| |
Effects of a Sustained Virologic Response on Outcomes of Patients With Chronic Hepatitis C
|
| |
| |
Download the PDF here
Published Online: June 13, 2011
CLINICAL GASTROENTEROLOGY AND HEPATOLOGY 20
VIVIAN NG* and SAMMY SAAB*
Department of Medicine,
Department of Surgery, David Geffen School of Medicine at University of California at Los Angeles, Los Angel
For patients with chronic hepatitis C virus infection, the goal of antiviral therapy is to achieve a sustained virologic response (SVR). We review the durability of the SVR and its effects on liver-related mortality, hepatic decompensation, and the development of hepatocellular carcinoma. We performed a systematic review of the effects of the SVR on liver-related hepatic outcomes and found the SVR to be durable (range, 98.4%-100%). An SVR reduced liver-related mortality among patients with chronic hepatitis C (3.3- to 25-fold), the incidence of hepatocellular carcinoma (1.7- to 4.2-fold), and hepatic decompensation (2.7- to 17.4-fold). An SVR can lead to regression of fibrosis and cirrhosis, and has been associated with a reduced rate of hepatic decompensation, a reduced risk for hepatocellular carcinoma, and reduced liver-related mortality.
Conclusions
The significance of SVR has been shown in the individual studies detailed in this review article. Long-term benefits of SVR have been shown in patients with chronic hepatitis C because SVR has been associated with reducing liver disease progression, development of HCC, and liver-related mortality. These benefits are seen in those with all degrees of liver fibrosis, and the effects are significant even in those with advanced fibrosis. SVR should continue to be the goal in treating patients with chronic hepatitis C because it is the best marker of successful therapy. Future studies of treatment regimens and outcomes should be conducted with aims to maximize the potential of achieving SVR.
Hepatitis C virus (HCV) is estimated to infect more than 170 million individuals worldwide.1 HCV is a leading cause of decompensated cirrhosis, hepatocellular carcinoma (HCC), liver transplantation, and liver-related death. The prevalence of infection is expected to increase as the survival time of the infected population continues to increase.2 It is proposed that the proportion of patients with cirrhosis will double by 2020 in the untreated population.2
Treatment for chronic HCV has been found to be cost effective and is associated with sustained viral clearance in approximately 54%-63% of patients overall.3, 4, 5 A recent 2010 study using a Markov model to compare the cost effectiveness of interferon (IFN) therapy for HCV over 17 years in a cohort of 4000 patients showed that treatment of hepatitis C with compensated cirrhosis resulted in 119 fewer deaths, 54 fewer HCCs, and 66 fewer transplants when compared with nontreated patients.6 Treatment for HCV is associated with a reduced risk of liver disease progression, with sustained virologic response (SVR) or without viral 7, 8, 9
Use of Sustained Virologic Response as a Marker of Successful Hepatitis C Virus Therapy
The goal of antiviral therapy is achieving an SVR, defined as undetectable HCV RNA levels at the end of 6 months of follow-up evaluation after cessation of treatment for chronic hepatitis C.10 Achieving SVR after treatment has been associated with improvements in disease progression, liver histology, health-related quality of life, and a reduced risk of HCC and liver-related 11, 12, 13, 14
Purpose
As more antiviral treatment regimens are developed and SVR is more easily achievable, the importance of understanding the impact of SVR on patient outcomes will become more pronounced. This article reviews the benefits of SVR on liver-related mortality, development of HCC, and liver disease progression and histologic changes stratified by degree of fibrosis.
Methods
Data Sources and Searches
We searched the MEDLINE database for all studies investigating SVR, durability of SVR, and liver-related outcomes for hepatitis C patients. We used combinations of the keywords: "cirrhosis," "liver fibrosis," "liver cancer," "liver malignancy," "hepatic decompensation," "hepatitis C," "durability," "hepatocellular carcinoma," "sustained viral response," and "sustained virological response." We searched all available data from January 1991 through March 2011. We also manually searched references cited in identified articles for additional studies that may have been missed with MEDLINE-assisted strategy.
