechanism of HCV transmission among Amsterdam MSM is highly unlikely. In a Swiss cohort study among HIV-infected MSM, increased HCV incidence was associated with sexual risk-taking and inconsistent condom use, especially in younger MSM [5]. Most likely, the current outbreak of HCV is triggered by the increase in sexual risk-taking observed among MSM after the introduction of HAART [20, 21].
How and when might sexual transmission of HCV occur? Among persons infected with HCV alone, >95% intermittently shed HCV RNA in saliva [30], and 42% of HIV/HCV-coinfected subjects shed HCV RNA in semen [31]. The rare event of sexual transmission among MSM, however, suggests that the rectal mucosa is relatively resistant to HCV infection. HIV-1 infection facilitates HCV transmission by increasing both (1) viral infectiousness due to higher viral loads, and (2) viral susceptibility through its detrimental effects to the gastrointestinal-associated immune system [32, 33]. Moreover, ulcerative STIs can enhance the sexual transmission of both HIV-1 and HCV [8, 34] by creating a portal of entry through the mucosal barrier. Quite possibly, HIV infection in itself makes the host vulnerable to HCV infection in combination with the above [35]. The observation that ulcerative STIs facilitate HCV transmission infection is consistent with the upsurge in STIs among MSM in countries where HAART is generally available [19, 20] and the high number of ulcerative coinfections reported by MSM with acute HCV infection [16, 17, 27]. However, STIs and sexual risk-taking have no higher incidence now than in the early 1980s, when the sexual transmission of HCV seemed absent. What else has changed?
First, current outbreaks of HCV coincide with the recent epidemic of LGV, a "new" STI that manifests as anorectal ulcers, hemorrhagic proctocolitis, and hyperplasia of intestinal and perirectal lymphatic tissue. Both HCV and LGV are strongly associated with a positive HIV status [36], and, in the Netherlands, acute HCV is reported among MSM coinfected with LGV [27]. This suggests a potential role for LGV in the outbreak of HCV reported in the Netherlands.
Second, successive outbreaks of syphilis, rectal gonorrhea, Shigella sonnei infection, LGV, and acute HCV among MSM in major European cities [16, 18, 19, 27, 36, 37] points to the presence of a high-risk "core group" of HIV-positive MSM. This "core group" functions as a reservoir for STIs and is highly susceptible to new or even inefficiently transmitted infections such as LGV and HCV. The popularity of the Internet, trans-European MSM weekend tourism, and organized sex parties may have increased contacts among HIV-positive MSM who share risky sex and are relatively unconcerned about acquiring "treatable" STIs [38]. Lesions in the rectal endothelial lineage could occur especially with rough sexual techniques like anal fisting, the use of toys, and group sex, which favor blood-blood contact and, hence, HCV transmission [14, 17, 27, 39]. In 2005, anal bleeding during sex was reported by 10% of young (<35 years old) HIV-negative participants in the ACS. The use of psychoactive substances, to enhance sexual pleasure and anal sphincter relaxation during receptive anal intercourse or to remove inhibition during rough sexual practices, might also cause mucosal trauma or bleeding, especially when substances are applied anally [40].
The increased HCV incidence among MSM has clinical implications. HIV/HCV coinfection is associated with lower rates of spontaneous HCV clearance, accelerated liver disease, and a less favorable HCV treatment outcome [41]. HCV combination therapy achieves a sustained virological response in only 17% of HIV-positive individuals infected with genotypes 1 or 4 [42]-the HCV strains that affect the majority (91%) of MSM in Amsterdam. In 90%-98% of HIV-negative patients, however, HCV chronicity can be prevented when treatment is initiated during the acute phase of infection [43]. The results of early interferon treatment in HIV-positive MSM infected with HCV genotypes 1 and 4 are conflicting but promising [11, 42, 44].
