 |
 |
 |
| |
About 40% Who Get Under 50 Copies Have Residual Viremia in French Cohort
|
| |
| |
20th International AIDS Conference, July 20-25, 2014, Melbourne
Mark Mascolini
Among 168 antiretroviral-naive people starting one of three standard once-daily regimens, 85% to 89% had a viral load below 50 copies after 48 weeks of treatment in an observational French cohort [1]. But only 45% to 54% of the group had a viral load below 1 copy (an undetectable RNA signal) at week 48. Response rates did not differ substantially with the three regimens, which included tenofovir/emtricitabine plus atazanavir/ritonavir, darunavir/ritonavir, or efavirenz.
Today's first-line antiretroviral regimens usually push viral loads below 50 copies/mL within months. But viral load assays now detect HIV RNA at sub-50 levels--down to less than 1 copy/mL if the assay returns no viral signal. The clinical meaning of completely undetectable HIV RNA in plasma remains unknown, as does the relative ability of today's first-line regimens to achieve that goal. To start addressing those questions, researchers in Paris analyzed virologic and pharmacologic outcomes in the first 48 weeks of treatment among people starting an initial regimen with efavirenz or ritonavir-boosted atazanavir or darunavir, each with tenofovir/emtricitabine.
Everyone in this observational cohort began treatment between November 2010 and June 2012. To measure viral load, the investigators used the COBAS AmpliPrep/COBAS TaqMan assay, which has a lower detection limit of 20 copies. They considered the viral load below 1 copy when this assay could not detect a viral target. The investigators measured 24-hour antiretroviral concentrations at the same times they measured viral load--4, 12, 24, 36, and 48 weeks after treatment began.
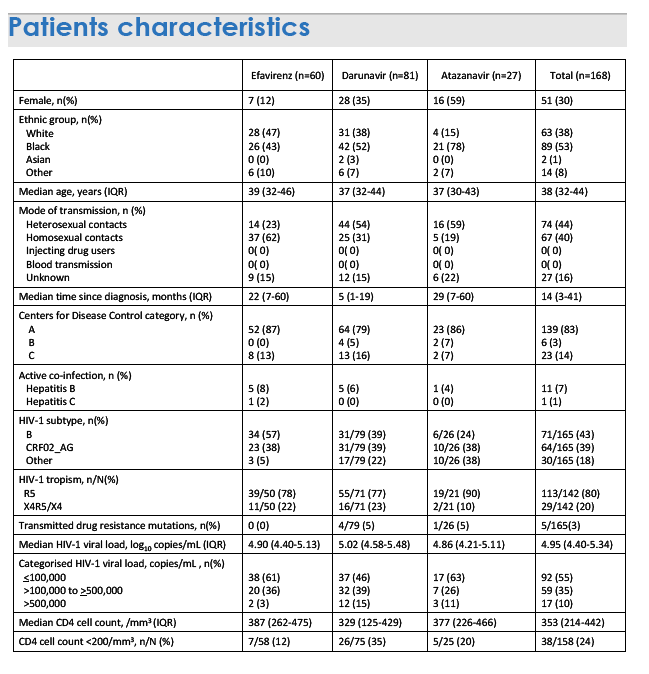
The analysis involved 60 people starting efavirenz (12% women), 81 starting darunavir (35% women), and 27 (59% women) starting atazanavir. Median ages of the three groups were 39, 37, and 37. Clinicians favored efavirenz for gay men (who made up 62% of efavirenz starters) and darunavir or atazanavir for heterosexuals (54% and 59% of those starting darunavir and atazanavir) (P < 0.0001). Median pretreatment viral loads were 4.9 log in the efavirenz group, 5.02 log in the darunavir group, and 4.86 log in the atazanavir group (about 79,000, 105,000, and 72,000 copies). Proportions starting treatment with a viral load above 100,000 copies were 39% with efavirenz, 54% with darunavir, and 36% with atazanavir (P = 0.022). Blacks made up 43% of those starting efavirenz, 52% of those starting darunavir, and 78% of those starting atazanavir (P = 0.006).
By treatment week 48, more than a twice higher proportion of people had stopped efavirenz (15%) than had stopped darunavir (6%) or atazanavir (4%). Most people who stopped any regimen did so for tolerance or simplification, with a viral load below 20 copies.