Study Selection
Two investigators reviewed the contents of 108 abstracts or full-text articles identified from the literature search to determine if they met inclusion criteria. Studies were included if they were published in scientific journals that provided information regarding the benefit of SVR in the development of HCC, liver-related mortality, and hepatic decompensation, along with studies detailing durability of SVR. We included studies that detailed special populations, such as human immunodeficiency virus (HIV) and HCV co-infection, advanced fibrosis, and those with normal serum alanine aminotransferase (ALT) levels. We also searched article reference lists for relevant articles or abstracts. We excluded data with follow-up evaluation of less than 2 years, and also studies with ongoing maintenance therapy, defined as therapy beyond standard acceptable courses (Figure 1
Data Extraction and Quality Assessment
Reviewers abstracted data from the identified studies, and extracted characteristics of each study and its participants. A formal scoring system to rate the study quality of each individual study was not used. Based on inclusion criteria, only studies with a long follow-up period (at least 2 years) were included. Of the studies included, most had cohorts of more than 100 patients, and only 3 cohort studies had fewer than 100 patients studied. Reviewers noted the following as outcomes of interest: rate of SVR, liver-related mortality, development of HCC, and hepatic decompensation. In addition, reviewers noted the degree of fibrosis of the patient population when stratified in the studies.
Data Synthesis
The investigators qualitatively synthesized the included studies and summarized the pertinent results into tables, stratifying the discussion of evidence by similar groups such as across all stages of fibrosis, and for those patients with advanced fibrosis.
Results
Durability of Sustained Viral Response
Sustained viral response is considered to be extremely durable. Yu et al15 described a cohort of patients and evaluated different doses of IFN, they showed no statistical dose-dependent difference in durability. A combined 63 of 64 patients maintained SVR, with a mean follow-up period of 6.81 years. Formann et al16examined 187 patients who had achieved SVR via IFN, IFN and ribavirin, and pegylated (PEG)-IFN and ribavirin. None of the patients had a relapse with a median follow-up period of 29 months.16 In a long-term follow-up study of patients previously enrolled in clinical trials,3, 4, 5, 17, 18, 19, 20, 21, 22 with patients receiving either IFN or IFN and ribavirin, it was found that only 12 of 1343 patients (0.9%) who initially achieved SVR were found to have re-infection during a mean follow-up period of 4.1 years.23 This large cohort of patients included different patient subsets such as those with normal ALT levels, and those with HIV and HCV co-infection. Desmond et al24 also showed this high durability, with 146 of 147 patients maintaining negative HCV RNA levels over a mean follow-up period of 2.3 years. Giannini et al25 studied a cohort of 231 chronic hepatitis C patients with at least 48 weeks of follow-up evaluation after therapy with PEG-IFN and ribavirin, and saw that SVR was maintained in 99% of their cohort (Table 1
