 |
 |
 |
| |
High SVR12 With Sofosbuvir/Ribavirin in HCV/HIV-Coinfected: PHOTON-2
|
| |
| |
20th International AIDS Conference, July 20-25, 2014, Melbourne
Mark Mascolini
Overall, more than 80% of HCV treatment-naive or experienced people with HIV infection attained a 12-week sustained virologic response (SVR12) with sofosbuvir plus ribavirin in the 274-person PHOTON-2 trial [1]. HCV therapy had no major impact on CD4 percent or HIV RNA in these coinfected people.
Sofosbuvir, a nucleotide analog NS5B polymerase inhibitor, is licensed for treatment of chronic HCV infection. Previous research established sofosbuvir efficacy in people with HCV genotype 1, 2, 3, or 4, including patients with hepatocellular carcinoma. A fixed-dose combination of sofosbuvir with the NS5A inhibitor ledipasvir is in development [2].
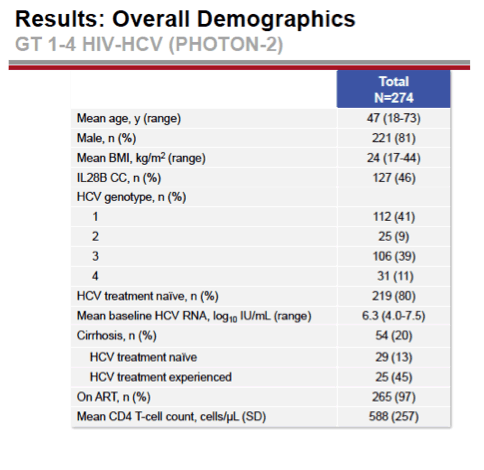
PHOTON-2 involved 274 HCV/HIV-coinfected people with HCV genotype 1, 2, 3, or 4 infection, with or without HCV treatment experience, and with or without compensated cirrhosis. Study participants took a 400-mg once-daily sofosbuvir tablet (the licensed dose) plus 1000 to 1200 mg of ribavirin daily. HCV treatment-naive people with genotype 1, 3, or 4 and treatment-experienced people with genotype 2 or 3 took sofosbuvir/ribavirin for 24 weeks. Treatment-naive people with genotype 2 HCV took the combination for 12 weeks. The primary endpoint in all study arms was sustained virologic response 12 weeks after treatment stopped (SVR12). All study participants had undetectable HIV RNA for at least 8 weeks on a stable antiretroviral regimen. Antiretroviral-naive people had a CD4 count above 500, and antiretroviral-treated people had a count above 200.
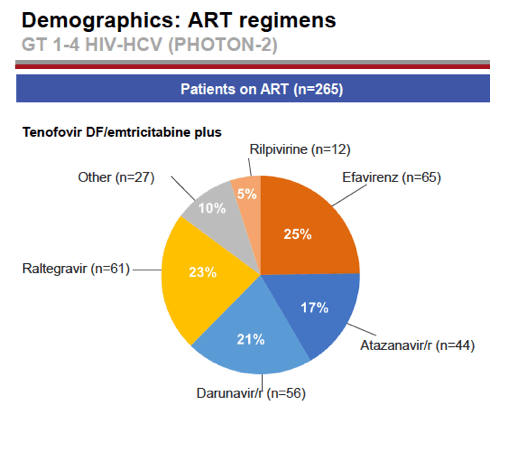
Most study participants, 81%, were men, and 97% were taking antiretrovirals. Baseline CD4 count averaged 588. HCV treatment-naive subgroups included 112 people with genotype 1, 19 with genotype 2, 57 with genotype 3, and 31 with genotype 4. Treatment-experienced groups included 6 with genotype 2 and 49 with genotype 3. Across those six groups, proportions with compensated cirrhosis were 15%, 5%, 5%, 26%, 33%, and 47% (20% overall). Respective pretreatment HCV RNA levels averaged 6.3 log10 IU/mL, 6.7 log, 6.3 log, 5.9 log, 6.4 log, and 6.3 log. More than 15% of participants were taking efavirenz (25%), raltegravir (23%), darunavir/ritonavir (21%), or atazanavir/ritonavir (17%).
