| |
HCV Treatment Cost-Effectiveness/ SOF/PR SVR
Reduces Liver Disease vs PI/PR Treatments
|
| |
| |
Reported by Jules Levin, NATAP
Cost-Effectiveness Analysis of Direct-Acting Antiviral Therapy for Treatment-Naïve Patients with Chronic Hepatitis C Genotype 1 Infection in the Veterans Health Administration
http://www.natap.org/2013/HCV/062613_01.htm
"In conclusion, our model indicates the upfront costs required for treatment with Boc/PR or Tel/PR are high; however the offsetting benefits of extending quality of life and lower costs due to liver-related morbidity indicate that these therapies have very acceptable incremental cost-effectiveness ratios compared to previous therapies in this managed care health care system. Further efforts to expand access to DAA therapy are warranted.
-----With the previously achieved inital treatment rate of 22 %, total system-wide costs to adopt Boc/PR or Tel/PR would be $708 million and $943 million, respectively. Increasing treatment rates to 50% would result in the total cost of antiviral therapies PR, Boc/PR and Tel/PR treatment to be $411 million, $1,610 million and $2,144 million, respectively (Figure 3). Without antiviral treatment, the expected total cost of care for hepatitis C-related liver disease is $3,729 million. Compared with no treatment using PR at a 50% treatment rate results in overall cost savings of $30 million over the VHA cohort lifetime. In contrast, using Boc/PR or Tel/PR at a 50% treatment rate results in net cost expenditures of $692 million or $1,175 million, respectively."
------------------------
A decision-analytic Markov model evaluated the health outcomes of SOF compared with current treatment options....."the number of cases of advanced liver disease were 77-81% lower for SOF+PR vs PR, 7078% lower for SOF+PR vs BOC+PR, and 60-67% lower for SOF+PR vs Telaprevir+PR"
AASLD 2013
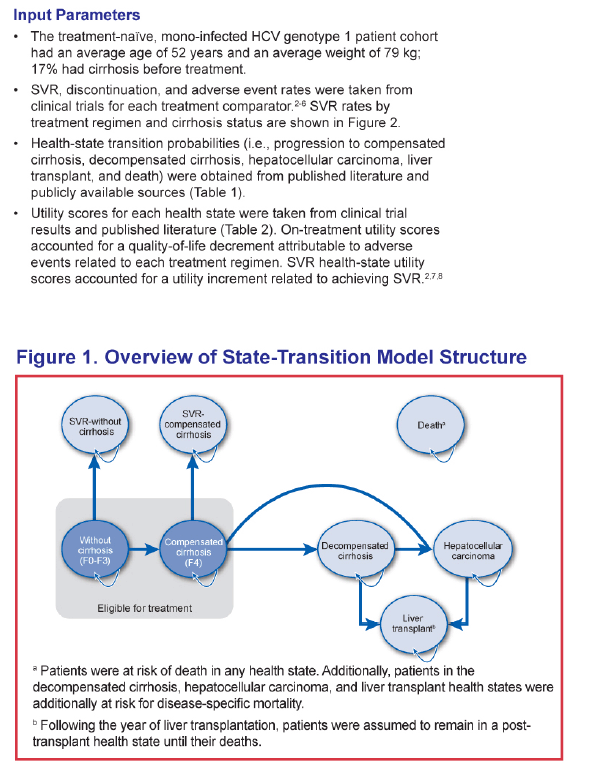
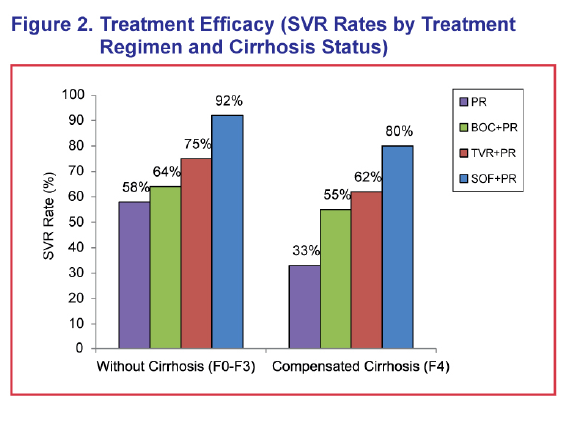
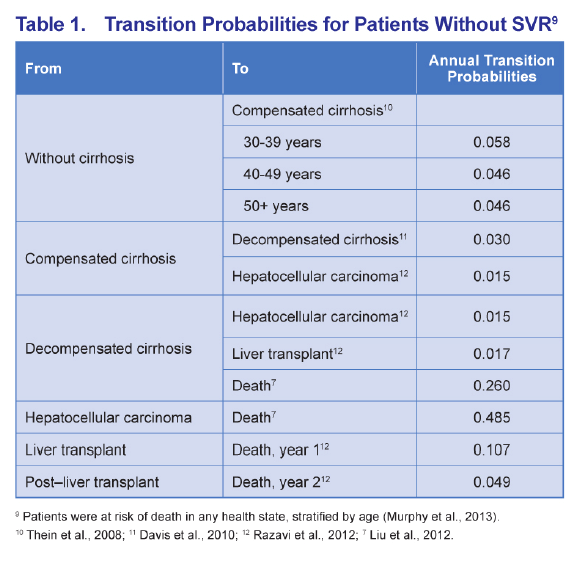
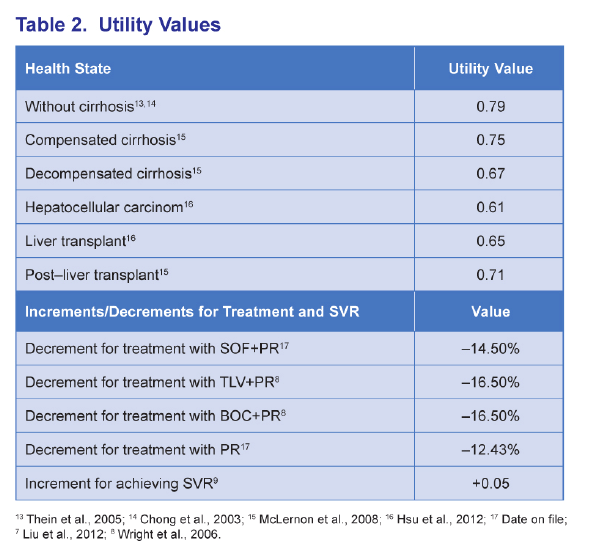
ABSTRACT - METHODS: The analysis modeled a cohort of treatment-naïve chronic HCV genotype 1 patients with an average age of 52 and 17% with cirrhosis to 100 years of age from a US third-party payer perspective. SOF+PR was compared with telaprevir (TVR) + PR for 24-48 weeks, boceprevir (BOC) + PR for 28-48 weeks, and PR for 48 weeks. Sustained virologic response (SVR) and adverse event rates were based on phase III clinical trials (SVR = 92% and 80% for SOF+PR, 75% and 62% for TVR+PR, 64% and 55% for BOC+PR, and 58% and 33% for PR in non-cirrhotics and cirrhotics, respectively). Transition probability and utility were based on a literature review and consensus by a panel of 4 hepatologists.