Patients With Normal Serum Aminotransferase Levels
It is estimated that approximately 30% of patients with chronic hepatitis C have normal serum ALT levels, and it is believed that their rate of disease progression to cirrhosis is reduced compared with patients with higher ALT levels.26, 27 Gordon et al28 studied 1744 patients with HCV treated with IFN therapy, and 105 patients (6%) had normal serum ALT levels. There was no difference in the SVR rate between patients with normal ALT levels (24.8%) compared with those with increased ALT levels (26.8%). Although most patients with normal ALT levels often have no fibrosis on liver histology, there are some patients with normal ALT levels with advanced fibrosis and cirrhosis on liver biopsy, placing them at increased risk for disease progression, and developing HCC.29 Therefore, the decision to treat with IFN should not be based solely on aminotransferase
Impact of Pretransplant Sustained Virologic Response After Liver Transplantation
In patients with cirrhosis and advanced liver disease, achievement of SVR before liver transplantation can improve outcomes after transplantation. There have been observations that treatment with IFN therapy for HCV before transplantation can be beneficial in preventing HCV recurrence, particularly if SVR is achieved.30 However, duration and dose adherence is limited because these patients have difficulty tolerating IFN-based therapy.31
Everson et al32 followed up 124 patients with advanced cirrhosis who were treated with IFN therapy. Of the 124 patients, 47 patients (37.9%) underwent liver transplantation, and of these 47 patients SVR was achieved in 12 (26%). Of the 4 patients who had achieved SVR before transplantation, none of these patients had recurrence of hepatitis in the long-term follow-up evaluation after liver transplantation. In a separate study by Nudo et al,33 patients who achieved an SVR, defined using a sensitive viral assay, and no evidence of viral recurrence after liver transplantation. The patients who had undetectable viral levels prior to liver transplantation (without achieving an SVR) were seen to have a significantly decreased risk of recurrent infection after liver transplantation when compared to patients with a measurable viral load at time of liver
Human Immunodeficiency Virus/Hepatitis C Virus Co-infection
Patients who are co-infected with HIV and HCV often have a higher rate of liver disease progression, cirrhosis, and HCC development.34, 35 Successful control of HIV infection with highly active antiretroviral therapy can reduce the rate of liver disease progression.36 In a review by Singal et al,37 the SVR rate in HIV/HCV co-infected patients was found to range between 17% and 53%. The pooled SVR rate from 7 randomized controlled trials or prospective cohort studies involving 784 HIV/HCV co-infected patients was 33.3% (range, 27.3%-44.2%) when treated with IFN and ribavirin.38 When measures are taken to increase treatment monitoring and compliance, SVR rates increased significantly and were equivalent to patients with HCV mono-infection. In a retrospective study conducted in a methadone maintenance treatment program of 73 patients with HCV infection, of whom 32% were co-infected with HIV, it was found that HIV/HCV co-infected patients achieved an SVR rate of 43%, and HCV mono-infected patients had an SVR rate of 46%.39