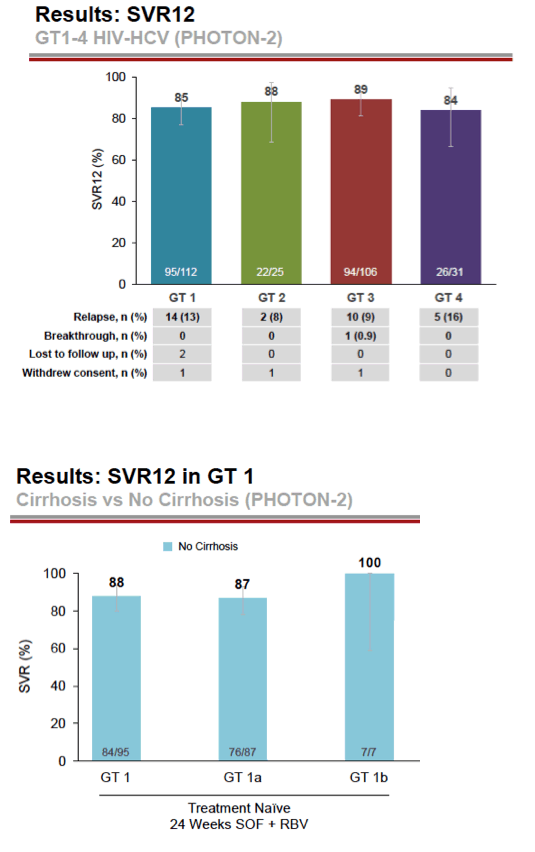
Almost all study participants had undetectable HCV RNA (below 25 IU/mL) after 4 weeks of treatment. Relapse rates ranged from 5% to 17% across the six study groups. Among the 38 people who did not attain SVR12, 31 had a relapse, 1 had breakthrough viremia, and 6 withdrew consent or stopped returning for study visits. SVR12 rates were high with all four genotypes:
-- Genotype 1: 85%
-- Genotype 2: 88%
-- Genotype 3: 89%
-- Genotype 4: 84%
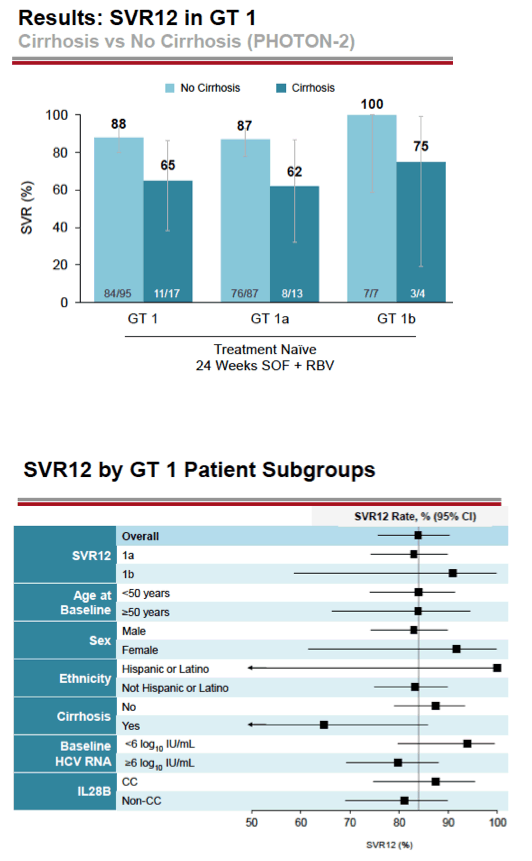
Among treatment-naive people with genotype 1 infection, SVR12 rates were higher in people without than with cirrhosis:
-- Genotype 1 overall: 88% without versus 65% with cirrhosis
-- Genotype 1a: 87% without versus 62% with cirrhosis
-- Genotype 1b: 100% without versus 75% with cirrhosis
SVR12 rates in cirrhotics and noncirrhotics did not differ as much with the other genotypes:
-- Genotype 2 naive: 89% without versus 100% with cirrhosis
-- Genotype 2 experienced: 75% without versus 100% with cirrhosis
-- Genotype 3 naive: 91% without versus 100% with cirrhosis
-- Genotype 3 experienced: 92% without versus 78% with cirrhosis
-- Genotype 4 naive: 83% without versus 88% with cirrhosis
Numbers of patients in most of these subgroups were small.
The most frequent adverse events were fatigue, insomnia, and nausea. Only 5 study participants (2%) stopped sofosbuvir and ribavirin because of adverse events; 2 stopped ribavirin because of adverse events. Only 15 people (6%) had grade 3 or 4 adverse events, and 15 had serious adverse events. Grade 3 or 4 lab abnormalities developed in 54 patients (20%). Hemoglobin fell below 10 g/dL in 26 patients (9.5%).
CD4 percent did not change appreciably through treatment and 12 weeks of follow-up. HIV RNA rebounded in 4 patients (1.5%) during that period, but these intermittent blips did not require antiretroviral changes. HIV RNA did not change significantly in people not taking antiretrovirals.
The PHOTON-2 team concluded that HCV treatment-naive and experienced people with HIV coinfection responded well to 12 or 24 weeks of sofosbuvir plus ribavirin and that the regimen proved safe with multiple antiretroviral regimens.
