| |
The patent system, Hepatitis C drugs not reaching poor - "access to drugs will depend on the development of cheap diagnostics"...... "Major donors have not yet signalled their intention to pay for the drugs"..... "It's possible it's really going to be up to the countries to fund their own programmes"......
|
| |
| |
"Major donors have not yet signalled their intention to pay for the drugs. Geneva-based UNITAID welcomed the new WHO guidelines, but noted that access to drugs will depend on the development of cheap diagnostics. "It's possible it's really going to be up to the countries to fund their own programmes, which is really going to be a limiting factor," says Wiktor."
Univ of Penn, Wharton School of Business (see full article below): "the patent system..... patents exist because the government permits firms to make original discoveries and get a temporary monopoly on them...... the patent system is controlled by the same people who are complaining about Sovaldi's price. "I find it a little ironic that members of Congress are giving speeches about how terrible this is," he says. "If we don't want companies charging high prices, or if someone thinks it's unethical, then Congress should change the patent system."..... the blame for high drug prices should be placed on the U.S. health care system instead ...... The government could, for example, offer a large financial prize to companies that develop groundbreaking cures...... existing patent system allows companies to recoup their high R&D costs, which then encourages them to search for more innovations. The commonly quoted figure is that it costs $1 billion to bring a new drug to market, though some recent estimates have put the real cost at $5 billion. "There's no doubt the process of clinical trials is expensive.... The motivation for doing [them] is high profits at the end of the road," Pauly says. "So I find the ethical discussion unrealistic, because it assumes you can make these value judgments after the fact, when really you ought to make an ethical judgment about the patent system as a whole."......"
Nature | News
Hepatitis C drugs not reaching poor
Treatment guidelines for virus highlight challenge of paying for expensive drugs in low-income countries.
· Ewen Callaway
15 April 2014

Saurabh Das/AP Photo
Campaigners in India have been calling for access to cheap hepatitis C treatments.
The publication last week of the first treatment guidelines for hepatitis C virus (HCV), and the advent of drugs that can cure most infections of the virus, have left public-health researchers with a touch of deja vu.
Three decades after wrestling to lower the cost of AIDS drugs (prices fell from about US$10,000 per patient per year in the 1990s to less than $100 in the mid-2000s), they are once again asking how expensive life-saving medicines can be made affordable for patients.
"We've been here before," says Stefan Wiktor, head of the global hepatitis programme at the World Health Organization (WHO) in Geneva, Switzerland, and lead author of the guidelines (see go.nature.com/bwhguw). And a similar success could follow, adds Andrew Hill, a pharmacologist at the University of Liverpool, UK, "if there's the political will to do it".
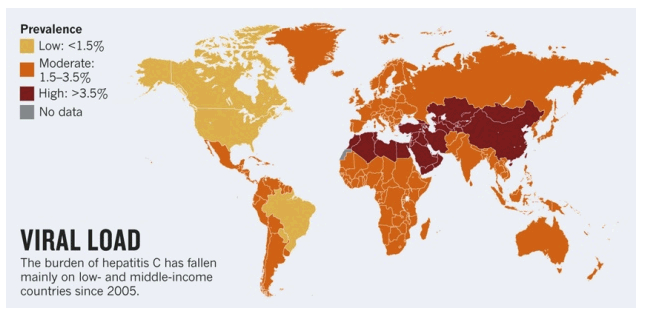
Discovered in 1989, HCV infects an estimated185 million people worldwide, more than 80% of whom live in low- and middle-income countries (see 'Viral load'). Around 15% of Egypt's population, for example, is infected. The blood-borne virus is a leading cause of liver cancer and cirrhosis.
Most of these HCV cases had been ignored, mainly because available treatments did not work well and were relatively expensive. The combination of an antiviral drug, ribavirin, and an immune-system modulator called interferon cures less than 50% of patients after a year, while causing fatigue, nausea and even depression.
But that is now set to change. First, a new breed of HCV medicine that directly attacks the virus - approved by US and European regulators in 2011 - pushed cure rates to around 75%, although it did not fully eliminate the need for the ribavirin-interferon therapy. And in recent months, regulators have approved two drugs that can cure most HCV infections after three months and do not require interferon in many cases.