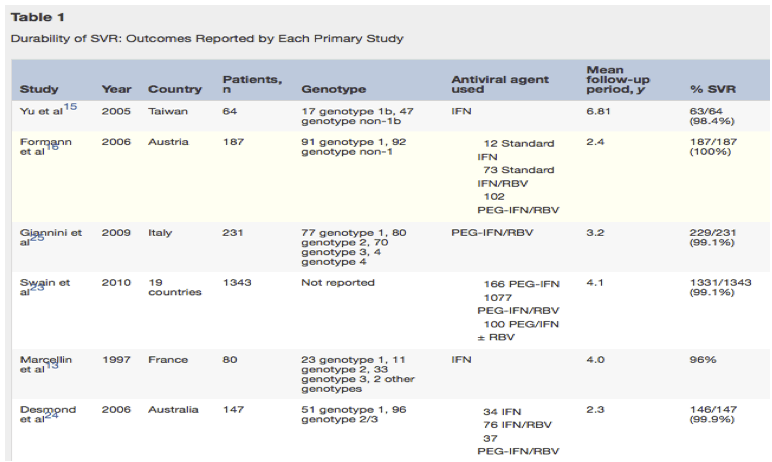
Liver-Related Mortality: All Stages of Fibrosis
Multiple studies have shown a positive effect of SVR on liver-related mortality, regardless of the stage of liver fibrosis (Table 2). Arase et al40 studied a cohort of 500 patients who received IFN therapy for hepatitis C in which 140 patients (28%) reached SVR and looked at long-term outcomes over a follow-up period of 7.4 years. The number of liver-related deaths was decreased significantly in the group that reached SVR, with 2 liver-related deaths of 9 total deaths in the SVR group (22% of deaths, 1.4% of SVR group) when compared with the group that did not attain SVR, with 32 liver-related deaths of 44 total deaths (73% of deaths, 8.9% of SVR group) (risk ratio, 1/0.13; 95% confidence interval [CI], 0.03-0.59; P = .007).40 In a study in Australia, 384 patients were followed up for 9 years after IFN therapy for hepatitis C, and of the 384 patients, there were 25 liver-related deaths. The risk of liver-related death was increased at least 3-fold for patients who did not attain SVR.41 In another retrospective cohort study of 2698 patients who received IFN with follow-up evaluation of more than 6 years, it was seen that the risk of death from liver-related causes was significantly lower for those with SVR than those who were untreated (risk ratio, 0.04; 95% CI, 0.005-0.301; P < .002).12 Yoshida et al42 performed a retrospective study of 2430 patients treated with IFN and 459 untreated patients, and found that liver-related mortality was reduced for those with SVR compared with untreated patients (risk ratio, 0.050; 95% CI, 0.012-0.216). A meta-analysis by Singal et al43quantitatively assessed the reduction in decompensated cirrhosis, development of HCC, and liver-related mortality in patients who achieve SVR compared with those who were nonresponders to therapy. Two investigators independently reviewed 26 studies and pooled rates of decompensated cirrhosis, development of HCC, and liver-related mortality in patients with SVR and nonresponders.12, 40, 41, 42, 44, 45, 46, 47, 48, 49, 50, 51, 52, 53, 54, 55, 56, 57, 58, 59, 60, 61, 62, 63, 64 They found that patients with SVR were less likely to have liver-related mortality when compared with those who did not respond to therapy for hepatitis C (relative risk, 0.23; 95% CI, 0.10-0.52).43 A study conducted by the US Department of Veterans Affairs followed up a large cohort of 16,864 patients with HCV treated with PEG-IFN and ribavirin, and looked at effects of SVR on all-cause mortality. They followed up 12,166 patients with HCV genotype 1, 2904 patients with HCV genotype 2, and 1794 patients with HCV genotype 3, with SVR rates observed to be 35% in the HCV genotype 1 group, 72% in the HCV genotype 2 group, and 62% in the HCV genotype 3 group. By using genotype-specific multivariate survival models, the study concluded that SVR was associated with a substantially reduced mortality risk for each genotype (genotype 1 hazard ratio, 0.70; P < .0001; genotype 2 hazard ratio, 0.64; P = .006; genotype 3 hazard ratio, 0.51; P = .0002).65