References
1. Molina JM, Orkin C, Iser DM, et al. All-oral therapy with sofosbuvir plus ribavirin for the treatment of HCV genotypes 1, 2, 3 and 4 infection in patients co-infected with HIV (PHOTON-2). AIDS 2014. 20th International AIDS Conference. July 20-25, 2014. Melbourne. Abstract MOAB0105LB.
2. Lawitz E, Poordad FF, Pang PS, et al. Sofosbuvir and ledipasvir fixed-dose combination with and without ribavirin in treatment-naive and previously treated patients with genotype 1 hepatitis C virus infection (LONESTAR): an open-label, randomised, phase 2 trial. Lancet. 2014;383:515-523. www.natap.org/2014/HCV/PIIS0140673613621212.pdf
-------------------------------------------
All-Oral Therapy with Sofosbuvir Plus Ribavirin for the Treatment of HCV Genotype 1, 2,3 and 4 Infection in Patients Coinfected with HIV (PHOTON-2)
Reported by Jules Levin
IAS 2014, Melbourne, Australia
Jean-Michel Molina1, Chloe Orkin2, David M. Iser3,Francisco.X. Zamora4, Mark Nelson5, Christoph Stephan6, Benedetta Massetto7, Anuj Gaggar7, Liyun Ni7, Evguenia Svarovskaia7, Diana Brainard7, G. Mani Subramanian7, John G. McHutchison7, Massimo Puoti8, J�rgen K. Rockstroh9
1University of Paris Diderot, Paris 7 and Department of Infectious Diseases, Saint-Louis Hospital, Paris, France; 2Barts Health NHS Trust, London, UK; 3Infectious Diseases Unit, Alfred Hospital, Melbourne, Australia; 4HIV Unit, Internal Medicine Service, Hospital Universitario La Paz, IdiPAZ, Madrid, Spain; 5Chelsea and Westminster Hospital, St. Stephens Centre, London, UK; 6Infectious Diseases Unit at Medical Department, Hospital of the Johann Wolfgang Goethe-University, Frankfurt, Germany; 7Gilead Sciences, Inc., Foster City, CA, USA; 8Division of Infectious Diseases, AO Ospedale Niguarda Ca' Granda, Milan, Italy; 9Department of General Internal Medicine I, University Hospital of Bonn, Bonn, Germany
- 88% (84/95) with GT1 without cirrhosis had SVR12: 88% Gt1a (76/87), 100% Gt1b (7/7)
- 89% (16/18) of treatment-naive GT2 without cirrhosis had SVR12, 24 weeks treatment
- 91% (49/54) treatment-naive Gt3 without cirrhosis had SVR12, 24 weeks treatment
- 83% (19/23) GT4 treatment-naive without cirrhosis had SVR 12, 24 weeks treatment
- GT2, 89% (16/18) treatment-naive & 75% (3/4) treatment experienced without cirrhosis had SVR12, 12 weeks treatment
see charts below for full results

---------------------------------------------
Program abstract
Background: HIV/HCV co-infected patients require effective interferon-free HCV therapy that is tolerable and easy to administer in combination with antiretroviral therapy (ART). This study evaluated the safety and efficacy of the oral HCV NS5B inhibitor sofosbuvir with ribavirin in patients coinfected with HIV and HCV genotypes (GT) 1-4.
Methods: 274 patients infected with HIV and HCV GT1-4, including those with compensated cirrhosis, were enrolled to receive sofosbuvir 400 mg QD and weight-based ribavirin 1000-1200 mg/day; GT 1, 3, 4 treatment-naïve and GT 2, 3 treatment-experienced patients received 24 weeks of therapy and GT 2 treatment-naïve 12 weeks. The primary efficacy endpoint was sustained virologic response 12 weeks after treatment (SVR12); safety assessments also included HIV RNA and CD4 cell levels.
Results: Baseline characteristics and SVR12 rates are shown in the table. 20% of patients were cirrhotic, 81% were male, 97% were receiving ART. Median CD4 was >500 cell/μL in all groups. SVR12 rates ranged from 83% to 91% across GT1-4. Of 38 patients not achieving SVR12, 31 patients relapsed, 1 had breakthrough and 6 were lost to follow-up or withdrew consent. HCV resistance testing is ongoing. Treatment discontinuations from sofosbuvir due to adverse events (AEs) were observed in 5/274 (2%) of patients and grade 3/4 AEs were reported in 15/274 (6%). No change in CD4% was observed through SVR12. Four patients had an HIV-RNA rebound that did not require ARV modification.
Conclusions: HCV GT 1-4 treatment naïve and experienced HIV coinfected patients achieved high rates of SVR12 with 12 or 24 weeks of an interferon-free, oral regimen of sofosbuvir + ribavirin. Sofosbuvir + ribavirin treatment was well-tolerated and safely coadministered with multiple ART regimens.
--------------------------------------------






|
| |
|
 |
 |
|
|