Source: K. M. Hanafiah et al. Hepatology 57, 1333-1342 (2013)
But the pricing of these new drugs has raised eyebrows even in Western countries, where private health insurers and public health-care systems tend to bear the costs of drugs. Sofosbuvir, made by Gilead Sciences in Foster City, California, debuted last December at $1,000 a pill - $84,000 for a 12-week course. And Janssen Pharmaceuticals, based in Beerse, Belgium, has priced its simeprevir at $66,000 for the same treatment period. Those prices are a non-starter for low- and middle-income countries. But it is not clear how the price will fall to a level that such countries can afford, says Wiktor.
Antiretroviral drugs that keep HIV in check reached less-wealthy countries only after pressure from non-governmental agencies, academics and patients. Generic-drug manufacturers showed that they could make the medicines cheaply, and funders such as the Global Fund to Fight AIDS, Tuberculosis and Malaria stepped in to pay for them. Some countries, such as Brazil and Thailand, even issued 'compulsory licences' to manufacture generic versions of HIV drugs, effectively ignoring patents.
These options will be open to HCV drugs, says Wiktor. "Competition and generic production really are the keys to reductions in prices."
In March, Gilead announced a deal with Egypt to provide sofosbuvir at $900 for a 12-week course. "I think it's an excellent first step, but it's only a first step," says Hill. He adds that treating every case of HCV in Egypt would far outstrip the country's annual health-care budget of around $4.5 billion.
Gregg Alton, executive vice-president of corporate and medical affairs at Gilead, says that the company is in negotiations with several Indian manufacturers to produce a generic version of sofosbuvir. But, according to a report from the Paris-based charity Medicins du Monde, the generic drug would be available to 60 mostly low-income countries, and not to many middle-income countries with high HCV burdens. Meanwhile, the Initiative for Medicines, Access & Knowledge, a non-profit group in New York, has filed a lawsuit with India's patent office seeking to prevent Gilead from receiving a patent for sofosbuvir.
Cheap generic HCV drugs are within reach. In an analysis published in February in Clinical Infectious Diseases, Hill and his team compared the cost of producing generic HIV medicines and applied this analysis to the potential cost of HCV drugs (A. Hill et al. Clin. Infect. Dis. 58, 928-936; 2014). They estimate that generic-drug manufacturers should be able to produce the pills at $100-250 for a 12-week course. But even at those prices generic manufacturers won't manufacture HCV medicines until there is large demand, Hill says.
Major donors have not yet signalled their intention to pay for the drugs. Geneva-based UNITAID welcomed the new WHO guidelines, but noted that access to drugs will depend on the development of cheap diagnostics. "It's possible it's really going to be up to the countries to fund their own programmes, which is really going to be a limiting factor," says Wiktor.
Jennifer Cohn, medical director of the drug-access programme for the charity Medecins Sans Frontieres in Geneva, notes that middle-income countries face the highest hurdles. Increasingly, inter-national donors are withdrawing support from these countries, and drug makers such as Gilead are likely to see them as untapped markets for their products and thus exclude them from generic licensing deals.
Cohn says that these countries should consider compulsory licensing, just as some countries did for HIV drugs. This practice is sanctioned by the World Trade Organization in certain cases - for example, to ensure affordable medicine prices - to protect public health.
But cost is not the only barrier. Most countries' health systems are not equipped to widely diagnose the disease or deliver the drugs. Wiktor says that it will be a missed opportunity if the global health community does not work out how to get the drugs to patients. "People are dying from liver cancer, from cirrhosis," he says, "and these deaths can be prevented."
Nature (17 April 2014)
-------------------
Sovaldi: Who's to Blame for the $1,000 a Day Cure?
http://knowledge.wharton.upenn.edu/article/sovaldi-whos-blame-1000-day-cure/
Apr 16, 2014
When Foster City, Calif.-based Gilead Sciences was developing its hepatitis C treatment, Sovaldi, the medical community was wowed by the 90% cure rate the drug racked up in clinical trials. Then Sovaldi hit the market early this year with an eye-popping price tag: $84,000 for a 12-week course of treatment, which amounts to $1,000 a day. Now insurers and patient advocacy groups are up in arms about what they say is a product that is vital to helping the estimated three million Americans with the disease, but is wildly unaffordable.
The Sovaldi controversy reached fever pitch in March, when U.S. Representative Henry Waxman, a Democrat from California, and several of his colleagues in Congress wrote Gilead CEO John Martin a searing letter demanding to know why the drug costs so much. Shortly thereafter, Steven Miller, the chief medical officer of St. Louis, Mo.-based Express Scripts, said his company was putting together a coalition to refuse to cover Sovaldi after lower-priced competitors hit the market, which could happen later this year. And administrators for some Medicaid plans are so worried they won't have enough resources to pay for any of the new treatments that they are pleading for financial assistance from their state legislators.