Several limitations should be considered when interpreting our results. Despite a significant increase in HCV incidence after the year 2000, the low number of HCV incident cases among ACS participants makes it necessary to investigate whether this trend continued after 2002. All HIV-positive ACS participants with extended follow-up after 2002 will be tested for HCV prospectively (or retrospectively at their last ACS visit). The high number of hospital cases collected after 2002, however, indicates sustained HCV transmission among HIV-positive MSM. Second, hospital cases were collected in a clinical setting and not for epidemiological research. Although high rates of concurrent STIs and fisting among hospital case patients are suggestive of permucosal transmission of HCV during sexual contact, a control group is needed to establish whether these rates are significantly higher than those in HIV-positive MSM without HCV coinfection. Additionally, HIV-positive MSM visit a physician more frequently and thus are more likely to receive HCV and STI testing than those who are HIV negative. Testing therefore might have occurred on indication and might have biased our conclusions about HCV and high levels of STIs. ACS data confirm, however, that HCV mainly circulates among HIV-positive MSM, given that no HCV seroconversions were identified among HIV-negative MSM. Third, retrospective data collection based on self-reported behavior may be compromised by report and report bias. However, all 39 recently infected MSM in the study denied IDU, which accounts for the vast majority of new HCV infections in western Europe. Moreover, phylogenetic analysis confirmed a non-IDU-related common source of infection. Data on possible other confounders (e.g., tattooing/piercing, acupuncture, or various nosocomial transmission routes) were not always available, but such general routes of incidental transmission cannot explain the common source of HCV infection observed among MSM. Potential subculture-related confounders like intranasal/anal drug and enema use need further investigation.
Despite these limitations, the present results indicate that HCV appears to be emerging as an STI among a restricted "core group" of HIV-positive MSM who practice risky sex. Given the absence of clear clinical symptoms during acute HCV infection, a significant proportion of the HCV epidemic may still remain unnoticed. Routine HCV screening among HIV-positive MSM is needed to identify the magnitude and character of the current epidemic and to enhance treatment. Although the exact biological mechanism of transmission has yet to be determined and should be examined in a prospective cohort or retrospective case-control study, targeted prevention can deter further spreading and increase awareness in HIV-positive MSM about the additional risks associated with acquiring serious bloodborne coinfections.
Increase in HCV Incidence among Men Who Have Sex with Men in Amsterdam Most Likely Caused by Sexual Transmission
J Infect Dis. (2007)
Abstract
We retrospectively screened 1836 men who have sex with men (MSM) participating in the Amsterdam Cohort Studies (1984-2003) for hepatitis C virus (HCV) antibodies. HCV incidence was 0.18/100 person-years (PY) in HIV-positive MSM (8/4408 PY [95% confidence interval [CI], 0.08-0.36]) but was 0/100 PY in MSM without HIV (0/7807 PY [95% CI, 0.00-0.05]). After 2000, HCV incidence among HIV-positive men increased 10-fold to 0.87/100 PY (5/572 PY [95% CI, 0.28-2.03]). Additional hospital cases (n=34) showed that MSM in Amsterdam who acquired HCV infection after 2000 reported high rates of ulcerative sexually transmitted infections (59%) and rough sexual techniques (55%), denied injection drug use, and were mainly infected with the difficult-to-treat HCV genotypes 1 (56%) and 4 (36%). Phylogenetic analysis showed 3 monophyletic clusters of MSM-specific HCV strains. The emergence of an MSM-specific transmission network suggests that HIV-positive MSM with high-risk sexual behaviors are at risk for sexually acquired HCV. Targeted prevention and routine HCV screening among HIV-positive MSM is needed to deter the spread of HCV.
Hepatitis C virus (HCV) infection occurs mainly by parenteral exposure, particularly injection drug use (IDU) [1]. Its association with sexual transmission remains controversial. Even in the presence of HIV coinfection, HCV is rarely transmitted by heterosexual intercourse [2]. HCV-serodiscordant partners in long-term monogamous heterosexual relationships show only slightly higher rates of HCV-infection than the general population [3]. Data obtained from men who have sex with men (MSM) are more conflicting [4, 5]. Previous cross-sectional and cohort studies have reported increased HCV prevalence among MSM and have highlighted unprotected anal intercourse, multiple sex partners, rough sexual techniques, and coinfection with HIV-1 and other sexually transmitted infections (STI) as potential risk factors [6-10]. However, these associations between sexual risk factors and HCV did not reach statistical significance and were often confounded by concurrent IDU [4].
A growing number of case reports from Europe [11, 12], the United States [13], and Canada [14] have suggested that HCV is emerging as an STI among HIV-1-positive MSM [15-17]. The availability of highly active antiretroviral therapy (HAART) in western countries has been followed by an increase in sexual risk behavior among MSM and, thereby, increased rates of STIs such as rectal gonorrhea and syphilis [18, 19]. A decrease in the perceived threat of HIV/AIDS appears to have weakened the safe-sex attitudes of MSM. High-risk sexual networks have emerged that are susceptible to the introduction of HCV and other infectious agents for which sexual contact is actually an inefficient route of transmission [20, 21].
