 |
 |
 |
| |
HPV VACCINATION IN HIV+ ADOLESCENTS AND YOUNG ADULTS
INDUCES STRONG HPV-SPECIFIC CELL-MEDIATED IMMUNE RESPONSES
|
| |
| |
IAC: Quadrivalent HPV Vaccine Elicits Antibody and Cell Responses in HIV+ Teens, Young Adults - written by Mark Mascolini - (07/23/14)
Reported by Jules Levin
20th International AIDS Conference, July 20-25, 2014, Melbourne
Veronica Rainone, PhD Milano University Medical School Chair of Immunology Department of Biomedical and Clinical Sciences L. Sacco

-------------
Program Abstract
Background: Human Papilloma Virus (HPV)-associated ano-genital infections represent the most common sexually transmitted disease in the general population. The incidence of HPV-associated cancers has been increasing in HIV-infected patients. HPV vaccination may be an important approach to reduce the risk of HPV-associated cancers in HIV-infected patients and a combined strategy of screening and HPV vaccination may guarantee a more adequate prevention of HPV-associated lesions. Immunogenicity of HPV vaccines in HIV-infected patients is still not adequately evaluated. We analysed immunogenicity of a quadrivalent HPV vaccine in HIV-infected patients without baseline molecular evidence of vaccine-type HPV infection focusing on both HPV-specific cell-mediated (CMI) and humoral immunity.
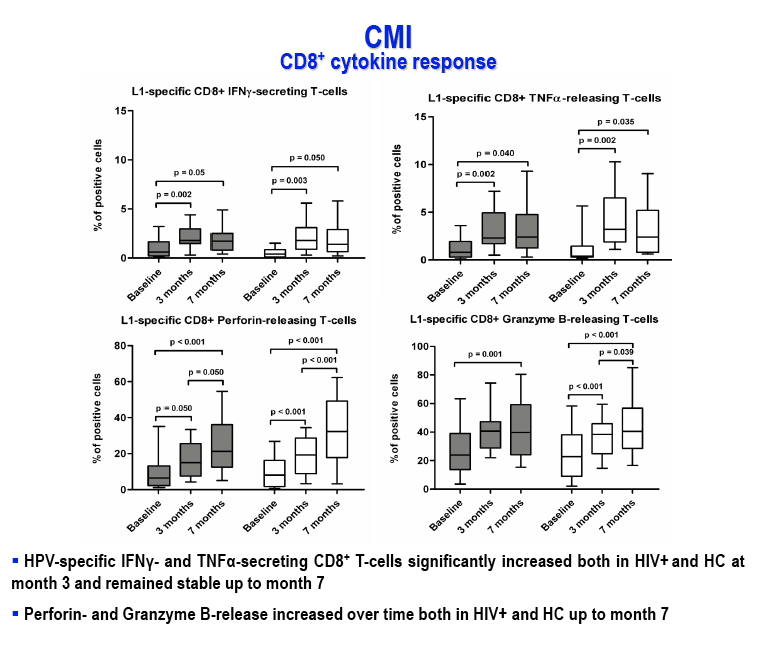
Methods: 31 ARV-treated HIV-infected adolescents (age range 28-14 years, mostly with undetectable viremia and effective CD4 recovery) and 25 sex- and age-matched HIV-seronegative healthy controls were enrolled in the study. Quadrivalent HPV-16/18/6/11 VLP vaccine (Gardasil®) was administered 3 times (baseline, 2 months and 6 months). Immune activation (CD4/CD25/HLADRII, CD8/CD25/HLADRII), naïve, effector and memory T-cell patterns, cellular immune responses (CD4/IFN-γ/IL-2, CD8/IFN-γ/TNF-α, CD8/Perforin/GranzymeB) and anti-HPV16/18/6/11 IgG titers were evaluated at baseline and after each immunization.