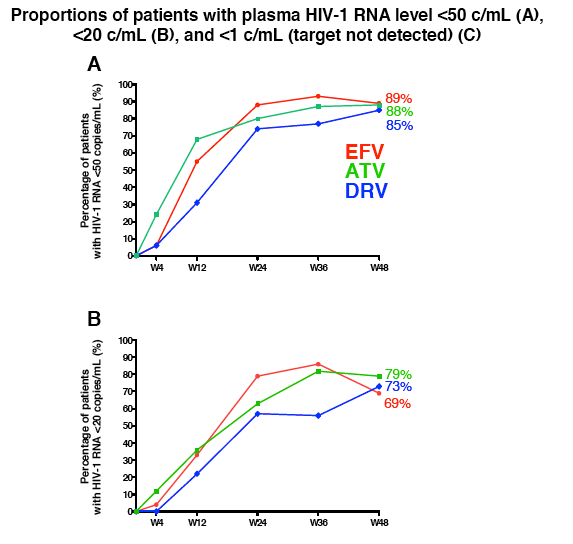
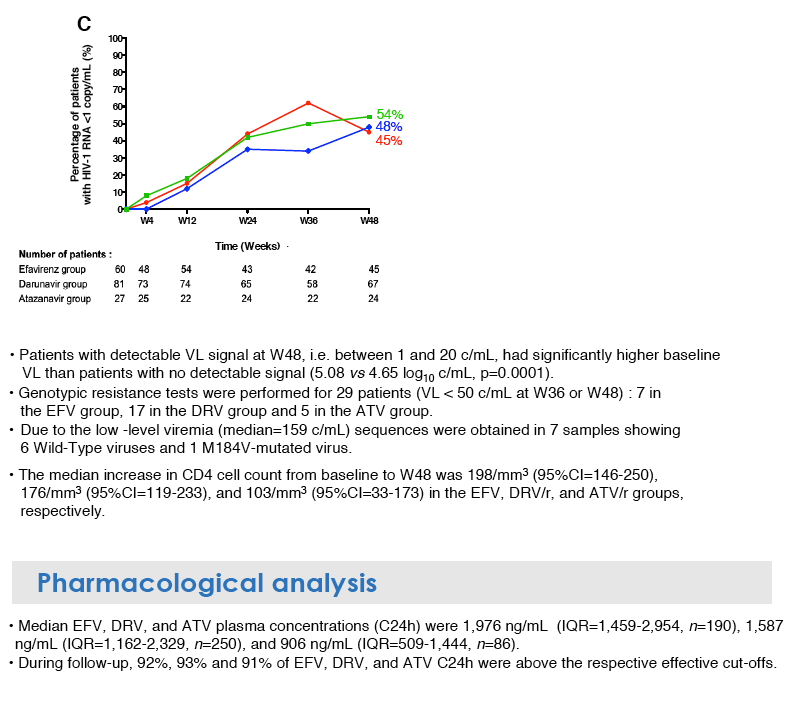
Similar proportions of people reached a viral load below 50 copies at week 48 after starting efavirenz (89%), atazanavir (88%) or darunavir (85%). Proportions reaching a load below 20 copies at week 48 were 69% with efavirenz, 79% with atazanavir, and 73% with darunavir. Proportions with less than 1 copy at week 48 were 45% on efavirenz, 54% on atazanavir, and 48% on darunavir. The drop in response rate from 50 copies to less than 1 copy was greatest with efavirenz (89% to 45%) but also substantial with atazanavir (88% to 54%) and darunavir (85% to 48%).
People who had a viral load of 1 to 20 copies at week 48 had a significantly higher pre-ART viral load than people with no detectable viral signal at week 48: 5.08 versus 4.65 log (about 120,000 versus 45,000 copies, P = 0.0001). Median CD4 gain through week 48 measured 198 with efavirenz, 176 with darunavir, and 103 with atazanavir.
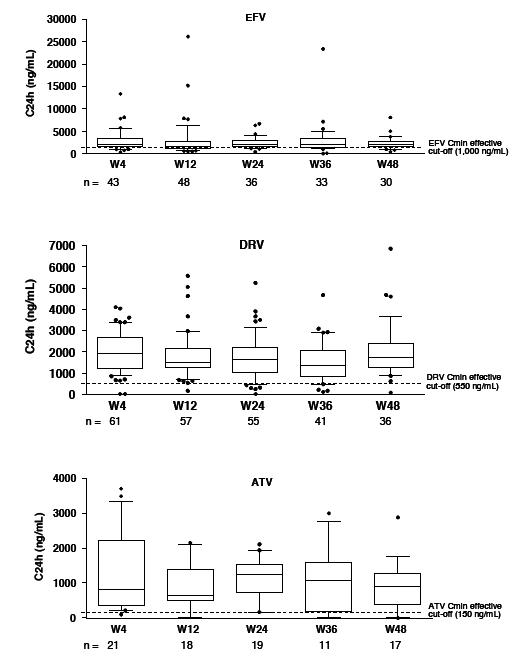
Throughout follow-up, 92% taking efavirenz, 93% taking darunavir, and 91% taking atazanavir had drug levels above the minimum effective concentrations for those antiretrovirals. Interpatient variability in drug concentrations was greatest for darunavir (171%), followed by atazanavir (133%) and efavirenz (91%). Intrapatient variability was 55% with atazanavir, 41% with darunavir, and 29% with efavirenz.
The investigators noted that virologic response rates in this cohort are similar to results of randomized trials. They stressed that people who started ART with a viral load above 100,000 copies ran a greater risk of residual viremia than people who started with a lower viral load.
Reference
1. Charpentier C, Choquet M, Joly V, et al. Virological outcome at week 48 of three recommended first-line regimens using ultra-sensitive viral load and drug concentrations level. AIDS 2014. 20th International AIDS Conference. July 20-25, 2014. Melbourne. Abstract WEPE043.




|
| |
|
 |
 |
|
|