Our study investigated HCV prevalence and incidence in a large prospective longitudinal cohort of both HIV-positive and HIV-negative MSM in Amsterdam, the Netherlands, over the course of nearly 2 decades (1984-2003). Additionally, 5 Amsterdam hospitals provided serum samples and data on parenteral and sexual risk behaviors of MSM diagnosed with an acute HCV infection after 2000. Phylogenetic analysis was used to obtain evidence of sexual transmission of HCV in MSM.
Subjects, Materials, and Methods
Study population I-the Amsterdam Cohort Studies (ACS) of homosexual men. The ACS of homosexual men is an open, ongoing prospective study [20]. Recruitment was started in October 1984 and uses "convenience sampling" (advertisements: brochures at the STI clinic and social venues for MSM) and "chain referral sampling" (recruitment by participants among their acquaintances). The local ethical committee approved the ACS, and all MSM participated only after they provided informed consent. Until 1995, the ACS included MSM of all ages with at least 2 male sex partners during the 6 months preceding enrollment, regardless of HIV status. After 1995, participation was restricted mainly to HIV-negative MSM ≤30 years old with at least 1 male sexual encounter during the preceding 6 months. Participants complete a self-administered standardized questionnaire at ACS visits every 6 months (if HIV positive, every 3 months) about personal health and sexual risk behavior, and blood was drawn for HIV testing and storage at -80°C.
Our study included all ACS participants who made at least 2 study visits between October 1984 and January 2003 (n=1836). The last available serum sample predating February 2003 was tested for HCV antibodies using a third-generation commercial MEIA system (AxSym HCV version 3.0; Abbott). HCV-positive test results were confirmed by TMA nucleic acid amplification (Versant; Bayer Healthcare) and/or Immunoblot (Chiron RIBA HCV 3.0 SIA; Ortho-Clinical Diagnostics). When a participant was found to be HCV positive, his sample from ACS entry and succeeding visits were tested to determine whether he was positive at entry or, if not, between which visits HCV seroconversion occurred.
Study population II-hospital case reports. In the Netherlands, the law requires that all acute HCV infections (until 2003, all newly diagnosed HCV infections) be reported to the local health service to enable source tracing and to prevent the further spread of HCV. Through review of these reports and through contact with physicians treating HCV in 5 major hospitals, including the STI clinic in Amsterdam, we identified 34 MSM who acquired HCV after the year 2000 and for whom blood was stored. Of these, 32 men had a confirmed seroconversion of anti-HCV antibodies and detection of HCV RNA after a previously HCV-negative sample. The other 2 men lacked a previous HCV-negative sample but had been diagnosed with acute HCV infection (jaundice, elevated liver enzymes, and exclusion of hepatitis A and B) by their physicians. To identify the probable route of transmission, additional data on possible IDU, blood-blood contact, STI coinfection, and sexual risk behaviors were obtained by review of medical charts and notification reports. For all patients, informed consent had been obtained by their treating physician.
Results
Study population-ACS. The study population included 1836 MSM with ≥2 ACS visits between October 1984 and January 2003. The mean ± SD age at entry was 31.8±8.1 years, 92% were of northwest European nationality, and 28% had at least a college degree. Median follow-up time in the ACS was 6.3 years (interquartile range [IQR], 2.8-11.2 years). These 1836 men made a total of 44.107 visits at a median interval of 91 days (IQR, 49-162 days). Most (89%) identified themselves as (almost) exclusively gay. Reported rates of risk behavior were high; at ACS entry, 69% had had >20 sex partners during the preceding 5 years, 28% tested positive for HIV-1, and 17% had a history of syphilis. Only 4 ACS participants (0.2%) reported IDU.
HCV prevalence-ACS. At study entry, 24 (1.3%) of 1836 MSM tested positive for HCV antibodies. Of these, 2 had injected drugs and 1 had received a blood transfusion before the introduction of standard HCV screening of blood donors (1991). HCV-positive MSM were older than HCV-negative MSM (36.1 vs. 31.7 years; P=.008) and were more likely to have a history of IDU (10% vs. 0.1%; P<.001) or to test positive for HIV (54% vs. 27%; P=.005). In multivariate analysis, only a history of IDU (odds ratio [OR], 60.5 [95% confidence interval [CI], 7.4-495]) and HIV positivity (OR, 4.1 [95% CI, 1.6-10.4]) were significantly associated with a positive HCV status at ACS entry. No other factors reflecting sexual risk behavior-such as the number of partners or a history of syphilis-were associated with HCV prevalence (table 1).