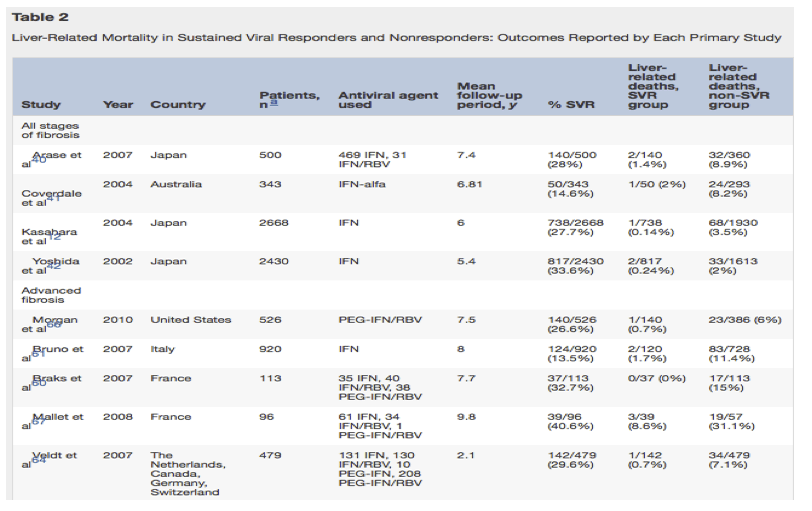
Hepatocellular Carcinoma Occurrence: All Stages of Fibrosis
Another important outcome of HCV treatment is the development of HCC, and multiple studies have looked at the effect of HCV therapy and SVR on the incidence of developing HCC (Table 3). Arase et al40 studied 500 Japanese patients with chronic hepatitis C in which 140 (28%) had SVR, and a total of 71 patients (14.2%) developed HCC during the follow-up period. A significant difference was seen in the incidence of HCC among those who were nonresponders compared with those with SVR (risk ratio, 1/0.22; 95% CI, 0.096-0.52; P < .0001). In the study by Coverdale et al,41 among the 384 patients treated with IFN therapy, 50 patients (15%) reached SVR, whereas 136 patients (40%) relapsed, and 157 patients (46%) were nonresponders. One patient (2%) in the SVR group developed HCC, whereas 5 patients (4%) in the relapse group and 18 patients (11%) in the nonresponse group developed HCC over a 9-year follow-up period. By using a univariate model, the chance of developing HCC was calculated as increased by a factor of 3.3 across each treatment response category (95% CI, 1.4-7.6; P = .004).41 Another study in Japan studied 594 patients who received IFN therapy, with a follow-up period of (mean ± standard deviation) 57.2 ± 13.9 months to assess for HCC development. Of the 594 treated patients, 175 patients (29.5%) had SVR, and it was seen that IFN therapy significantly decreased the incidence of HCC among patients with SVR (hazard rate ratio, 0.16; 95% CI, 0.09-0.79; P < .001) compared with nonresponders.68 A retrospective cohort study in 2007 of 1124 patients who received IFN therapy showed a 3.5% rate of HCC development in the 373 patients with SVR, compared with an 8.1% rate in the patients who did not have SVR.69 Hung et al11 also showed a similar difference in HCC incidence rates. Of 132 patients treated with IFN, 73 patients (55%) achieved SVR, and during a median follow-up period of 37 months, HCC developed in 5 patients with SVR (6.8%), and in 11 patients who did not have SVR (18.6%) (P = .0178). Bruno et al61 enrolled 920 patients, of whom 124 patients (13.5%) achieved SVR, and during a mean follow-up period of 96.1 months, the rate of HCC per 100 person-years of follow-up evaluation was 0.66 (95% CI, 0.27-1.37) for those with SVR and 2.10 (95% CI, 1.75-2.51) for those without SVR (P < .001). Therefore, patients who did not achieve SVR had a 2.59-fold increased rate of HCC development than those with SVR.61
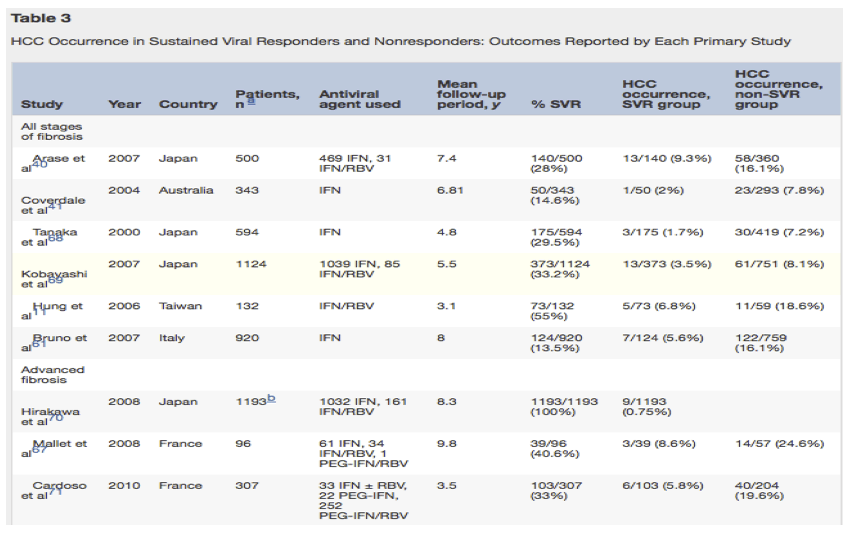
Hepatocellular Carcinoma Occurrence: Advanced Fibrosis
There are few studies that have examined the incidence of HCC development in patients with advanced fibrosis treated with IFN therapy and have achieved SVR. This may be owing to the small subset of patients with advanced fibrosis who typically are treated with IFN therapy. A 2008 study of 1193 patients who achieved SVR after IFN therapy showed the rate of developing HCC was significantly higher in the 41 patients with cirrhosis (liver fibrosis stage, F4), than in the 1106 patients with