| |
Updates to the HCV/HIV Section in HHS ART Adult/Adolescent
Guidelines Nov 13 2014
|
| |
| |
http://aidsinfo.nih.gov/guidelines/html/1/adult-and-adolescent-arv-guidelines/26/hiv-hcv
--------------------
New Guidelines for the Use of Antiretroviral Agents in HIV-1-Infected Adults and Adolescents:
http://aidsinfo.nih.gov/guidelines/html/1/adult-and-adolescent-arv-guidelines/0
On November 13, 2014, updates to the
Hepatitis C (HCV)/HIV Coinfection section of the Adult and Adolescent ARV
guidelines were published. For details, please visit the What's New in the Guidelines section.
This interim revision of the Guidelines includes a revised section on Hepatitis C Virus (HCV)/HIV Coinfection, with emphasis on considerations for use of antiretroviral (ARV) drugs in patients who also receive treatment for HCV infection. A new table (Table 12) provides clinicians with guidance on the concomitant use of HCV drugs and ARV drugs, with a focus on potential pharmacokinetic drug interactions. The Panel refers clinicians to
http://www.hcvguidelines.org for guidance on the diagnosis and treatment of HCV infections.
----------------------
Panel's Recommendations
All HIV-infected patients shold be screened for hepatitis C virus (HCV) infection. Patients at high risk of HCV should be
screened annually and whenever HCV infection is suspected.
Antiretroviral therapy (ART) may slow the progression of liver disease by preserving or restoring immune function and
reducing HIV-related immune activation and inflammation. For most HIV/HCV-coinfected patients, including those with
cirrhosis, the benefits of ART outweigh concerns regarding drug-induced liver injury (DILI). Therefore, ART should be
initiated in most HIV/HCV-coinfected patients, regardless of CD4 T lymphocyte (CD4) cell count (BII).
Initial ART combination regimens recommended for most HIV/HCV-coinfected patients are the same as those
recommended for individuals without HCV infection. However, when treatment for both HIV and HCV is indicated,
consideration of potential drug-drug interactions and overlapping toxicities should guide ART regimen selection or
modification (see discussion in the text below and in Table 12)......("......in HIV treatment-naive patients with CD4 counts >500
cells/mm , some clinicians may choose to defer ART until HCV treatment is completed to avoid drug-drug
interactions (CIII)")
Combined treatment of HIV and HCV can be complicated by drug-drug interactions, increased pill burden, and
toxicities. Although ART should be initiated for most HIV/HCV-coinfected patients regardless of CD4 cell count, in ARTnaive
patients with CD4 counts >500 cells/mm some clinicians may choose to defer ART until HCV treatment is
completed (CIII).
In patients with lower CD4 counts (e.g., <200 cells/mm ), ART should be initiated expeditiously (AI) and HCV therapy
may be delayed until the patient is stable on HIV treatment (CIII).
---------------------------
Drug-Drug Interaction
Considerations for the concurrent use of ART and recommended HCV agents (per Error! Hyperlink reference not valid.) are as follows:
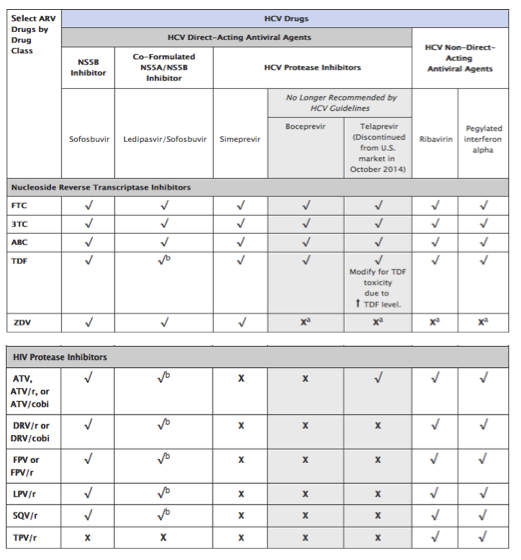
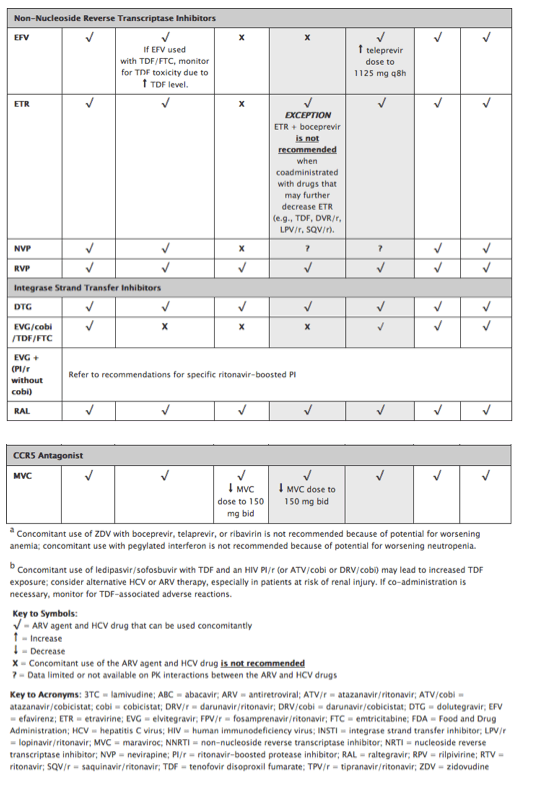
· Simeprevir (similar to boceprevir and telaprevir) is a HCV NS3/4A PI that has been studied in HIV/HCV-coinfected patients.36 Simeprevir is a substrate and inhibitor of CYP3A4 and p-glycoprotein (p-gp) enzymes, and therefore may have significant interactions with certain ARVs that are metabolized by the same pathways. Simeprevir is also an inhibitor of the drug transporter OATP1B1/3. On the basis of drug-drug interaction studies in healthy volunteers, simeprevir can be coadministered with raltegravir (RAL), dolutegrevir, rilpivirine (RPV), and tenofovir (TDF).37 However, coadministration of simeprevir with efavirenz (EFV), etravirine, HIV PIs, cobicistat (cobi), or elvitegravir/cobiscistat/tenofovir/emtricitabine (EVG/cobi/TDF/FTC) is not recommended (see Table 12 for recommendations on the concomitant use of selected drugs for treatment of HCV and HIV infection).
