 |
 |
 |
| |
Does the Addition of HCV Testing to a Rapid
HIV Testing Program Impact HIV Test Acceptance?
A Randomized Controlled Trial
|
| |
| |
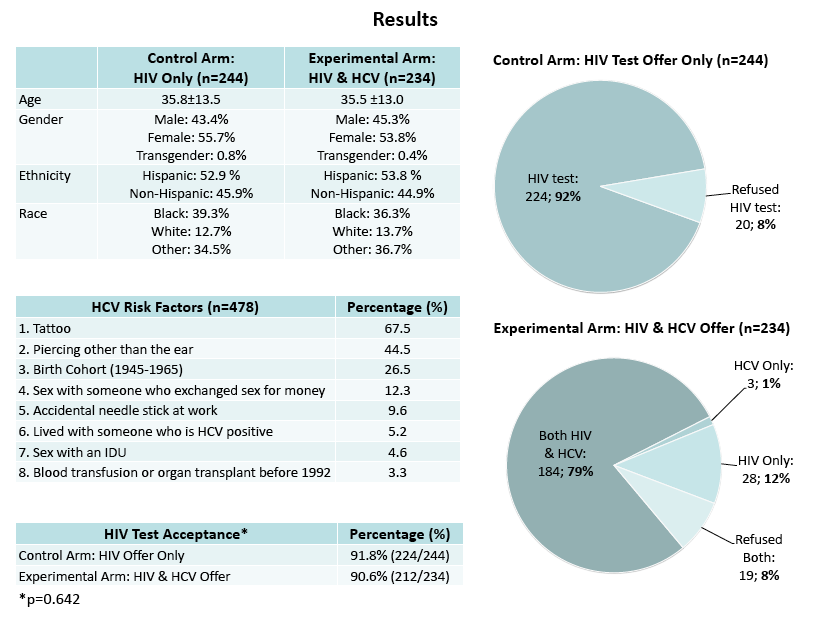
"There was no significant difference in HIV test acceptance between the HCV and HIV (91.8%, 224/244) and HIV-only (90.6%, 212/234) groups (p=0.642). .........Offering rapid HCV testing along with HIV testing did not significantly impact HIV test acceptance in an urban ED with an existing rapid HIV testing program. HCV risk and test acceptance were high. Future screening efforts for HCV could be integrated into current HIV testing models without negatively affecting HIV testing rates."
Reported by Jules Levin
20th International AIDS Conference, July 20-25, 2014, Melbourne
Yvette Calderon MD MS,1,2 Ethan Cowan MD MS,1,2 Mark Iscoe BA,1 Sara Rahman BA,1 John Y. Rhee BS,1 Lisa N. Glass MS BS,2 Mathew Barbery BA BS,2 Jason Leider MD PhD2,3 1Department of Emergency Medicine, Jacobi Medical Center, Bronx, NY, USA; 2Albert Einstein College of Medicine, New York, NY, USA; 3Department of Internal Medicine, Jacobi Medical Center, Bronx, NY, USA
 ------------------
------------------
Program Abstract
Background: Merging two critical public health screenings, HIV and Hepatitis C (HCV), could be crucial in identifying positive patients in high-risk urban settings. Considering the efforts that have been made in expanding HIV testing in the US, utilizing existing HIV models might be an effective way of identifying HCV. A new rapid, point-of-care HCV test opens the possibility of integrating HCV testing into current rapid HIV testing programs. The purpose of this study was to determine how offering HCV testing along with HIV testing in an urban emergency department (ED) would impact HIV test acceptance.
Methods: We conducted a 2-armed randomized controlled trial on a convenience sample of patients age 18 and above in a Bronx, New York ED. Participants were randomized to an offer of both HIV and HCV testing or HIV testing alone. The primary outcome, HIV test acceptance, was compared between the groups. Secondary outcomes included HIV and HCV prevalence and HCV test acceptance, refusal, risk, and knowledge.
Results: Of 666 eligible patients, 478 agreed to participate. There was no significant difference in HIV test acceptance between the HCV and HIV (91.8%, 224/244) and HIV-only (90.6%, 212/234) groups (p=0.642). There were also no significant differences in HIV test acceptance based on gender, race, or ethnicity. HCV test acceptance was high (79.9%, 187/234). Majority of participants (76.6%, 366/478) reported at least one HCV risk factor. No participants tested positive for HIV, and 1 (0.5%) tested positive for HCV. The participants were knowledgeable in recognizing the existence of latent HCV infection (74.3% correctly responded), 70.7% knew alcohol could damage the livers of those with HCV, 66.9% knew HCV could be sexually transmitted, 55.9% knew HCV was treatable, 45.8% knew HCV could be cured, and 43.9% knew there is no HCV vaccine.
Conclusions: Offering rapid HCV testing along with HIV testing did not significantly impact HIV test acceptance in an urban ED with an existing rapid HIV testing program. HCV risk and test acceptance were high. Future screening efforts for HCV could be integrated into current HIV testing models without negatively affecting HIV testing rates.
-------------------



|
| |
|
 |
 |
|
|