| |
Patterns and Correlates of PrEP Drug Detection among MSM and Transgender Women in the Global iPrEx Study.
|
| |
| |
Download the PDF here
JAIDS Journal of Acquired Immune Deficiency Syndromes: Sept 16 2014
Liu, Albert M.D., M.P.H.; Glidden, David V. Ph.D.; Anderson, Peter L. Pharm.D.; Amico, K.R. Ph.D.; McMahan, Vanessa B.S.; Mehrotra., Megha M.P.H.; Lama, Javier R. M.D., M.P.H.; MacRae, John M.D; Hinojosa, Juan Carlos M.D.; Montoya, Orlando M.Sc.; Veloso, Valdilea G. M.D., Ph.D.; Schechter, Mauro M.D., Ph.D.; Kallas, Esper G. M.D., Ph.D.; Chariyalerstak, Suwat M.D.; Dr. P.H.; Bekker, Linda-Gail M.B., Ch.B., Ph.D.; Mayer, Kenneth M.D.; Buchbinder, Susan M.D.; Grant, Robert M.D., M.P.H.; for the iPrEx Study Team
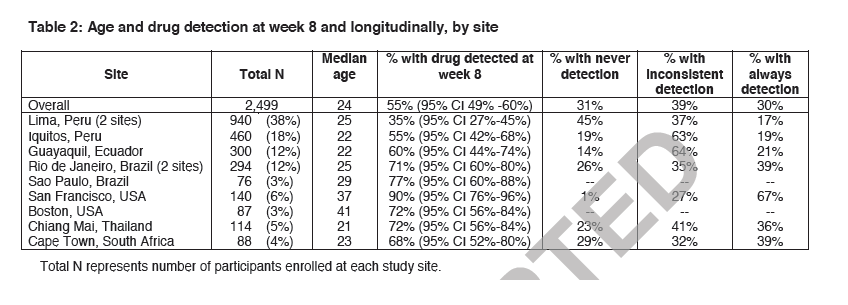
"In the longitudinal analyses, markers of risky sexual behavior (e.g. reporting ncRAI, ≥2 sexual partners, or having an STI) were associated with more frequent drug detection at some or all study visits, suggesting that participants who had some level of risk were more likely to attempt to take the study medication. Similarly, study drug adherence was lower during periods of no sexual activity in the Partners PrEP study, suggesting that adherence to PrEP may be lower during periods of low perceived risk.26 Greater drug detection among those engaging in the highest risk sexual practices is expected to increase the impact and cost-effectiveness of PrEP.29.......The analysis of pharmacologic levels of antiretroviral drugs has emerged as an objective measure of study product use in PrEP trials1-4 and may help identify groups at risk for non-adherence, who may benefit from adherence support, and help identify those most likely to adhere to the PrEP regimen...... Rates of drug detection varied significantly by site (Table 2), ranging from 35% in Lima, Peru, to 90% in San Francisco, USA"
Abstract
Background: Adherence to pre-exposure prophylaxis (PrEP) is critical for efficacy. Antiretroviral concentrations are an objective measure of PrEP use and correlate with efficacy. Understanding patterns and correlates of drug detection can identify populations at risk for non-adherence and inform design of PrEP adherence interventions.
Methods: Blood antiretroviral concentrations were assessed among active-arm participants in iPrEx, a randomized, placebo-controlled trial of emtricitabine/tenofovir in men who have sex with men (MSM) and transgender women in 6 countries. We evaluated rates and correlates of drug detection among a random sample of 470 participants at week 8 and a longitudinal cohort of 303 participants through 72 weeks of follow-up.
Results: Overall, 55% (95% CI 49-60%) of participants tested at week 8 had drug detected. Drug detection was associated with older age and varied by study site. In longitudinal analysis, 31% never had drug detected, 30% always had drug detected, and 39% had an inconsistent pattern. Overall detection rates declined over time. Drug detection at some or all visits was associated with older age; indices of sexual risk, including condomless receptive anal sex; and responding 'don't know' to a question about belief of PrEP efficacy (0-10 scale).
Conclusions: Distinct patterns of study-product use were identified, with a significant proportion demonstrating no drug detection at any visit. Research literacy may explain greater drug detection among populations having greater research experience, such as older MSM in the US. Greater drug detection among those reporting highest-risk sexual practices is expected to increase the impact and cost-effectiveness of PrEP.