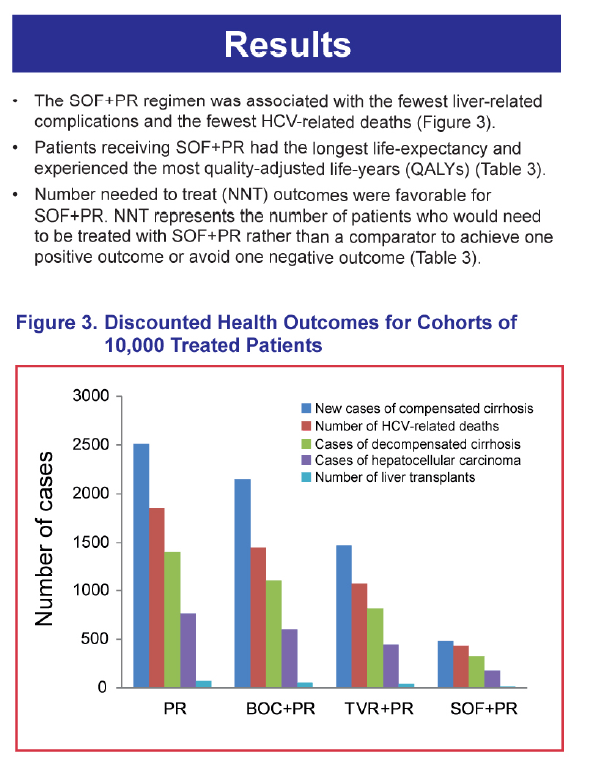
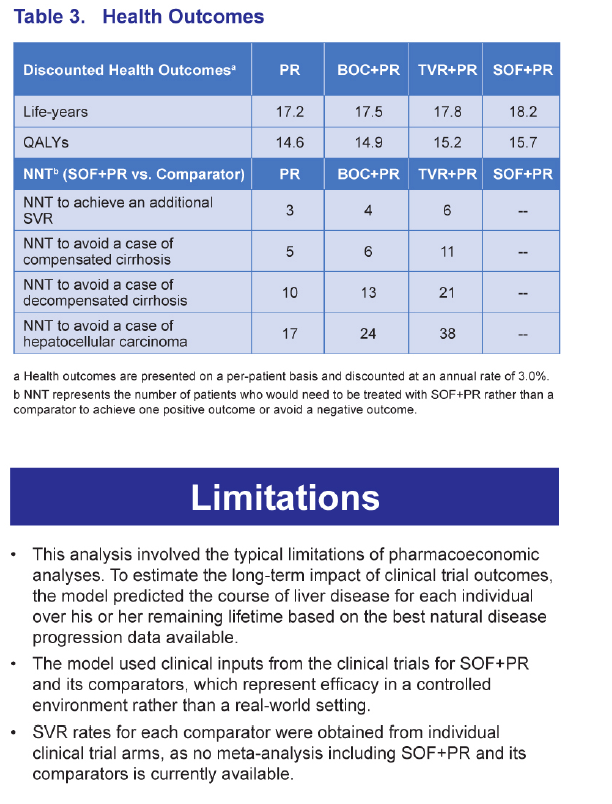
RESULTS: The SOF+PR regimen resulted in the best outcomes compared with TVR+PR, BOC+PR, and PR, with the lowest numbers of patients with liver-related complications. In addition, analysis of the number needed to treat (NNT), which is the number of patients who would need to be treated with SOF+PR rather than the comparator to achieve one positive outcome or avoid one negative outcome, also indicated favorable outcomes with SOF+PR.
CONCLUSIONS: The SOF-based regimen was projected to yield better health outcomes. Large discrepancies in efficacy, side effect, and adherence rates have been reported for currently available regimens in the real-world vs. clinical trial settings. Additional analyses are necessary to determine the potential impact of the greater expected real-world differences between the SOF-based regimen and other therapies.




|
|
| |
| |
|
|
|