HCV incidence-ACS. Between October 1984 and January 2003, 8 MSM seroconverted for HCV and 5 seroconverted after December 2000; all 8 HCV seroconverters were HIV positive; their median age was 38.7 years (IQR, 34.5-42.6 years). Overall, HCV incidence was 0.07/100 PY (95% CI, 0.03-0.13/100 PY); it was 0/100 PY among HIV-negative men (95% CI, 0-0.05/100 PY) and 0.18/100 PY among HIV-positive men (95% CI, 0.08-0.36/100 PY). The HCV incidence among HIV-positive MSM increased significantly after the year 2000, compared with previous years-0.87/100 PY in 2000-2003 (95% CI, 0.28-2.03/100 PY), compared with 0.08/100 PY in 1984-1999 (95% CI, 0.02-0.23/100 PY; P=.001) (table 2). The total number of HCV seroconverters in the ACS was too small to allow for a statistical analysis of sexual risk factors.
Hospital case reports. For the 34 participants with hospital stays, the median duration between the last HCV-negative test and the first HCV-positive test was 201 days (IQR, 98-472 days). All but 1 subject were HIV positive at the moment of HCV seroconversion; their median age was 40.0 years (IQR, 35.3-46.6 years). Medical chart reviews, performed for 27 of 34 hospital cases, found no IDU during the 6 months preceding seroconversion; 1 MSM reported possible blood-blood contact after assisting at an accident involving a presumed drug user. Sexual risk behaviors among the hospital subjects was high; at least 16 (59%) of 27 had had 1 or even 2 ulcerative coinfections within the 6 months preceding HCV seroconversion: lymphogranuloma venereum (LGV; 10/27), syphilis (8/27), and/or herpes simplex virus 2 (3/27). Other STIs mentioned were rectal gonorrhea (10/27), acute hepatitis B virus (2/23), rectal non-LGV Chlamydia trachomatis (3/27), and genital warts (3/27). Of the 27 men, 15 (55%) practiced anal fisting. In total, 23 (85%) of 27 reported either an ulcerative coinfection and/or anal fisting; 2 of the remainder reported neither IDU, blood-blood contact, nor high-risk sexual contact.
Genotyping and phylogenetic analysis. All HCV seroconversion samples (8 ACS participants) and samples obtained at the time of HCV diagnosis (34 hospital case patients) tested positive for HCV RNA, as did 15 (63%) of 24 MSM who were positive at ACS entry. Table 3 shows the HCV genotype distribution among Amsterdam MSM infected before and after 2000. HCV infections predating 2000 were mainly of subtypes 1b and 3a (56%), whereas, after 2000, subtypes 1a and 4d predominated (85%).
Figure 1A represents a phylogenetic tree of 56 partial HCV NS5B sequences obtained from the Amsterdam MSM participating in the study. MSM who acquired HCV infection after 2000 had a high degree of clustering, showing 3 monophyletic clusters (cluster I-III) and, additionally, 3 pairs (a-c) of nearly identical HCV strains. When MSM who contracted HCV before 2000 were compared with those who contracted HCV after 2000, 0 of 17 HCV isolates from before 2000 and 33 (85%) of 39 HCV isolates from after 2000 had at least 1 nearly identical HCV isolate. The 2 MSM (S11 and S39) with recent HCV infection who reported no IDU, blood-blood contact, or high-risk sexual contact did not have a clustered or paired HCV isolate, suggesting unrelated routes of transmission. The MSM (S18) who assisted in the accident with the presumed IDU, who also reported unprotected sex, was part of cluster III.
The largest cluster (III) includes 14 MSM infected with HCV genotype 4d. To address whether their HCV strains resemble strains circulating in other risk groups, we constructed a phylogenetic tree (figure 1B) of all 51 HCV genotype 4 sequences in our database [26]: from MSM in the present study (n=16), IDUs in Amsterdam (n=17) and other European cities (n=10), and Dutch blood donors (n=8). For genotype 4, but also for genotype 1a (data not shown), different risk groups can be distinguished on the basis of phylogeny. Although MSM and IDUs were both infected with strains of HCV subtype 4d, the 14 MSM (cluster III) who contracted HCV infection after 2000 form a separate cluster within this subtype (bootstrap value, 91). Sequences obtained from 2 MSM who acquired HCV infection before 2000 (P2 and P7) were interspersed with sequences of IDUs from Amsterdam; both of them had reported IDU. Interestingly, geographic clustering was observed of IDUs from Amsterdam and those from southern Europe.
|
|
| |
| |
|
|
|