It may be tempting to pronounce Gilead guilty of prioritizing profits over patient need, but many Wharton experts say the blame for high drug prices should be placed on the U.S. health care system instead. "Companies obviously have an obligation to their shareholders to maximize profits," says Patricia Danzon, Wharton professor of health care management. "That generally means doing the best that you can within the reimbursement environment that exists in any particular country. In the U.S., we have established a system of reimbursement for pharmaceuticals that unfortunately puts absolutely no limits on the prices that companies can charge."
Gilead declined to be interviewed by Knowledge@Wharton but said in a statement: "We believe the price of Sovaldi reflects the value of the medicine. Sovaldi represents a significant therapeutic advance over other available therapies, as it has shortened the duration of treatment to as little as 12 weeks and has reduced or completely eliminated the need for interferon injections, depending on the patient's genotype." The company added that the price of Sovaldi plus interferon and ribavirin (which are used in conjunction) is consistent with that of protease inhibitors that are often used to treat hepatitis C.
Gilead has also argued that by curing hepatitis C, Sovaldi should actually save the health care system money over time by reducing long-term complications of the disease, such as liver failure and the need for liver transplants.
"Generally we do not adjust prices in a market-based system based on the number of consumers. Price is more related to value." -Patricia Danzon
There's little doubt, Danzon notes, that the drug will help a lot of people, and therein lies the crux of the debate. This is far from the first astronomically priced drug on the market - cancer drugs are routinely priced at tens of thousands of dollars per course of treatment, as are products aimed at "orphan" diseases, which affect small patient populations. But in those cases, "the argument is made that the very high prices are justified because you have a small number of patients and the cost of [research and development] needs to be covered," Danzon says. "The problem occurs when you get a very large disease class like hepatitis C. Should the argument then reverse - i.e., that the price has to be lower because it's a very high volume of people? Generally we do not adjust prices in a market-based system based on the number of consumers. Price is more related to value."
In the U.S., Danzon adds, value is often defined as whatever price the market will bear. That's not the case in other countries, though. Gilead has reported that in the United Kingdom, it charges $57,000 for a 12-week course of Sovaldi. She points out that average wages are lower in the U.K. than they are in the U.S., and that drug pricing is largely controlled by the National Institute for Health and Care Excellence (NICE), which evaluates every new drug and makes rulings as to whether the country's health care system should pay for it. "Charging less [for Sovaldi] in the U.K. is consistent with differential pricing and with the rules NICE has set," Danzon says.
In the U.K. and virtually every other developed country, there is a mechanism for setting limits on what drugs should cost, which is often defined as a willingness to pay a certain amount for a "quality adjusted life year" (QALY) saved, according to Danzon. QALY is a formula that takes into account not just total life expectancy that will be gained by a particular health intervention, but also the quality of life patients are likely to enjoy during those years, based on their health status. Using QALY and other related measures "is an incentive for innovation," Danzon says. "Any new product that delivers benefits can command a higher price that is commensurate with the degree of innovation defined in terms of health gain. But it's not just a blank check."
The blank check issued to Gilead and its eventual competitors in the U.S. could have dire financial consequences, some insurers argue. Express Scripts estimates that spending on hepatitis C drugs will jump 1,800% from 2014 through 2016, possibly hitting $300 billion - which CMO Miller has told the press is more than what the U.S. spends on all other drugs combined.
If anyone in the U.S. can put pressure on drug companies to lower prices, it's the insurers, Danzon says. But there is no guarantee that reactionary measures put in place by Express Scripts or any other benefits provider will actually work. "It is difficult for any one insurer to do it because they compete with other insurers in the marketplace," she notes. "That's why Express Scripts is going out and trying to form a coalition. But any one of those payers will say over and over again, 'We do not have the market power to do this.' If the physicians prescribe it, they feel they have to pay."
A Tricky Patent System
In January, researchers in Italy published a study concluding that a three-drug treatment for hepatitis C that includes Sovaldi is cost-effective based on a number of measures, including QALY. Wharton health care management professor Mark V. Pauly believes such evidence may be getting lost in all the griping over the drug's price.
More importantly, he says, the debate has overlooked one key aspect of the health care system that would have to be changed in order for the U.S. to take more control over drug pricing: the patent system. Sovaldi "is a patented drug. And patents exist because the government permits firms to make original discoveries and get a temporary monopoly on them," Pauly notes. "The general idea is to allow companies to charge monopoly prices for a period of time to motivate innovation. That's what Gilead is doing - they're charging the monopoly price that is presumably based on what they think the market is willing to pay for their product."