· Sofosbuvir is an HCV NS5B nucleotide polymerase inhibitors that is not metabolized by the cytochrome P450 enzyme system and, therefore, can be used in combination with most ARV drugs. Sofosbuvir is a substrate p-gp. P-gp inducers, such as tipranavir, may decrease sofosbuvir plasma concentrations and should not be co-administered with sofosbuvir. No other clinicially significant pharmocokinetic intractions between sofosbuvir and ARVs have been identified. Drug-drug interaction studies in healthy volunteers did not find any significant interaction between sofosbuvir and darunavir/ritonavir, EFV, RPV, RAL, TDF, or FTC38 (see Table 12 for recommendations on the concomitant use of selected drugs for treatment of HCV and HIV infection).
· A fixed drug combination of sofosbuvir and ledipasvir has been approved by the Food and Drug Administration.39 Ledipasvir is an HCV NS5A inhibitor and, similar to sofosbuvir, is not metabolized by the cytochrome P450 system of enzymes and is a substrate for p-gp. The potential for clinically significant drug-drug interactions is low. However, the coadministration of sofosbuvir/ledipasvir and ARV regimens containing TDF together with an HIV PI boosted with either RTV or cobi is associated with increased exposure to TDF (see Table 12 for recommendations on the concomitant use of selected drugs for treatment of HCV and HIV infection). In some patients, alternative HCV or ARV drugs should be considered to avoid increases in TDF exposures. If the drugs are co-administered, the patient should be monitored for potential TDF-associated renal injury by assessing measurements of renal function (i.e., estimated creatinine clearance, serum phosphorus, urine glucose, and urine protein) before HCV treatment initiation and periodically during treatment.
Table 12. Recommendations for Concomitant Use of Selected Antiretroviral Drugs and All Food and Drug Administration (FDA)-Approved Drugs for Treatment of Hepatitis C in HIV-Infected Adults
These recommendations for concomitant use of selected HIV drugs with FDA-approved HCV drugs are based on available data on pharmacokinetics interaction or on predictions based on the known metabolic pathway of the agents. In some cases, there are not enough data for any recommendations; these cases are indicated in the table. Whenever HIV and HCV drugs are used concomitantly, patients should be closely monitored for HIV and HCV virologic efficacy and potential toxicities. Given the rapidly evolving field of HCV therapy, readers should also refer to the latest drug product labels and HCV guidelines (www.hcvguidelines.org/) for updated information on the concurrent use of HIV and HCV drugs.
When to Start Antiretroviral Therapy
The rate of liver disease (liver fibrosis) progression is accelerated in HIV/HCV-coinfected patients, particularly
in individuals with low CD4 counts (≤350 cells/mm ). Data largely from retrospective cohort studies are
inconsistent regarding the effect of ART on the natural history of HCV disease. However, ART may slow
the progression of liver disease by preserving or restoring immune function and by reducing HIV-related
immune activation and inflammation Therefore, ART should be initiated in most HIV/HCV-coinfected
patients, regardless of CD4 count (BII). However, in HIV treatment-naive patients with CD4 counts >500
cells/mm , some clinicians may choose to defer ART until HCV treatment is completed to avoid drug-drug
interactions (CIII). Compared to patients with CD4 counts >350 cells/mm , those with CD4 counts <200 had
lower HCV treatment responses and higher toxicity rates to PegIFN/RBV. Data regarding HCV treatment
response to combination therapy with DAA agents in those with advanced immunosuppression is lacking. For
patients with lower CD4 counts (e.g., <200 cells/mm ), ART should be initiated expeditiously (AI) and HCV
therapy may be delayed until the patient is stable on HIV treatment (CIII)
|
|
| |
| |
|
|
|