| |
FDA Approval DAA IFN-free Regimens Expected This Fall 2014 & Future Ongoing New Drug Development
|
| |
| |
And here are the actual study data & results reported at the liver meeting called EASL in London in April 2014 for key HCV new DAA INF-free regimens:
We can expect the FDA to approve in the Fall 2014: daclatasvir, sofosbuvir/ledipasvir and the Abbvie 3D regimen. Sofosbuvir+Simeprevir has been a potent regimen currently in use (COSMOS Study) with both drugs approved in Dec 2013. Daclatasvir+sofosbuvir is a potent regimen with phase 3 studies ongoing now & as mentioned above daclatasvir expected to be approved in the Fall 2014. Studies for all these regimens with the latest results are linked to below:
EASL Presentations Phase 3 Studies GT1, treatment-naive & treatment-experienced:
EASL: All Oral Fixed-Dose Combination Ledipasvir/Sofosbuvir With Or Without Ribavirin for 12 or 24 Weeks in Treatment-Naive Genotype 1 HCV-Infected Patients: the Phase 3 ION-1 Study - (04/14/14)
EASL: All Oral Fixed-Dose Combination Ledipasvir/Sofosbuvir With or Without Ribavirin for 12 or 24 Weeks in Treatment-Experienced Genotype 1 HCV-Infected Patients: The Phase 3 ION-2 Study - (04/14/14)
EASL: Ledipasvir/Sofosbuvir With and Without Ribavirin for 8 Weeks Compared to Ledipasvir/Sofosbuvir for 12 Weeks in Treatment-Naïve Noncirrhotic Genotype-1 HCV-Infected Patients: The Phase 3 ION-3 Study - (04/11/14)
EASL: TURQUOISE-II: SVR12 RATE OF 92-96% IN 380 HEPATITIS C VIRUS GENOTYPE 1-INFECTED ADULTS WITH COMPENSATED CIRRHOSIS TREATED WITH ABT-450/r/ABT-267 AND ABT-333 PLUS RIBAVIRIN - (04/14/14)
EASL: SAPPHIRE-I: PHASE 3 PLACEBO-CONTROLLED STUDY OF INTERFERON-FREE, 12-WEEK REGIMEN OF ABT-450/r/ABT-267, ABT-333, AND RIBAVIRIN IN 631 TREATMENT-NAïVE ADULTS WITH HEPATITIS C VIRUS GENOTYPE 1 - (04/11/14)
EASL: SAPPHIRE-II: PHASE 3 PLACEBO-CONTROLLED STUDY OF INTERFERON-FREE, 12-WEEK REGIMEN OF ABT-450/r/ABT-267, ABT-333, AND RIBAVIRIN IN 394 TREATMENT-EXPERIENCED ADULTS WITH HEPATITIS C VIRUS GENOTYPE 1 - (04/10/14)
Here are the results from studies of daclatasvir+sofosbuvir & ongoing phase 3 studies:
Daclatasvir+Sofosbuvir - (05/09/14)
COSMOS - Simeprevir+Sofosbuvir
EASL: Once-daily simeprevir (TMC435) plus sofosbuvir (GS-7977) with or without ribavirin in HCV genotype-1 prior null responders with METAVIR F0-2: COSMOS study subgroup analysis - (04/14/14)
EASL: Simeprevir plus sofosbuvir with/without ribavirin in HCV genotype-1 prior null-responder / treatment-naïve patients (COSMOS study): primary endpoint (SVR12) results in patients with METAVIR F3-4 (Cohort 2) - (04/14/14)
Janssen Initiates Phase 3 OPTIMIST Trials of Once-Daily Simeprevir in Combination with Once-Daily Sofosbuvir for the Treatment of Genotype 1 Chronic Hepatitis C - (04/03/14)
HELIX-2, a phase II all-oral combination study of Simeprevir, TMC647055 and Samatasvir (IDX719) for the treatment of hepatitis C has been initiated...... http://www.natap.org/2013/HCV/120313_02.htm
EASL: New DAA Phase 3 Studies for genotype 1 at EASL - in both Treatment-naives, treatment-experienced; simultaneous publications - (04/21/14)
---------------------
In phase 3 & further back in development are:
Merck
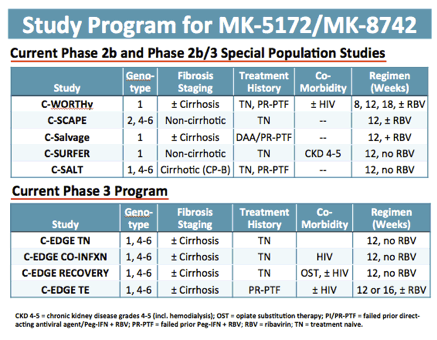
New study:
Efficacy and Safety of MK-5172, MK-8742, and Sofosbuvir for Chronic Infection With Hepatitis C Virus Genotypes 1 and 3 (MK-5172-074) - (05/15/14)
In phase 3 now:
Efficacy and safety of MK-5172 and MK-8742± RIBAVIRIN IN Hepatitis C Genotype 1 Infected Patients with cirrhosis or previous null response: the C-WORTHY Study - (04/11/14)
EASL: SAFETY AND EFFICACY OF THE ALL-ORAL REGIMEN OF MK-5172 / MK-8742± RIBAVIRIN IN TREATMENT-NAIVE, NONCIRRHOTIC PATIENTS WITH HEPATITIS C VIRUS GENOTYPE 1 INFECTION: THE C-WORTHy STUDY - (04/14/14)
EFFICACY AND SAFETY OF THE ALL-ORAL REGIMEN, MK-5172/MK-8742 +/- RBV FOR 12 WEEKS IN GT1 HCV/HIV CO-INFECTED PATIENTS: THE C-WORTHY STUDY - (04/11/14)
------------------
Idenix
Ongoing phase 2 program:
Idenix Announces Promising Clinical Data and Continued Progress in Nucleotide Prodrug Development Programs for the Treatment of Hepatitis C - (04/07/14)
EASL: Favorable Preclinical Profile of IDX21437, a Novel Uridine Nucleotide Prodrug, for Use in a Direct-Acting Antiviral (DAA) Regimen for HCV - (04/16/14)
EASL: A Phase II Study of Samatasvir (IDX719) in Combination with Simeprevir and Ribavirin in Treatment-Naïve HCV-Infected Subjects with Genotypes 1b and 4 (HELIX-1 Study) - (04/16/14)
EASL: Pharmacokinetic (PK) Drug-Drug Interaction between Samatasvir (IDX719), a Pan-Genotypic NS5A Inhibitor, and Simeprevir in Healthy Volunteers and HCV-Infected Subjects - (04/16/14)
-----------------
Achillion
Ongoing phase 2 program:
Achillion Advances ACH-3422, Uridine-Analog Nucleotide Inhibitor, Into Clinical Trial; Initiates Phase 2 Pilot Study With ACH-3102, NS5A Inhibitor (+sofosbuvir), for HCV - (05/01/14)
APASL: A Single Direct-acting Anti-viral Agent, ACH-3102, with Ribavirin is Able to Achieve a Robust Anti-viral Response in Subjects with Genotype 1b Chronic Hepatitis C Infection - (04/17/14)
APASL: ACH-3102 and Ribavirin in Genotype-1b Hepatitis C Patients: Confirmation of the High Barrier to Viral Breakthrough in Genotype-1b HCV - (04/17/14)
APASL: SVR4 Results for the Combination of ACH-3102 and Sovaprevir, With Ribavirin, in Patients With Genotype 1 Chronic Hepatitis C Infection - (04/17/14)
|
|
| |
| |
|
|
|