liver fibrosis stages F0 to F3 (hazard ratio, 12.9; 95% CI, 5.5-30.6; P < .001). The cumulative HCC development rate in patients with cirrhosis after attaining SVR was 15.5%, 24.2%, and 39.4%, at 5, 10, and 15 years after SVR, respectively, and the rates for those with liver fibrosis stages F0 to F3 were 1.00%, 1.68%, and 1.68% at 5, 10, and 15 years after SVR, respectively.70 In a study by Mallet et al,67 96 patients with biopsy-proven cirrhosis (liver fibrosis stage, F4) treated with IFN therapy for hepatitis C were followed up for a median period of 118 months. Of this group, 39 patients (40.6%) achieved SVR, and 3 patients (8.6%) with SVR developed HCC during the follow-up period, compared with 14 patients (23.3%) in the nonresponder group (P = .097). Cardoso et al71 evaluated 307 patients with chronic hepatitis C, of whom 127 patients (41.4%) had bridging fibrosis and 180 patients (58.6%) had cirrhosis. Cox regression analysis was used to assess the impact of IFN therapy on the incidence of HCC. SVR was seen in 37% of patients with bridging fibrosis and 30% of patients with cirrhosis P = .186), and over a median follow-up period of 3.5 years, the incidence rates per 100 person-years of HCC was 1.24 for those with SVR and 5.85 among non-SVR patients (log-rank test, P < .001). The data from these studies show that achieving SVR in advanced fibrosis decreases the risk of HCC
Liver Disease Progression and Hepatic Decompensation: All Stages of Fibrosis
Treatment for HCV is associated with a reduced risk of liver disease progression7, 8, 9 (Table 4). Bruno et al72 followed up 47 patients who attained SVR over 102 months, and observed that liver histology progressively improved in the patients with SVR. In all 47 patients, there were no decompensated events, and no deterioration in the histologic scores over time, and an improvement was noted in 88% of the patients (P < .0001). A retrospective study by Shiratori et al73assessing changes in hepatic fibrosis after IFN therapy saw that of the 183 patients who attained SVR, activity grade on histology was improved in 89% of those patients, whereas untreated patients had an unchanged activity grade (95% CI, 83%-93%). Those with SVR had an associated mean reduction in fibrosis score at more than 3 years of follow-up evaluation. Only 2 of the 183 patients who attained SVR had increased disease activity on biopsy (1.1%) compared with 58 of the 304 patients without SVR who had increased disease activity on repeat biopsy (19.1%). They also found that among patients without pre-existing cirrhosis, 10.9% of the 274 treated patients without SVR progressed to cirrhosis over 38 months, whereas none of the 159 patients with SVR developed cirrhosis. George et al74 conducted a study of 150 patients with SVR followed up for 5 years after therapy for chronic HCV and monitored liver-related outcomes and evidence of biochemical or virologic relapse. Of the 150 patients, virologic relapse was not seen, and in a blind rescoring of 49 paired biopsies (taken pretreatment and at long-term follow-up evaluation), 40 patients (82%) had a decrease in fibrosis score. Only 1 patient had an increase in inflammation seen on histology and developed HCC. None of