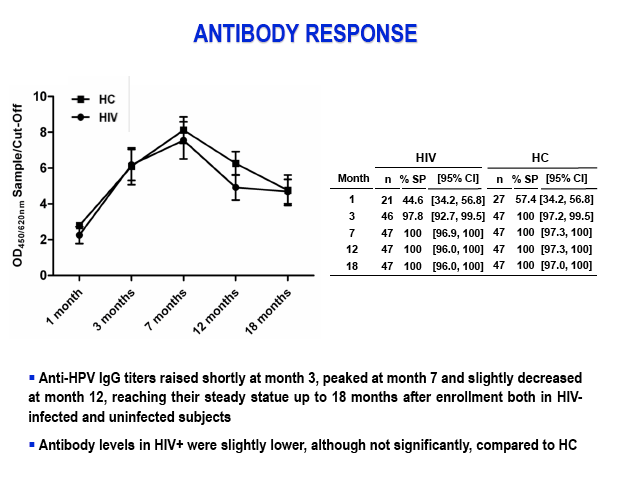
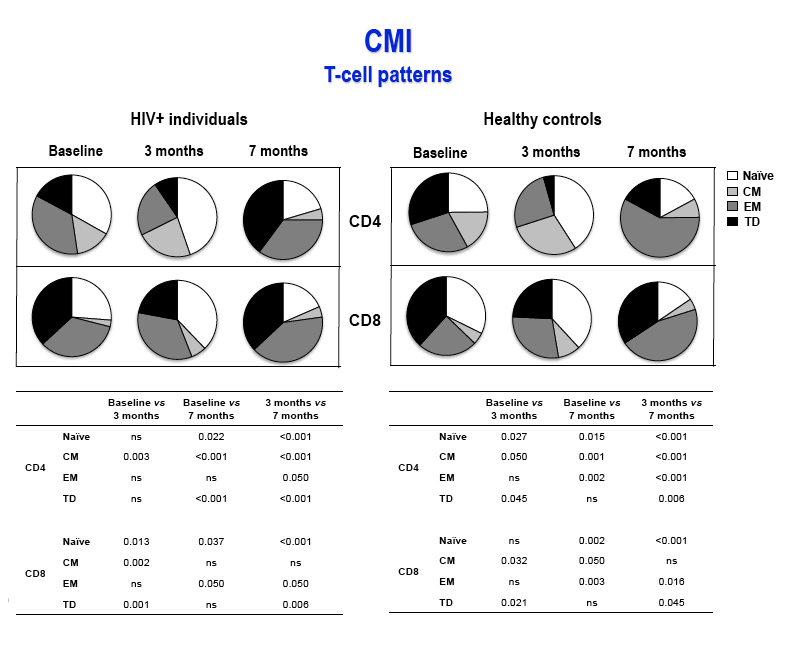
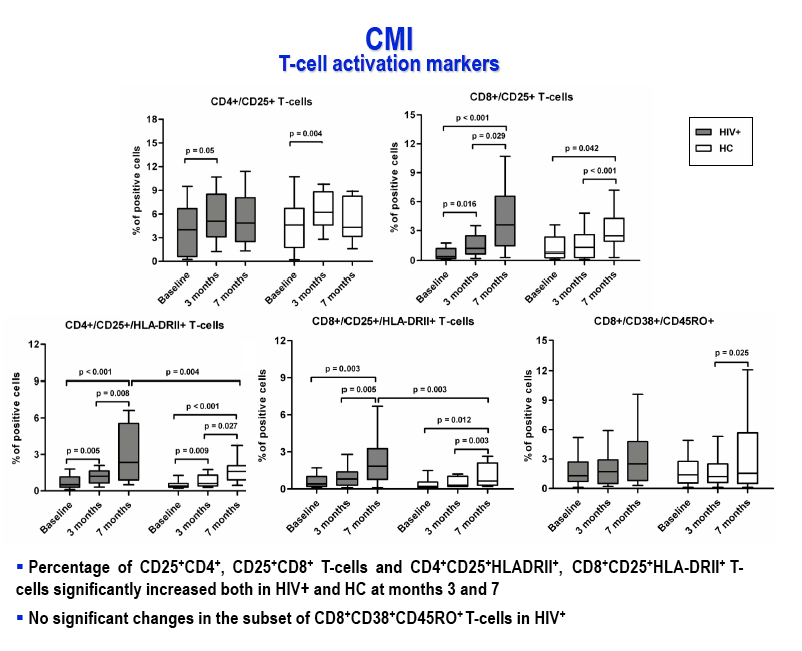
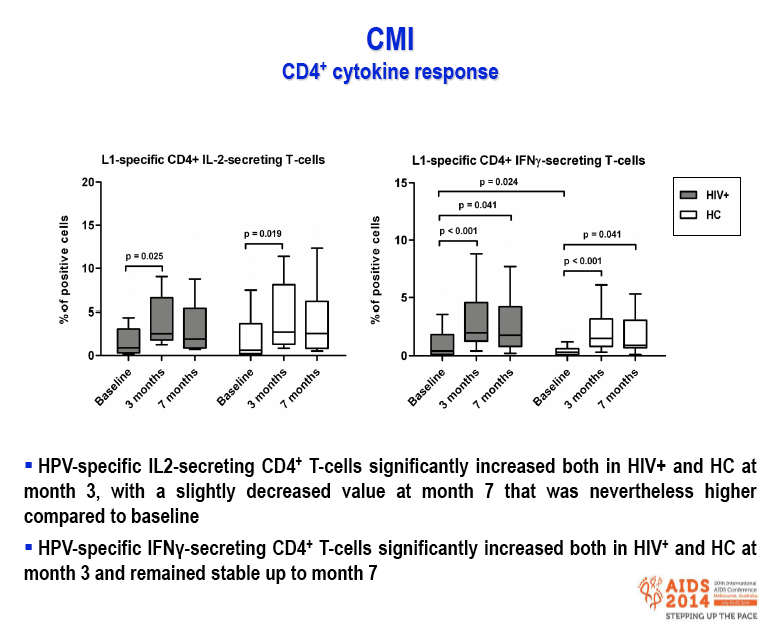
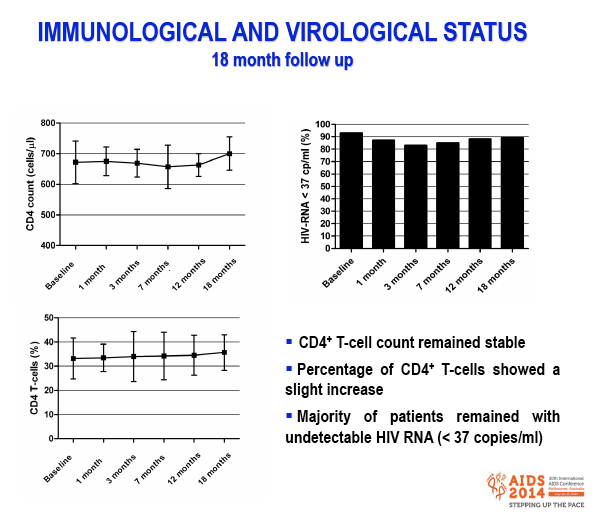
Results: After the third immunization HIV-infected individuals showed: 1) no changes in CD4 counts, percentage of CD4 cells and HIV viral load; 2) a significant increase in naïve CD8 T-cells, activated CD8 T-cells and in central memory CD4 and CD8 T-cells; 3) a significant reduction in terminally differentiated CD8 T-cells; 4) a significant increase in unstimulated and in HPV-specific IL2+/CD4+, IFN-γ+/CD4+, IFN-γ+/CD8+ and TNF-α+/CD8+ T-cells; 5) a significant increase in HPV-specific Perforin- and Granzyme B-secreting CD8 T-cells. High titers of anti-HPV16/18/6/11 IgG raised shortly after the first immunization and increased over time until the last immunization. Notably, results obtained in HIV-infected patients were comparable to those seen in HC.
Conclusions: Quadrivalent HPV-16/18/6/11 VLP vaccine induces strong HPV-specific cell-mediated and humoral immune responses in ARV-treated HIV-infected individuals that are comparable to those observed in HIV-seronegative controls.
HPV-specific CMI is likely an important component of the protective effect of this vaccine, beside the generation of a broad antibody response, data herein indicating that this arm of immunity is not impaired in ARV-treated HIV infected individuals.
-----------------





|
| |
|
 |
 |
|
|