The use of antiretroviral pre-exposure prophylaxis (PrEP) for HIV prevention has generated great interest and promise, with four recently completed randomized clinical trials demonstrating its safety and efficacy across different populations.1-4 A key finding from these studies is the critical relationship between adherence and PrEP efficacy.5,6 For example, in the Global iPrEx study, the efficacy of daily oral emtricitabine/tenofovir disoproxil fumarate (FTC/TDF) was 42% on an intention-to-treat basis, but was estimated to be >90% among those with drug detected at any concentration in blood.4,7 Similarly, in the CAPRISA 004 microbicide study, having a tenofovir (TFV) level of >1000 ng/ml in cervicovaginal fluid was associated with significant efficacy (74%), while a TFV level below this threshold was not protective.7,8 While adherence and efficacy were both high among HIV-uninfected men and women in serodiscordant couples in the Partners PrEP study,1 the FEM-PrEP and VOICE studies were unable to demonstrate PrEP efficacy in African women, due in part to low study product adherence.9,10
Although self-reported and pill-count based adherence data have been commonly used in research settings, including PrEP studies, these measures are subject to social desirability bias and potential manipulation11,12 and have been shown to over-estimate PrEP use across a number of studies.13 The analysis of pharmacologic levels of antiretroviral drugs has emerged as an objective measure of study product use in PrEP trials1-4 and may help identify groups at risk for non-adherence, who may benefit from adherence support, and help identify those most likely to adhere to the PrEP regimen.
The Global iPrEx Study, the only PrEP efficacy trial among men who have sex with men (MSM) and transgender women, was conducted in 6 countries in North and South America, Asia, and Africa.4 We evaluated rates and correlates of PrEP drug detection in blood early in the study and longitudinally among participants in this trial.
Serum drug detection at week 8
Overall, 55% (95% CI 49-60%) of week 8 samples tested had drug detected. Detection of TFV and FTC were concordant in 99% samples. Rates of drug detection varied significantly by site (Table 2), ranging from 35% in Lima, Peru, to 90% in San Francisco, USA. The proportion of participants with drug detection according to socio-demographic variables and reported behaviors is shown in Table 3. Drug detection was significantly associated with older age (Table 3). Level of education, number of male sex partners, ncRAI, alcohol or drug use, creatinine clearance, perceived treatment assignment and PrEP efficacy, being transgender, living situation, and concern about having a job or place to live were not significantly associated with drug detection at week 8. Reporting gastrointestinal symptoms (nausea, vomiting, diarrhea, flatulence, or abdominal pain) or headache at 4 or 8 weeks after PrEP initiation was not associated with drug detection at week 8 (all p values >0.50).
Patterns of drug detection over time
For the 303 study participants included in the longitudinal analyses, the average number of samples tested was 3.8 (range 2-6). Out of 1168 visits tested, 467 (40%) visits had both PBMC and plasma samples available for testing, 688 (59%) visits had only plasma available, and 13 (1%) had PBMCs only. Overall, 31% did not have drug detected in any sample, 30% had drug detected in all samples, and 39% had inconsistent drug detection (at some time-points but not others). Patterns of drug detection varied by site: San Francisco had the highest proportion with drug detection at all time-points; Lima, Peru had the highest proportion never having drug detected; and Iquitos, Peru and Guayaquil, Ecuador had the highest proportion with inconsistent drug detection pattern (Table 2). In univariable analyses, having drug detection at some or all visits was associated with older age, baseline ncRAI, and responding "don't know" to a question on PrEP efficacy, when compared with those who never had drug detected (Table 4). Having drug detected at all visits was also associated with reporting ≥2 sex partners and reporting an
STI at baseline, and was marginally associated with greater HIV risk perception. Results were similar in multivariable analyses, except that reporting an STI was no longer significant for always having drug detection. In a model adjusting for site and baseline ncRAI, there was a trend towards increased drug detection at follow-up visits where ncRAI was also reported (OR 1.60, 95% CI 0.98-2.62).
The proportion of participants with drug detection declined over the first 72 weeks of study participation, from a detection rate of 59% at week 8 to 44% at week 72 (Figure 1a). Trajectories of drug detection over time for individual participants in the longitudinal cohort are shown graphically in Figure 1b. Among the 163 participants with drug detected at the first visit (orange lines), 53 (32%) showed a pattern of discontinuation (drug detection stopped and remained undetectable at subsequent visits), with a median time of stopping of 24 weeks. The remaining two-thirds (68%) had drug detection at some or all subsequent follow-up visits.
Among participants who had drug detected at week 12, reporting GI symptoms between weeks 12 and 24 was positively associated with drug detection at week 24 (AOR 4.32, 95% CI 1.07-17.49, p=0.04), while reporting headache during this time period was not significantly associated with week 24 detection (p=0.36). Of the 139 participants who had no drug detected at the first time-point (green lines), the majority (68%) did not have drug detection at any subsequent time-point.
|
|
| |
| |
|
|
|