the patients in the study had decompensated liver disease. Huang et al75 studied biopsy-proven, noncirrhotic, chronic hepatitis C patients who received IFN-based therapy to evaluate cirrhosis prevention. The large-scale, nationwide, multicenter, retrospective-prospective study enrolled 1386 patients, of whom 892 patients received IFN therapy. The annual incidence of cirrhosis in the untreated and IFN-treated groups was 2.26% and 1.11%, respectively, over 5 years of follow-up evaluation. Patients without SVR had a cirrhosis incidence rate of 1.99% versus 0.74% in those with SVR. The 14.5-year cumulative incidence of cirrhosis was significantly lower in the SVR group (4.8%), compared with nonresponders (21.6%) P =
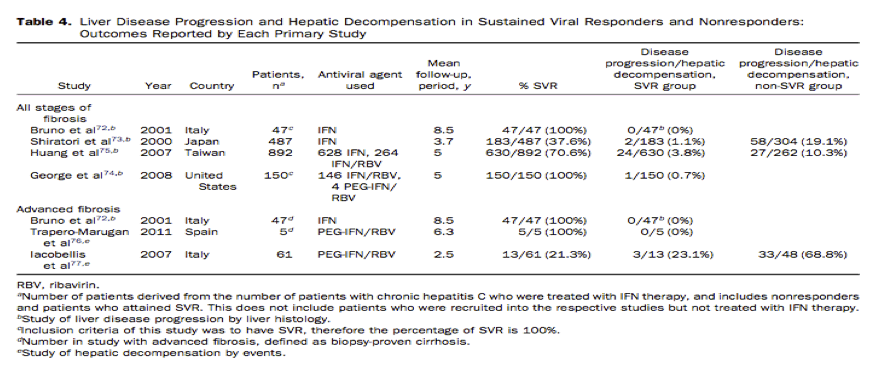
Liver Disease Progression and Hepatic Decompensation: Advanced Fibrosis
For patients with advanced fibrosis and chronic hepatitis C, attaining SVR with IFN has shown promising data on preventing progression or even leading to regression of disease. In the study by Bruno et al72 of the 47 patients who attained SVR with IFN therapy, 10 of the patients had biopsy-proven liver cirrhosis at the time of initial evaluation, and at follow-up evaluation 5 patients had a reduction of their fibrosis score, and 4 patients did not show any change. None of these patients showed progression of their liver disease. A study conducted in Spain followed up a group of 153 patients with SVR after IFN therapy over a mean follow-up period of 76 ± 13 months (SD), in which no evidence of virologic relapse was seen after the therapy period. Five patients (3.26%) had biopsy-proven cirrhosis before treatment and none of the patients showed evidence of hepatic decompensation during the follow-up period.76 Another study by Iacobellis et al77 studied patients with decompensated cirrhosis as a result of hepatitis C, in which 129 patients were enrolled and 66 patients received IFN therapy. After a follow-up period of 30 months, it was found that decompensated events occurred in 52, 33, and 3 of controls, nonresponders, and SVR patients, respectively. The annualized incidence of death was 2.34, 1.91, and 0 per 1000 patient-years, respectively, in controls, nonresponders, and SVR patients. Because patients with decompensated cirrhosis have a high risk of adverse events on IFN therapy, liver transplantation is still recommended, but these limited data do provide a promising alternative for those who are not transplantation candidates and warrants further
Conclusions
The significance of SVR has been shown in the individual studies detailed in this review article. Long-term benefits of SVR have been shown in patients with chronic hepatitis C because SVR has been associated with reducing liver disease progression, development of HCC, and liver-related mortality. These benefits are seen in those with all degrees of liver fibrosis, and the effects are significant even in those with advanced fibrosis. SVR should continue to be the goal in treating patients with chronic hepatitis C because it is the best marker of successful therapy. Future studies of treatment regimens and outcomes should be conducted with aims to maximize the potential of achieving SVR.
|
|
| |
| |
|
|
|