Pauly points out that the patent system is controlled by the same people who are complaining about Sovaldi's price. "I find it a little ironic that members of Congress are giving speeches about how terrible this is," he says. "If we don't want companies charging high prices, or if someone thinks it's unethical, then Congress should change the patent system."
"If we don't want companies charging high prices, or if someone thinks it's unethical, then Congress should change the patent system." -Mark V. Pauly
That would be a huge change - but not impossible, Pauly adds. The government could, for example, offer a large financial prize to companies that develop groundbreaking cures, rather than giving them multi-year monopolies via the patent system. "There's some general sentiment that the patent system may not actually encourage innovation as much as a system of open and free competition would," he notes. "Some economists argue that you could give companies a prize and then order them to sell the drugs at cost thereafter."
But the flipside to that argument is that the existing patent system allows companies to recoup their high R&D costs, which then encourages them to search for more innovations. The commonly quoted figure is that it costs $1 billion to bring a new drug to market, though some recent estimates have put the real cost at $5 billion. "There's no doubt the process of clinical trials is expensive.... The motivation for doing [them] is high profits at the end of the road," Pauly says. "So I find the ethical discussion unrealistic, because it assumes you can make these value judgments after the fact, when really you ought to make an ethical judgment about the patent system as a whole."
Managing Corporate Reputation
Inherent in the debate over Sovaldi's price is one key question: How important will it be for Gilead's corporate reputation to respond to the criticism? Arnold J. Rosoff, an emeritus professor of legal studies and business ethics at Wharton, says finding the right balance between being a good corporate citizen and earning good returns for shareholders can be difficult. "A company that has invested tens of millions of dollars developing a drug will want to get that money back," Rosoff notes. "You're walking a fine line between people who need the drug and can't afford to pay full price for it and everybody's desire to have drug companies continue to forge ahead."
Gilead said in its statement to K@W that the company has established "one of the most comprehensive patient assistance programs in the industry to help ensure cost is not a barrier to Sovaldi for patients in the U.S." The components of that system include providing coupons to bring the co-pay for the drug as low as $5 per month, paying the entire cost of the drug for some patients who lack insurance, and providing financial support to an independent non-profit organization that offers assistance for patients who cannot cover their out-of-pocket medication costs.
"A company that has invested tens of millions of dollars developing a drug will want to get that money back." -Arnold J. Rosoff
This Sovaldi controversy doesn't just affect the U.S. The World Health Organization estimates that more than 150 million people worldwide suffer from chronic hepatitis C and that up to 500,000 people die from complications of the disease every year. In developing countries that can't afford the drug, Gilead has shown some willingness to bend on price. At a recent medical meeting in London, the non-profit Doctors Without Borders confirmed it will be able to obtain the drug at a cost of $900 for the 12-week course for use in countries such as Kenya, Mozambique, Myanmar and India.
Gilead does have a history of making its innovations obtainable in poor countries: In 2011, it was the first company to form a deal with the Medicines Patent Pool, a United Nations-backed organization founded with the goal of increasing access to HIV medicines. The firm donated the patents to four of its HIV medicines to the organization so inexpensive versions could be manufactured and distributed in poor countries.
According to Rosoff, fellow drugmaker Merck proved the value of making innovations affordable for those who need them the most with its drug to treat river blindness, a common disease in Africa. Former Merck CEO Roy Vagelos decided in the 1980s to give the drug away in Africa. "The drug had already made its mark and returned for Merck the majority of the development costs," Rosoff notes. "At that point, all you're talking about is the cost of manufacturing and distributing the drug. In third-world countries, a company can be very generous without undercutting its home market."
Vagelos has long preached the value of the positive publicity that came out of the river blindness program. "All the drug companies understand that example and would like to bathe in the same glow that Merck did," Rosoff says. "But the question is, practically [speaking], what are the pros and cons of doing it? You want to grab all the glory, but at a cost you can afford."
In the meantime, the controversy over Sovaldi's price has not hurt its sales. Gilead has pulled off one of the most successful drug launches of all time, and analysts are now predicting Sovaldi will hit anywhere from $1 billion to $2 billion in first quarter sales - putting it well on track to surpass the top-selling drug of all time, Pfizer's cholesterol blockbuster, Lipitor.
|
|
| |
| |
|
|
|